First Gene-Edited Dogs Reported in China
Man’s best friend is now his newest genetic engineering project.

Scientists in China say they are the first to use gene editing to produce customized dogs. They created a beagle with double the amount of muscle mass by deleting a gene called myostatin.
The dogs have “more muscles and are expected to have stronger running ability, which is good for hunting, police (military) applications,” Liangxue Lai, a researcher with the Key Laboratory of Regenerative Biology at the Guangzhou Institutes of Biomedicine and Health, said in an e-mail.
Lai and 28 colleagues reported their results last week in the Journal of Molecular Cell Biology, saying they intend to create dogs with other DNA mutations, including ones that mimic human diseases such as Parkinson’s and muscular dystrophy. “The goal of the research is to explore an approach to the generation of new disease dog models for biomedical research,” says Lai. “Dogs are very close to humans in terms of metabolic, physiological, and anatomical characteristics.”
Lai said his group had no plans breed to breed the extra-muscular beagles as pets. Other teams, however, could move quickly to commercialize gene-altered dogs, potentially editing their DNA to change their size, enhance their intelligence, or correct genetic illnesses. A different Chinese Institute, BGI, said in September it had begun selling miniature pigs, created via gene editing, for $1,600 each as novelty pets.
The Chinese beagle project was led by Lai and Gao Xiang, a specialist in genetic engineering of mice at Nanjing University. The dogs are being kept at the Guangzhou General Pharmaceutical Research Institute, which says on its website that it breeds more than 2,000 beagles a year for research. Beagles are commonly used in biomedical research in both China and the U.S.
Genome editing refers to newly developed techniques that let scientists easily disable genes or rearrange their DNA letters. The method used to change the beagles, known as CRISPR-Cas9, is particularly inexpensive and precise (see “10 Breakthrough Technologies 2014: Genome Editing”).
Last month, Duanqing Pei, a representative of the Chinese Academy of Sciences, highlighted Lai’s work as part of what he called a large Chinese effort to modify animals using CRISPR. The list of animals already engineered using gene editing in China includes goats, rabbits, rats, and monkeys. Pei described the efforts as a national scientific priority and part of China’s effort to establish world-class research.
The ease with which gene-editing can be carried out has raised worries that humans could be next (see “Engineering the Perfect Baby”). Those fears were stoked in April when another Chinese team reported altering human embryos in the laboratory in an attempt to correct a genetic defect that causes beta-thalassemia (see “Chinese Team Reports Gene-Editing Human Embryos”).
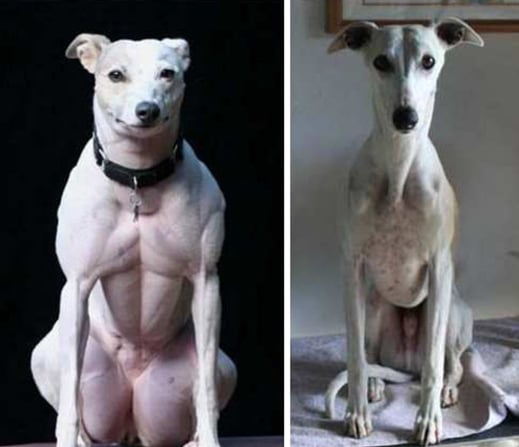
The dog researchers took much the same approach, directly introducing the gene-editing chemicals—a DNA snipping enzyme, Cas9, and a guide molecule that zeroes in to a particular stretch of DNA—into more than 60 dog embryos. Their objective was to damage, or knock out, both copies of the myostatin gene so that the beagles’ bodies would not produce any of the muscle-inhibiting protein that the gene manufactures.
In the end, of 65 embryos they edited, 27 puppies were born, but only two, a female and a male, had disruptions in both copies of the myostatin gene. They named the female Tiangou, after the “heaven dog” in Chinese myth. They named the male Hercules.
Lai and his colleagues reported that in Hercules, the gene editing was incomplete, and that a percentage of the dog’s muscle cells were still producing myostatin. But in Tiangou, the disruption of myostatin was complete and the beagle “displayed obvious muscular phenotype,” or characteristics. For example, her thigh muscles were large compared to those of her littermates.
The effects of losing the myostatin gene are well known from nature. One breed of ultra-beefy cattle called Belgian Blues normally lack the gene and grow to hulking size. Among dogs, the mutation occurs naturally only in whippets, says Eva Engvall, a retired scientist and whippet breeder who in 2007 helped identify the mutation affecting that breed. The double-muscled dogs are called “bully whippets.”
In rare cases, a person can also be born without any working copy of myostatin. In 2004 doctors reported a newborn who “appeared extraordinarily muscular, with protruding muscles in his thighs and upper arms.” They confirmed he was missing the myostatin gene and noted that by four and a half years of age, the boy could extend his arms while holding three-kilogram dumbbells.
Because the myostatin gene is well-studied—and because double-muscling isn’t known to have obvious drawbacks—it is frequently cited in debates over hypothetical future “gene-doping” among athletes. U.S. doctors are already attempting to block myostatin in gene-therapy experiments that seek to slow muscle loss in boys suffering from Duchenne muscular dystrophy.
Engvall says she was impressed by the Chinese work, which she says shows how gene editing might also be used to correct genetic illnesses that affect some dog breeds. “The point of this work was not to re-create myostatin knockouts,” says Engvall, “The point was to use the CRISPR technology in dogs. And in this project, the authors overcame some obstacles, in that dogs are a bit tricky when it comes to manipulating embryos.”
However, Eliane Ostrander, a scientist at the National Institutes of Health who has studied myostatin in dogs, says it remains unclear to her, based on the publication, whether the beagles were displaying extremely noticeable increases in muscle mass, as is seen in whippets. “The number of dogs is still small,” she says. “It will be interesting to see what types of variation come up as more dogs undergo the process.”
When DNA alterations are introduced at the embryo stage, they can affect every cell in an animals’ body, including sperm and egg, the cells of the “germline.” Lai’s team says it checked Hercules’s sperm and found that, if he was bred, he would pass the myostatin mutation onward. “The favorable traits that result from gene editing can pass generation by generation,” says Lai, and it will be possible to breed large numbers of gene-edited dogs “which can be commercialized.”
People have been influencing the genetics of dogs for millennia. By at least 36,000 years ago, early humans had already started to tame wolves and shape the companions we have today. Charles Darwin frequently cited dog breeding in The Origin of Species to demonstrate how evolution gradually occurs by a process of selection. With CRISPR, however, evolution is no longer gradual or subject to chance. It is immediate and under human control.
It is precisely that power that is stirring wide debate and concern over CRISPR. Yet at least some researchers think that gene-edited dogs could put a furry, friendly face on the technology. In an interview this month, George Church, a professor at Harvard University who leads a large effort to employ CRISPR editing, said he thinks it will be possible to augment dogs by using DNA edits to make them live longer or simply make them smarter.
Church said he also believed the alteration of dogs and other large animals could open a path to eventual gene editing of people. “Germline editing of pigs or dogs offers a line into it,” he said. “People might say, ‘Hey, it works.’ ”
Deep Dive
Biotechnology and health
How scientists traced a mysterious covid case back to six toilets
When wastewater surveillance turns into a hunt for a single infected individual, the ethics get tricky.
An AI-driven “factory of drugs” claims to have hit a big milestone
Insilico is part of a wave of companies betting on AI as the "next amazing revolution" in biology
The quest to legitimize longevity medicine
Longevity clinics offer a mix of services that largely cater to the wealthy. Now there’s a push to establish their work as a credible medical field.
There is a new most expensive drug in the world. Price tag: $4.25 million
But will the latest gene therapy suffer the curse of the costliest drug?
Stay connected
Get the latest updates from
MIT Technology Review
Discover special offers, top stories, upcoming events, and more.