A medical app uses your smartphone’s camera to carry out lab tests at home
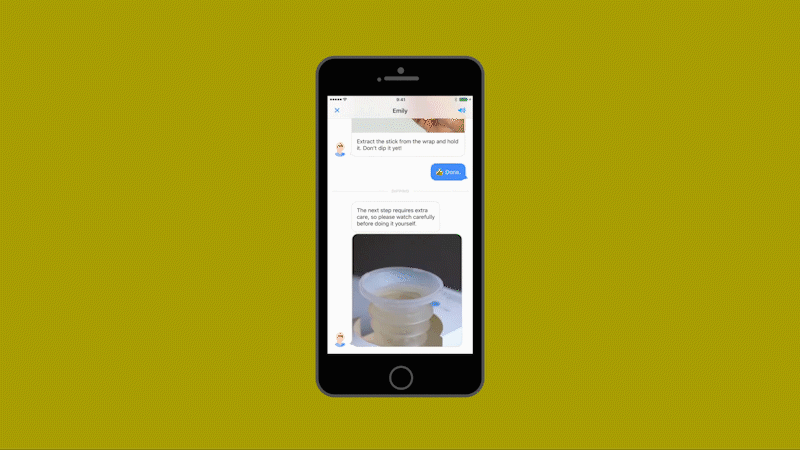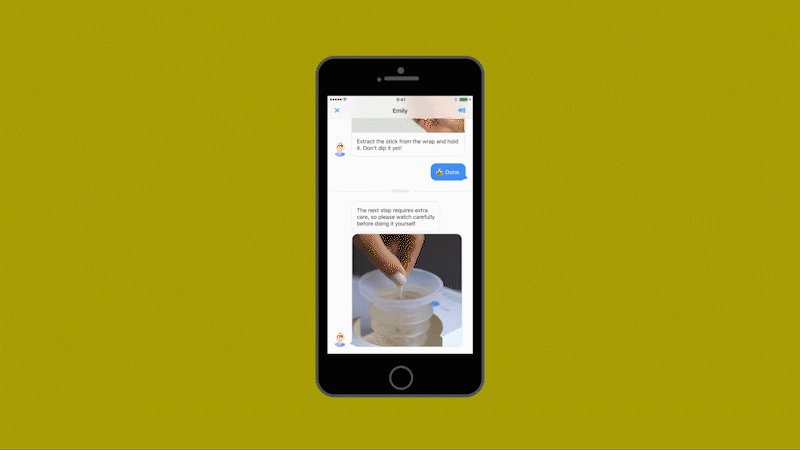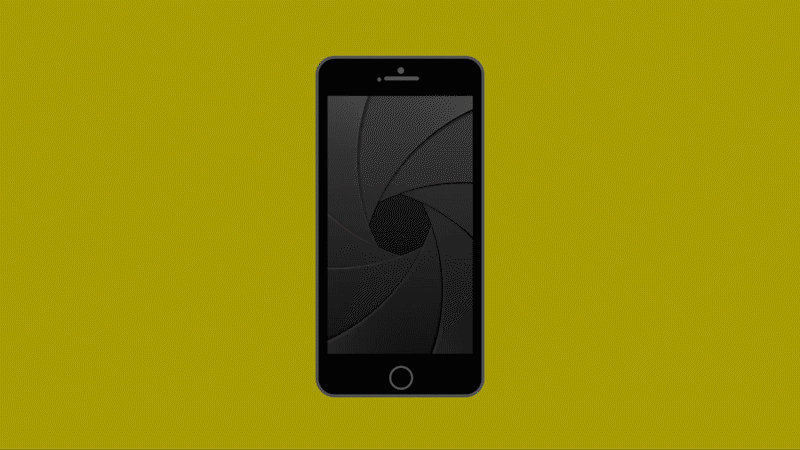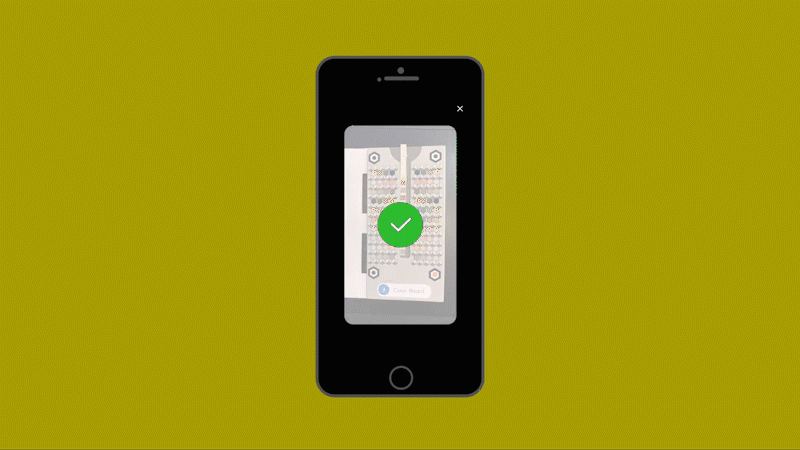
Millions of laboratory urine tests are carried out each year for pregnant women, patients at risk of kidney failure, and people who think they may have urinary tract infections. The tests are expensive and time consuming for patients and medical practitioners alike, involving a visit to a physician, a referral to a lab, and up to three days waiting for the results.
This inconvenience deters people from annual screening that could save them dialysis or even a transplant by detecting early signs of renal failure. At present, only 40% of at-risk patients in the UK respond to reminders to visit their doctor for an annual urine test, while the response rate in the US is even lower, around 30%.
The first commercial deployment of a system that transforms an ordinary smartphone camera into a laboratory-grade scanning device could help. A home urine-testing kit developed by Tel Aviv–based Healthy.io could save health providers hundreds of millions of dollars a year by detecting disease in time to prevent kidney failure. Dip.io, as it’s called, should also save pregnant women and those with UTIs time and discomfort with instant analysis, avoiding the need for a doctor’s appointment. It is the first smartphone-based system to receive approval from the US Food and Drug Administration as a Class II clinical-grade diagnostic device.
Engagement boost
Today, the UK’s National Health Service announced that its first commercial rollout—with more than 2,000 diabetic patients in the Hull region of northeast England—raised response rates to 72% among patients who previously had failed to respond to reminders. Of these, 10% were found to have elevated levels of protein in their urine, an early sign of chronic kidney disease that, left undiscovered, would necessitate either dialysis or transplant in about five years.
Healthy.io’s system uses a standard smartphone camera and a dipstick for urine tests that can identify 10 separate indicators of disease, infection, and pregnancy-related complications. The patient takes a photo of the dipstick against a color card. The app uses machine learning to correct the color, taking into account the type of camera, the lighting conditions, and numerous other variables (including whether the patient has dipped the stick for the recommended two seconds or longer.) The app then gives an instant analysis. In clinical trials as part of the FDA’s approval process, the system’s accuracy matched the results of a professional laboratory—at a fraction of the time and cost and at minimal inconvenience to the patient.
To boost engagement, Healthy.io uses behavioral algorithms of the kind developed by Facebook and Google. At-risk patients who hadn’t shown up for the test in 18 months were contacted with an automated phone call and invited do the test at home. About a quarter took the test in the first week; the rest were prompted with nudges based on age, where they live, and even the type of smartphone they have. These boosted compliance to around 70%.
Patients liked the speed and convenience of the service, with benefits including fewer appointments, less travel, and reduced anxiety about supplying samples, says Rachel Barnes, a nurse and leader of research and clinical development at the Hull branch of Modality, an NHS GP super-practice that used the kits with its patients. “There were diabetic patients in our pilot cohort that we knew we had never seen in the surgery whose results came back as abnormal,” she says.
More than 1.8 million people have been diagnosed with chronic kidney disease in England, and it’s estimated that a million more have the disease but are not yet diagnosed, says Yonatan Adiri, founder and CEO of Healthy.io. In the US, about 30 million adults are believed to be affected, but most are not aware of it.
If adopted nationwide in the UK alone, the results indicate, the technology could lead to early diagnosis of more than 33,000 cases over five years, prevent more than 1,300 deaths, and allow more than 11,000 people to avoid dialysis or transplant—with estimated savings of more than £670 million ($867 million) over the same period, according to an analysis by York Health Economics Consortium.
An earlier trial involving nearly 1,000 people in the US, carried out by Healthy.io with the National Kidney Foundation and health-care provider Geisinger, found a similar 72% compliance rate and proved popular with patients.“This new test has the potential to help millions of patients find out they have chronic kidney disease while there is still time to prevent progression to kidney failure,” says Kerry Willis, the foundation’s chief scientific officer.
Following its success in Hull, Healthy.io is planning to roll out the system across the NHS and extend it to hypertension and maternity care patients. Meanwhile, a pilot program is under way with the pharmacist Boots UK. This enables women who suspect they have urinary tract infections to self-test and immediately get treatment from a pharmacist without seeing a doctor or waiting for lab results. The final results of that program will be announced in May.
DIY testing, without the hassle
Several other companies are also developing urine tests based on smartphone cameras, including Testcard in the UK and Scanwell in the US, which has Class I FDA approval for a two-parameter dipstick that identifies urinary tract infections.Following Apple’s announcement that it will be placing a 3D sensor on the back of its phones, Adiri says Healthy.io is already working on new applications for its technology, including measurement of chronic wounds. The firm says it has received orders from medical providers for commercial use by 100,000 patients in Israel, the UK, and the Netherlands over the next six months.
“By democratizing laboratory technologies, as long as the quality control is good, it’s very important,” says John Halamka, a professor at Harvard Medical School and chief information officer at Beth Israel Deaconess Medical System in Boston. Still, he says, “we want to make sure those tests are interpreted by a professional when you get a result.”
Nigel Hughes, scientific director at Janssen Clinical Innovation in Belgium, also warns that such technology needs regulatory oversight. “You can’t mess around with this,” Hughes says. “Breaking things quickly and learning fast has worked in other industries, but not when you’re putting a patient’s life at risk. It takes time.”
Deep Dive
Biotechnology and health
How scientists traced a mysterious covid case back to six toilets
When wastewater surveillance turns into a hunt for a single infected individual, the ethics get tricky.
An AI-driven “factory of drugs” claims to have hit a big milestone
Insilico is part of a wave of companies betting on AI as the "next amazing revolution" in biology
The quest to legitimize longevity medicine
Longevity clinics offer a mix of services that largely cater to the wealthy. Now there’s a push to establish their work as a credible medical field.
There is a new most expensive drug in the world. Price tag: $4.25 million
But will the latest gene therapy suffer the curse of the costliest drug?
Stay connected
Get the latest updates from
MIT Technology Review
Discover special offers, top stories, upcoming events, and more.