Choosing the Good Eggs
By watching the tiny, pulsing motions of a newly fertilized mouse egg, researchers in a new study could determine which eggs stood the best chance of producing healthy mice. The same procedure should also work with human eggs, the researchers said yesterday, opening up the possibility of dramatically improving the success rates of in-vitro fertilization.
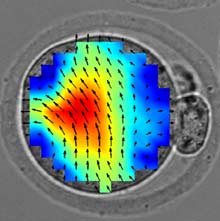
In a paper in the current issue of Nature Communications, the researchers found that the insides of a newly fertilized egg slowly vibrate, and that the speed and direction of these movements was associated with the egg’s likelihood of success.
Using high-speed photography, the researchers could predict within hours of fertilization whether an egg would be viable—the fastest and earliest method ever devised, says the paper’s senior author, Magdalena Zernicka-Goetz, of the University of Cambridge. The method is potentially safer than current techniques, which require removing cells from the developing embryo to determine their viability.
The researchers have not tried this approach yet with human eggs, but say there’s no reason it wouldn’t work. “The same type of movements do happen in human eggs upon fertilization—we checked it,” Zernicka-Goetz said at a news conference. “We are in the process of discussing with an in-vitro fertilization clinic in the U.K. to initiate such tests on human embryos.”
Doctors have long sought ways to judge whether an embryo is worth implanting. This is particularly important in the United States because insurance often doesn’t cover in-vitro fertilization, and many prospective parents can’t afford the costly procedure more than once or twice, says Andrew La Barbera, scientific director for the American Society for Reproductive Medicine.
Doctors often implant multiple embryos to increase a woman’s chances of a successful pregnancy. But this also increases the chances of twins and other multiple births, which are riskier for the babies.
By using a rapid-speed camera, the researchers could see that the cytoplasm—the liquid surrounding the cell’s organelles—began to move more rapidly after fertilization than before. That pulsing continued for about four hours, though it waxed and waned in three distinct stages. The direction of the movement also changed during these periods. The vibrations slowed once a cell nucleus was formed.
La Barbera agrees that human embryos likely show similar movements, and says he found the paper elegant and novel. But he doubts that this kind of test could be as predictive in human eggs as the Cambridge researchers suggest it is in mice. Human couples have far more genetic diversity than the inbred mice used in the research, La Barbera says, and older women can have defects in their eggs that might not be visible with this technique.
“This study has great promise,” he says. “It remains to be seen how well these results can be translated into humans.”
Janice Evans, an associate professor at the Johns Hopkins Bloomberg School of Public Health, shares La Barbera’s concerns about the applicability of the work to human embryos. The researchers’ insight is profound, Evans says, but it is still too soon to tell if their method will work with human embryos.
Deep Dive
Biotechnology and health
How scientists traced a mysterious covid case back to six toilets
When wastewater surveillance turns into a hunt for a single infected individual, the ethics get tricky.
An AI-driven “factory of drugs” claims to have hit a big milestone
Insilico is part of a wave of companies betting on AI as the "next amazing revolution" in biology
The quest to legitimize longevity medicine
Longevity clinics offer a mix of services that largely cater to the wealthy. Now there’s a push to establish their work as a credible medical field.
There is a new most expensive drug in the world. Price tag: $4.25 million
But will the latest gene therapy suffer the curse of the costliest drug?
Stay connected
Get the latest updates from
MIT Technology Review
Discover special offers, top stories, upcoming events, and more.