A new way to make steel could cut 5% of CO2 emissions at a stroke

A lumpy disc of dark-gray steel covers a bench in the lab space of Boston Metal, an MIT spinout located a half-hour north of its namesake city.
It’s the company’s first batch of the high-strength alloy, created using a novel approach to metal processing. Instead of the blast furnace employed in steelmaking for centuries, Boston Metal has developed something closer to a battery. Specifically, it’s what’s known as an electrolytic cell, which uses electricity—rather than carbon—to process raw iron ore.
If the technology works at scale as cheaply as the founders hope, it could offer a clear path to cutting greenhouse-gas emissions from one of the hardest-to-clean sectors of the global economy, and the single biggest industrial source of climate pollution.
After working on the idea for the last six years, the nine-person company is shifting into its next phase. If it closes a pending funding round, the startup plans to build a large demonstration facility and develop an industrial-scale cell for steel production.
Cutting carbon
In the main approach to steelmaking today, iron oxide is placed into a blast furnace with coke, a hard, porous substance derived from coal. Under high temperatures, the coke turns into carbon monoxide, which plucks oxygen off the iron, producing an intermediate metal known as “pig iron”—along with carbon dioxide that wafts into the atmosphere.
This and other steps in the process pump around 1.7 gigatons of carbon dioxide into the atmosphere annually, adding up to around 5 percent of global carbon dioxide emissions, according to a recent paper in Science (see “We still have no idea how to eliminate more than a quarter of energy emissions”). And that’s before taking into account the fuels required to fire the furnaces.
“Cars, buildings, and bridges are all pretty well dependent on having steel,” says Steven Davis, lead author of that study and an earth system scientist at the University of California, Irvine. “So unless we change that—and there’s no sign we are—we need to figure out a way to decarbonize the process.”
But completely eliminating emissions will require either technology that can capture carbon dioxide before it leaves the steel mill, which is both expensive and technically fraught, or the use of alternative materials to remove oxygen from iron oxide.
Moonshot
Donald Sadoway, a chemist at MIT, began inadvertently working on a solution in the mid-2000s.
NASA had offered a quarter-million-dollar prize to the first research team that could figure out how to extract oxygen from the moon’s surface, a precondition for establishing lunar bases. Sadoway proposed using an electrolytic cell—which produces an electric current to break down compounds—to extract oxygen from lunar rocks. The by-product was molten metal, a realization that led him to explore the possibility of using a similar approach to process metals back on Earth.
But producing something like steel would require an anode made from cheap materials that wouldn’t corrode under high temperatures or readily react with iron oxide. In 2013, Sadoway and MIT metallurgy researcher Antoine Allanore published a paper in Nature concluding that anodes made from chromium-based alloys might check all those boxes.
The previous year, Sadoway, Allanore, and another partner had cofounded a startup known as Boston Electrometallurgical, which would become Boston Metal.
The company has raised $13 million to date, primarily from Brazilian investor Ingo Wender, the US Department of Energy, and the National Science Foundation. The firm declined to disclose the source of a pending investment until that transaction is finalized.
A hesitant industry
One of its electrolytic cells sits in the back room of Boston Metal. It’s a squat metal cylinder, with a chimney-like tube emerging from the top and an ovular opening in front. It’s designed to produce ferroalloys, a high-margin material used to produce certain grades of steel— and the startup’s initial target market.
The “chimney” is actually an anode. A thin layer of metal along the bottom forms the cathode. These positive and negative electrodes together act like a sort of pump, pushing electrons through the electrolyte in the chamber, a mix of metallic minerals and other oxides.
The precise ingredients in that electrolyte are a critical part of the company’s core technology. In the case of steel, the other oxides act as a solvent at high temperatures, dissolving the iron oxide without decomposing themselves.
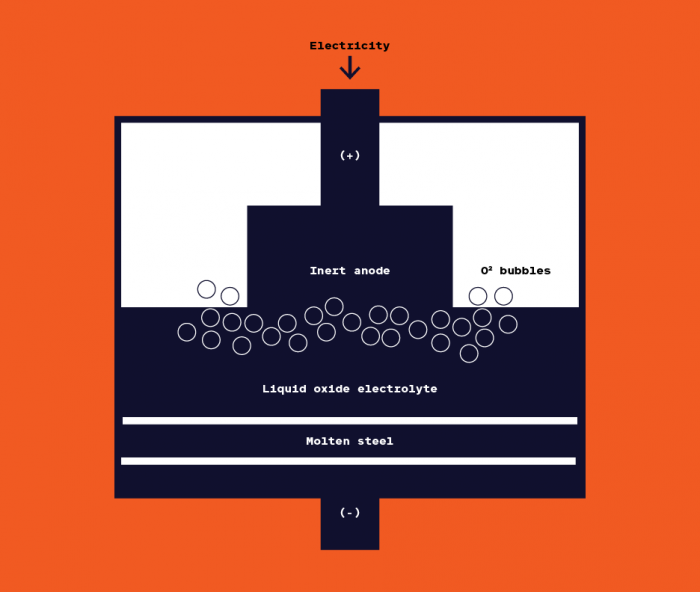
As the electric current heats up that soup, oxygen freed from the iron bubbles up to the top, and the resulting metal accumulates at the bottom. Once operators “tap,” or crack through, a lining through the hole in the front, molten metal pours forth in a blazing orange line.
An upside to using carbon in steelmaking is that it imparts strength to the final product, at least when it’s in the right proportion. Adam Rauwerdink, the company’s director of strategy, said in an e-mail that in order to produce specific grades of steel, carbon and other ingredients can simply be added in as the metal cools.
But some wonder how convincing that will be to the notoriously conservative steel industry, which after all is producing a material that needs to hold up skyscrapers.
“Carbon is required at some level for the mechanical properties of the steel itself,” said Nathan Lewis, a chemist at the California Institute of Technology, in an e-mail. “And understandably, the industry is hesitant to change that process.”
Next steps
Last year, Tadeu Carneiro joined Boston Metal as chief executive officer. He previously led Brazil-based CBMM, the largest producer of niobium, a metal used in superconducting alloys.
Carneiro, who wears a pair of dark, heavy glasses above a set of dark, heavy eyebrows, scribbles chemical equations on a whiteboard as he describes the company’s business strategy in a Brazilian accent. The three-year plan is to build a demonstration facility to produce ferroalloys, he says. Meanwhile, the company will also start designing and engineering a full-scale cell for producing steel, a milepost he hopes to reach within seven years.
If Boston Metal achieves these benchmarks, it could pursue a number of business models, including licensing the technology, partnering with metal manufacturers, selling cells, or producing metals directly.
But as always in science, until the product is actually built and tested at commercial scale, it’s too early to say how well or affordably it will really work. Moreover, merely producing a green version of a product for around the same price won’t be enough to transform the industry, given the billions of dollars in sunk costs in steel mills that can operate for decades—and the fact that much of this industry is based in relatively poor nations or struggling financially.

“Even if the perfect technology came on the scene today, it’d probably be several decades before we could effectively transition to it,” Davis says.
Carneiro acknowledges that the company still must surmount a number of remaining technical challenges, including improving the “faradaic efficiency,” which means increasing the percentage of electrons that actually produce metal; enhancing thermal efficiency, or reducing the kilowatt-hours of electricity required to produce a given volume of metal; and scaling up a chromium alloy anode that’s only been demonstrated at the lab level to date.
He’s confident, however, that Boston Metal will solve these challenges, prove the technology is cheaper, and ultimately convince the industry to adopt its approach.
Transforming the nearly $1 trillion steel sector is still a long shot. But if the startup can demonstrate its process works at an industrial scale, it could at least provide a small hope for solving one of the hardest parts of the climate puzzle.
Deep Dive
Climate change and energy
The problem with plug-in hybrids? Their drivers.
Plug-in hybrids are often sold as a transition to EVs, but new data from Europe shows we’re still underestimating the emissions they produce.
Harvard has halted its long-planned atmospheric geoengineering experiment
The decision follows years of controversy and the departure of one of the program’s key researchers.
Why hydrogen is losing the race to power cleaner cars
Batteries are dominating zero-emissions vehicles, and the fuel has better uses elsewhere.
Decarbonizing production of energy is a quick win
Clean technologies, including carbon management platforms, enable the global energy industry to play a crucial role in the transition to net zero.
Stay connected
Get the latest updates from
MIT Technology Review
Discover special offers, top stories, upcoming events, and more.