The US is about to kick-start its controversial covid booster campaign
There are three groups of Americans now eligible for a booster shot—but many experts say that the doses would be better off being used elsewhere
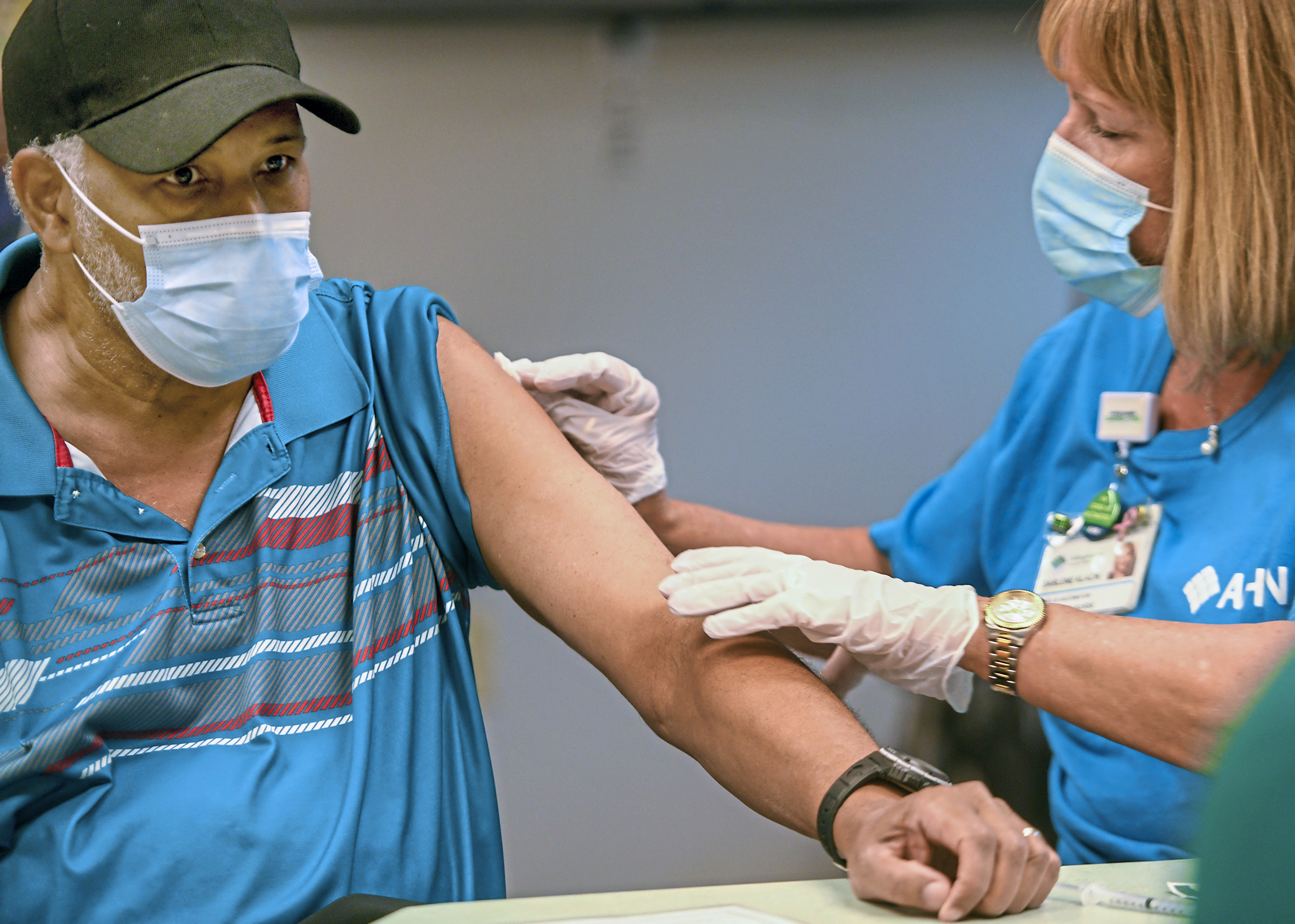
The news: The White House is set to kick off its booster shot campaign today, after Centers for Disease Control and Prevention director Rochelle Walensky overruled her own agency’s advisors in favor of recommending third doses of the Pfizer-BioNTech vaccine for frontline workers.
Who gets it: There are three groups of Americans now eligible for a booster shot: those 65 and older, some adults with underlying health conditions, and people at higher risk of exposure because of their jobs. The CDC’s advisory panel had only recommended booster shots for the first two categories, but Walensky disagreed and added the third, justifying her decision by pointing to the fact it aligns with an FDA ruling released earlier this week.
The shots should be given at least six months after the second dose, the CDC recommended. It has yet to assess either the Moderna or Johnson & Johnson vaccines—but these decisions are expected to come soon
The Biden administration has been keen to offer booster shots to as wide a population as possible, with plans drawn up to offer them to all Americans over the age of 16, but there’s little scientific evidence to support such a move. Despite some evidence that post-vaccine immunity wanes over time, the vaccines seem just as effective as ever at keeping people alive and out of hospitals. More than 2 million immunocompromised Americans have already had a booster.
Disagreements: Booster shots have been controversial. A group of top scientists, including experts at the FDA and WHO, published a review in The Lancet on Monday arguing that booster shots are unnecessary since vaccines are still very effective at preventing severe disease and death. Furthermore, they say, vaccine supplies could save more lives if they’re used for unvaccinated people rather than as boosters for the vaccinated. That’s why the WHO has been pleading with rich countries to stop handing them out until more of the world is vaccinated.
Unequal distribution: The US joins the UK, the UAE, France, Germany, and Israel, which have also launched booster programs. In the UK, for example, a rollout of booster shots to all over-50s is about to begin after officials gave the green light last week. Meanwhile, less than 4% of Africa’s population is fully vaccinated, compared with 70% of adults in the EU. In the US, it's 55%, a figure that has stubbornly failed to significantly budge in recent weeks. Earlier this week, President Biden announced that the US would buy a further 500 million doses of vaccine to distribute to other parts of the world, bringing its total commitment to more than 1 billion.
Scramble: Millions of Americans are likely to try to get a third shot. A YouGov poll this summer found that three in five vaccinated Americans will get one if it’s available. Given the chaotic nature of the US vaccine rollout, it will be hard to prevent people from gaming the system to get a third shot even if they aren't technically eligible.
Deep Dive
Biotechnology and health
How scientists traced a mysterious covid case back to six toilets
When wastewater surveillance turns into a hunt for a single infected individual, the ethics get tricky.
An AI-driven “factory of drugs” claims to have hit a big milestone
Insilico is part of a wave of companies betting on AI as the "next amazing revolution" in biology
The quest to legitimize longevity medicine
Longevity clinics offer a mix of services that largely cater to the wealthy. Now there’s a push to establish their work as a credible medical field.
There is a new most expensive drug in the world. Price tag: $4.25 million
But will the latest gene therapy suffer the curse of the costliest drug?
Stay connected
Get the latest updates from
MIT Technology Review
Discover special offers, top stories, upcoming events, and more.