This company can encode your favorite song in DNA—for $100,000
At her office overlooking San Francisco’s Mission Bay neighborhood, Emily Leproust leaned over to show me a silver lozenge barely the size of a pill.
“This one capsule of DNA stores as much data as an entire Facebook data center,” she said.
Leproust is cofounder and CEO of Twist Bioscience, a five-year-old startup that is, by some estimates, the world’s largest supplier of manufactured DNA strands, or synthetic genes.
Custom-ordered genes are the raw material of synthetic biology and the starting point of every biotech drug, food, or fragrance. The DNA in the capsule represents a new market—potentially a huge one—for storage drives made out of DNA.
“We are the leader in DNA writing,” said Leproust, in a commanding voice carrying the accent of her native France. “And we intend to remain the leader.”
As Leproust led me through a curtain into a production room, we passed an Applied Biosystems Model 394 DNA synthesizer (state of the art in 1991). It could make four short DNA strands in a day, she told me. Today Twist makes three million strands a day.

Around a corner, we met Twist’s technology. Encased in glass, it resembles an ink-jet printer hacked together, with exposed circuit boards and nozzles aimed at postcard-size wafer of black silicon. As the machine kicked into life, little tanks squeezed droplets of adenine, cytosine, guanine, thymine—the A, C, G, and T of DNA—into 9,600 nano-wells, each the width of a strand of hair.
Thanks to this device, Twist is a key player in the race to make manufacturing DNA cheaper, selling it for seven to nine cents per DNA letter.
“She has been able to do what everyone [has] been dreaming about for 10 years—to lower the cost by an order of magnitude,” says John Cumbers, founder of the industry network SynBioBeta.
Making parts
Leproust earned her PhD in organic chemistry in Texas and then landed at Agilent Technologies. That company later accused her of stealing the technology behind Twist. Her lawyers say the lawsuit is meritless and an attempt to “suffocate” and “diminish” her success, which includes raising $259 million from investors.
The CEO compares her company to Intel, saying it sells the genetic equivalent of transistors to others who use them for new and profitable applications. “I ask my customers, ‘Do you have more ideas or more money?’ she says. “When we lower our price, they don’t save the difference. They buy more DNA.”
Cheaper DNA means more ambitious experiments. Last October, Twist signed a deal to supply the Boston bio-foundry Ginkgo Bioworks with one billion base pairs of manufactured DNA—the largest supply agreement in the industry’s history.
Ginkgo adds Twist’s genes to yeast or bacteria in a factory-like effort to identify new enzymes. Other scientists hope DNA will become so cheap they can afford to construct entire genomes, even human ones.
Twist’s innovations have helped create a challenging marketplace in which, industry insiders say, prices for DNA are falling faster than demand is going up. In January 2017, the tough economics claimed a competitor when Cambridge-based synthetic-DNA company Gen9 was sold off for parts only six months after signing its own major supply deal.
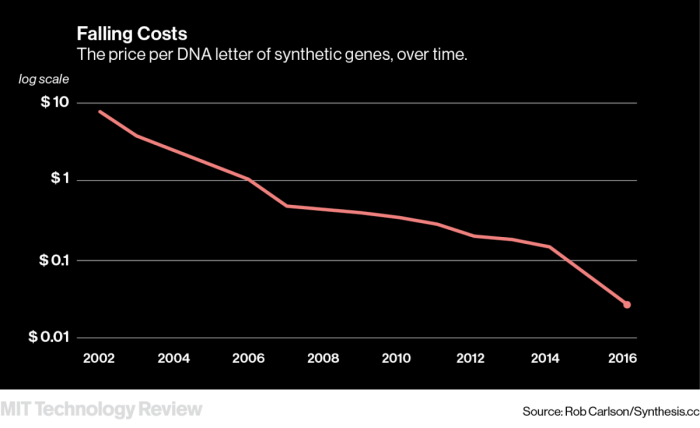
“There’s still a lack of understanding of what these tools can do,” says David Berry, a partner at Flagship Pioneering in Cambridge. “In the meantime, it’s a commodity race to the bottom.”
Storing data in DNA
That’s where the $30-billion-a-year data storage market comes in. The research group IDC predicts the world will be creating 163 zettabytes of digital data by 2025, enough to cause “memory full” warnings on 1.2 trillion iPhones.
Instead of archiving that data on magnetic tapes, it may be possible to encode it in DNA—each bit translated into genes, to be sequenced when the data needs to be retrieved.
As a data storage medium, DNA is incredibly dense. Theoretically, all the data in existence could fit in the bland conference room where Leproust was showing me the silver pill. “For archival data you want to store for a long time, there’s nothing better,” says Luis Ceze, a computer scientist at the University of Washington.
Since 2016, Twist has supplied 20 million custom-coded DNA strands to Microsoft Research, now working with Ceze to turn DNA storage into a business. The team has already encoded music videos, songs from the Montreux Jazz Festival, and the Universal Declaration of Human Rights, among other files.
This February, the group achieved another milestone, demonstrating in a paper published in Nature Biotechnology that they could recover individual data files, error-free, from over 200 megabytes of data stored in DNA. Such “random access” is necessary if DNA is to serve as a commercial storage medium.
But DNA will also need to get much, much cheaper. Leproust underscores that fact when she tells me that Twist has been offering customers the opportunity to store 12 megabytes of data in DNA, for a mere $100,000.
Leproust says that won’t always be the case—not if her plans to improve the technology work out. “In a few years it won’t be $100,000 to store that data,” she says. “It will be 10 cents.”
Deep Dive
Biotechnology and health
How scientists traced a mysterious covid case back to six toilets
When wastewater surveillance turns into a hunt for a single infected individual, the ethics get tricky.
An AI-driven “factory of drugs” claims to have hit a big milestone
Insilico is part of a wave of companies betting on AI as the "next amazing revolution" in biology
The quest to legitimize longevity medicine
Longevity clinics offer a mix of services that largely cater to the wealthy. Now there’s a push to establish their work as a credible medical field.
There is a new most expensive drug in the world. Price tag: $4.25 million
But will the latest gene therapy suffer the curse of the costliest drug?
Stay connected
Get the latest updates from
MIT Technology Review
Discover special offers, top stories, upcoming events, and more.