Biotech’s Coming Cancer Cure

When Milton Wright III got his third cancer diagnosis, he cried until he laughed. He was 20 and had survived leukemia twice before, first when he was eight and again as a teen. Each time he’d suffered through years of punishing chemotherapy.
But now he had checked himself in to Seattle Children’s Hospital. An aspiring model, he had taken a fall before a photo shoot and found he couldn’t shake off the pain in his ribs. When the doctors started preparing him for a spinal tap, he knew the cancer was back. “I said, Oh, man, they are going to tell me I relapsed again,” he recalls. “They’re going to give me my six months.”
The third time wasn’t good, he knew. He’d seen enough sick kids at the Ronald McDonald House to know that when leukemia comes back like this, it’s usually resistant to chemotherapy. Hardly anyone survives.
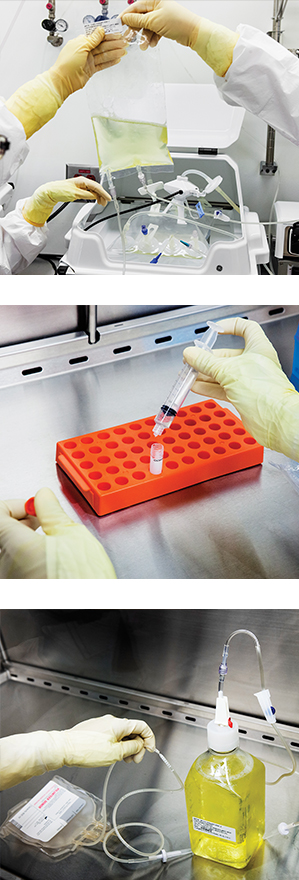
Top: A bioreactor bag holds a leukemia patient’s T cells. The cells have been genetically modified to fight cancer. A new receptor has been added.
Middle: A sample of a patient’s T cells is prepared for quality tests.
Bottom: A bottle of nutrients is used to feed the T cells, which are grown for about 10 days, until they number in the billions. Then they can be reinfused into a patient’s veins.
But Wright did. In 2013 his cancer, acute lymphoblastic leukemia, was destroyed with a new type of treatment in which cells from his immune system, called T cells, were removed from his blood, genetically engineered to target his cancer, and then dripped back into his veins. Although Wright was only the second person at Seattle Children’s to receive the treatment, earlier results in Philadelphia and New York had been close to miraculous. In 90 percent of patients with acute lymphoblastic leukemia that has returned and resists regular drugs, the cancer goes away. The chance of achieving remission in these circumstances is usually less than 10 percent.
Those results explain why a company called Juno Therapeutics raised $304 million when it went public in December, 16 months after its founding. In a coup of good timing, the venture capitalists and advisors who established Juno by licensing experimental T-cell treatments in development at Seattle Children’s, the Fred Hutchinson Cancer Research Center, and hospitals in New York and Memphis took the potential cancer cure public amid a historic bull market for biotech and for immunotherapy in particular. Its IPO was among the largest stock market offerings in the history of the biotechnology industry.
The T-cell therapies are the most radical of several new approaches that recruit the immune system to attack cancers. An old idea that once looked like a dead end, immunotherapy has roared back with stunning results in the last four years. Newly marketed drugs called checkpoint inhibitors are curing a small percentage of skin and lung cancers, once hopeless cases. More than 60,000 people have been treated with these drugs, which are sold by Merck and Bristol-Myers Squibb. The treatments work by removing molecular brakes that normally keep the body’s T cells from seeing cancer as an enemy, and they have helped demonstrate that the immune system is capable of destroying cancer. Juno’s technology for engineering the DNA of T cells to guide their activity is at an earlier, more experimental stage. At the time of its IPO, Juno offered data on just 61 patients with leukemia or lymphoma.
Juno is located in South Lake Union, a Seattle neighborhood dominated by Amazon.com, whose CEO, Jeff Bezos, was an early investor in the company. During a day spent at Juno’s labs and offices in May, the phrase I heard repeated over and over was “proof of principle.” That’s what cases like Wright’s have provided. The studies are small, with no control groups, no comparisons, but also no other explanation than T cells for why the cancer disappears. “It’s proved that the T cell is the drug,” says Hans Bishop, a former Bayer executive who is the company’s CEO.
Bishop argues that medicine is entering a new phase in which cells will become living drugs. It is a third pillar of medicine. The pharmaceuticals that arose from synthetic chemistry made up the first pillar. Then, after Genentech produced insulin in a bacterium in 1978, came the revolution of protein drugs. Now companies like Juno are hoping to use our own cells as the treatment. In the case of T cells, the tantalizing evidence is that some cancers could be treated with few side effects other than a powerful fever.
Medicine is entering a new phase in which cells will become living drugs.
Moving beyond the proof of principle won’t be easy. No one has ever manufactured a cellular treatment of any commercial consequence. It’s not certain what the best way to make and deliver such personalized treatments would be. Nor is it clear whether engineered T cells can treat a wide variety of cancers; this year Juno and others are launching new studies to find out. Even in leukemia, cancer that affects the bone marrow and blood, it’s too early to declare a cure. The majority of patients receiving the therapy have been treated only in the last 12 months. About 25 percent have seen their cancers roar back, sometimes mutated in a way that makes them immune to the T cells. At 18 months since his treatment, Wright, who hopes to become a police officer, is one of the longest survivors.
Juno isn’t the only company chasing the T-cell idea. More than 30 companies have started clinical tests or are planning them, including Novartis, which says it may file for approval for a competing leukemia treatment in 2016. The U.S. Food and Drug Administration last summer gave both Novartis and Juno so-called breakthrough designation, meaning that their leukemia treatments could be approved after only one larger clinical trial.
If early results hold, tests of engineered T cells in blood cancers may lead to one of the fastest approvals in the history of drug development. It could take as little as seven years, whereas the average drug takes closer to 14 years. “That is unheard of in the industry,” says Usman Azam, head of gene and cell therapy for Novartis.

At Juno I met the CFO, Steven Harr, who before joining the company was an investment banker specializing in biotech at Morgan Stanley. I asked whether he’d ever paid attention to cell therapy companies while on Wall Street. No, he said. Just the opposite. They were considered dogs, chasing an idea that didn’t work—and even if it did, it was too complicated to commercialize. The FDA lists 14 approved cell therapies, most of which are skin grafts or involve storing umbilical cords.
But Harr says he “jumped on the bandwagon” when he saw the data from the leukemia patients. Now he thinks Juno will find an economic advantage by solving the difficult problem of how to commercialize cellular treatments. “It’s a living thing—it’s different from a pill,” he says.
The treatment
“They hyped it up, like it was going to be amazing,” Wright remembers. He’d signed up for the clinical trial right away, but he didn’t tell anyone he was at the hospital. His mom was texting him: “Where you at? What’s up?” After a few days he finally told her. “I’m at Children’s. I’m getting ready for a trial.” Wright underwent a two-hour process known as leukapheresis, in which his blood was passed through a device to separate out the T cells. The cells were taken to a lab, where a strand of new DNA was inserted using a virus. Two weeks later he got the treatment: a 10-minute drip from an IV bag to reinfuse the cells. Easy stuff compared with chemotherapy. And at first, nothing happened.
A sign of how potent the T-cell treatments are is that most patients suffer from “cytokine release syndrome,” a storm of molecules generated as the cells fight the cancer. At least seven patients have been killed by the syndrome. Wright’s doctors kept checking in to see if he had developed a fever, which would signal that the T cells were working. “They were pressuring me—‘Come on, call us,’” he says. Two weeks later it came on like a body-flattening flu. He was admitted to the ICU and says he was barely lucid when smiling doctors told him they couldn’t find cancer in his body.
Carl June, the University of Pennsylvania doctor who publicized some of the first successful treatments with engineered T cells, has likened what’s happening inside patients’ bodies to “serial killing” and “mass murder.” As the billions of T cells in a dose multiply, they can locate and kill several pounds of tumor.

That’s something normal T cells don’t do. One reason is that they’re trained not to harm your body, an effect known as tolerance. The training occurs in the thymus, the organ for which T cells are named. Each cell bristles with thousands of copies of a single receptor, its shape generated at random by shuffled DNA (a quintillion possible arrangements are possible). T cells whose receptor attaches strongly to surface markers, called antigens, on the body’s own cells are discarded. The rest head out to patrol for foreign-looking viruses, bacteria, or infected cells, which they stick to and destroy. “The problem is that cancer is you,” says Michel Sadelain, a researcher at Memorial Sloan Kettering Cancer Center and one of Juno’s scientific founders. “The antigens on cancer just aren’t that enormous and juicy.”
Credit for the idea of getting around tolerance with an engineered T cell goes to an Israeli scientist named Zelig Eshhar. In a study published in 1989 in the Proceedings of the National Academy of Sciences, he replaced the T cell’s natural receptor with one that he chose. Eshhar realized that with his technique, a T cell could be engineered to attach to whatever it was instructed to attach to.
It’s an idea as dangerous as it is powerful. The reason is that few antigens appear exclusively in cancer cells. In 2009, a woman given T cells engineered to recognize colon cancer suddenly went into respiratory distress; she died five days later. Doctors at the National Cancer Institute quickly canceled the study, concluding that the T cells had encountered their antigen in her lungs and attacked.
Scientists like Sadelain soon zeroed in on one ideal antigen, called CD19. It appears nowhere in the body except on B cells, the same kind that go awry in lymphoma and in the leukemia that afflicted Wright. And it turns out that wiping out a person’s B cells isn’t life-threatening. With shots of immunoglobulin, you can live without any for years.
By 2010, doctors at Memorial Sloan Kettering, Penn, and the National Cancer Institute had begun trying to treat leukemia patients with T cells bearing a doctored receptor for CD19. To the inside of the receptor, they’d added another snippet of DNA that stimulates the cells to divide. No one is sure how the stimulation works, but without it, the modified T cells don’t do much. Early case reports eventually multiplied into trials that have treated about 350 leukemia and lymphoma patients. The results are remarkable, partly because they’re so consistent, even though each lab uses slightly different DNA designs.
Fast follower
Penn’s first results were well publicized and drew the attention of Novartis, the world’s second-largest drug company. In August 2012, it agreed to give the university $20 million to build a new cell-therapy center as part of an alliance through which Penn’s T-cell therapies will be sponsored and owned by the Swiss pharmaceutical giant. The deal was notable for being struck on the basis of published data from just three patients, and now it looks like a bargain.
It also makes Juno a “fast follower,” in startup parlance. Incorporated in August 2013, it is “a company of many fathers,” says Lawrence Corey, an infectious-disease doctor who was then the president of Fred Hutchinson. Corey, aided by the venture capitalist Bob Nelsen and Richard Klausner, the former director of the National Cancer Institute and now chief medical officer of the DNA-sequencing company Illumina, created Juno by buying up patents and licensing rights to T-cell trials under way in Seattle and at Sloan Kettering in New York.
It’s an idea as dangerous as it is powerful. At least seven patients have been killed.
Since its IPO, Juno’s stock market value has surged above $6 billion, reflecting intense speculation that engineered T cells will prove to be a new way to treat many types of cancer, not only the relatively rare leukemia Wright suffered from. Juno’s executives believe that they can quickly come up with new T cell designs and obtain a “fast readout” by testing them in terminal cancer patients, where risks are easy to justify. The company plans to have 10 studies of six different T-cell designs in progress by next year. “We are looking for breakthroughs,” says Mark Frohlich, a doctor who is Juno’s vice president for strategy. “We aren’t going to say, ‘Okay, two months survival.’”

The big question mark is whether T cells will work in cancers other than those of the blood. The week before I visited Juno, investors briefly sent its shares tumbling by 35 percent after Novartis and Penn reported that low doses of engineered T cells had no dramatic effects in five patients with cancers of the pancreas, ovaries, or lung. Still, the data were too preliminary to indicate much. “We know it’s feasible. But how many cancers can you apply this to? That we don’t know,” says Sadelain. “What’s changed is that everyone now knows what to do. I think that partly explains the frenzy around T cells.”
The goal is to find the next CD19. But that’s not easily done. Since few antigens appear only on tumor cells, any targeted T cell runs the risk of wiping out vital organs, as happened to the colon cancer patient in 2009. The Recombinant DNA Advisory Committee, a federal body that oversees gene therapy, called a meeting this June to debate how scientists planned to avoid these and other side effects. One way to lessen the risk is already being tested in patients: “suicide switches,” which let doctors rapidly kill off all the engineered T cells should any serious problems arise. This spring, Michael Jensen, a pediatric cancer doctor at Seattle Children’s whose cell-therapy center treated Wright, opened a study to treat neuroblastoma, the most common cancer affecting infants. He says T cells will target an antigen found on nerve cells. If the T cells do unexpected damage, they can be inactivated with a dose of the drug Erbitux.
Safety isn’t the only obstacle. How can engineered T cells be made to persist in a person’s body to provide permanent protection? So far they don’t seem to linger in many patients, something Frohlich terms a “big problem.” And dense organ tumors can saturate their surroundings with signals, like a molecule called PD-L1, that turn T cells off. This defense is the process that checkpoint inhibitors, the new immunotherapy drugs sold by Merck and Bristol-Myers Squibb, interfere with. But DNA engineering may offer clever solutions as well. Jensen says he rewired the DNA of T cells so this “off” signal instead provokes them to kill even more.
Jensen is optimistic that rapidly improving techniques for modifying genes, and for handling and growing cells, will let researchers conquer solid tumors. “What is in the clinic now with leukemia is version 1.1 of this operating system,” he says. “But back in the labs, that is already antiquated technology.”
Given that it took 20 years to come up with the results in leukemia, Sadelain told me, “it would be naïve to expect a breakthrough every quarter.” Yet a dozen newly launched studies on T cells mean some big results could be in the wings.
One study I heard about is led by Marcela Maus, an oncologist at Penn, who this year tested engineered T cells in five patients with glioblastoma, an incurable brain cancer. When one of these patients underwent brain surgery, Maus discovered that the tumor had been mostly killed. No cancer cells with the marker she’d aimed at remained at all. So was this proof that T cells can treat brain cancer, too? Maus is reluctant to answer that question. “Potentially,” she says. It’s just too soon to know if these patients will live any longer than they would have otherwise. “It’s hard to exercise patience, but that is what is needed,” she says.
Commercial barriers
When Wright’s white blood cells were collected in late 2013, they headed to a processing facility at Seattle Children’s. Workers laboring in masks and safety suits placed them into bioreactors and used a virus to insert the new DNA. Then the cells were grown for 10 days inside plastic sacks fed with human blood serum. If a half-dozen academic centers hadn’t built specialized clean rooms like this one, there wouldn’t be any clinical trials, or any IPOs. But Jensen’s center is no commercial operation: it can prepare cells for only 10 patients a month. It costs $75,000 to manufacture cells for each one.
“There’s no model for how much it costs. But remember, we get to utter words like ‘cure.’”
Jensen says about a quarter of the children whose parents want to enter them in the Seattle study aren’t accepted. Sometimes the reasons are medical, but not always: capacity is simply limited. “I wish every kid could get it,” Jensen says of the cell treatment. “The major barrier is the commercial end of it—having their factories built, having their trials done, and it being something that a doctor would be able to write a prescription for in any part of country.”

In fact, no one is quite sure how a personalized cell therapy will be commercialized at a large scale. Schematics outlining how it would work typically show not just a dozen complex laboratory steps but two airplanes, to get cells to and from patients. That explains why the largest number of Juno’s employees are involved in process engineering. One of them, Chris Ramsborg, gave me a tour around what he called the “sandbox” where new ideas for growing and packaging cells are being worked out. But most of the equipment was hidden from sight. “The manufacturing technology and how we are deploying it is the secret of Juno,” he said. “The techniques to make these products don’t really exist yet.”
Several members of Juno’s staff, including Ramsborg, Frohlich, and Hans Bishop, worked at another Seattle biotech named Dendreon, which developed a T-cell treatment for prostate cancer. (The cells, instead of being engineered, were exposed to cancer antigens and then multiplied. The treatment was only modestly effective.) Even though Dendreon charged $93,000 for its treatment, it cost half that much to manufacture. The company filed for bankruptcy last year.
Dendreon’s manufacturing plant in New Jersey was scooped up by Novartis, which has started using it to process cells for patients involved in its leukemia study. Azam says Novartis, which has 400 people working in gene and cell therapy, is already studying the logistics of how a personalized cell therapy could be offered globally. “We have been mapping out how we would do the patient journey, the individual cell journey,” he says. “It’s a new way to treat patients, but also a new way of practicing business.”
It may one day be possible to mass-produce off-the-shelf T cells or even do genetic engineering at a patient’s bedside. Some labs are working with instruments to pump genetic material into cells using electricity or pressure. Others have shown they can generate T cells in a lab dish and use them to cure mice, raising the possibility of T-cell factories. For now, though, all the engineered T-cell treatments in clinical testing use a patient’s own cells.
So how much will a dose of genetically engineered immune cells cost? One Citigroup analyst estimated that the price could exceed $500,000. That would be more expensive than nearly any existing cancer drug. Yet it might be considered cheap if a 10-minute drip could effectively treat leukemia without causing permanent damage to the patient. Current chemotherapy treatments last for a year or more and can weaken a person’s heart and body for a lifetime. The hospital bills for leukemia patients can top $2 million.
Harr, Juno’s CFO, was well known on Wall Street for criticizing the high cost of cancer drugs; he warned that the government might step in and set prices if they weren’t reined in. When I asked him about the T-cell treatments, he said it was too soon to guess at a price. It depends how well they work and how hard they are to make. “There’s no model for how much it costs,” he said. “But remember, we get to utter words like ‘cure.’ And at this point, it’s a single dose.”
—Antonio Regalado
Deep Dive
Biotechnology and health
How scientists traced a mysterious covid case back to six toilets
When wastewater surveillance turns into a hunt for a single infected individual, the ethics get tricky.
An AI-driven “factory of drugs” claims to have hit a big milestone
Insilico is part of a wave of companies betting on AI as the "next amazing revolution" in biology
The quest to legitimize longevity medicine
Longevity clinics offer a mix of services that largely cater to the wealthy. Now there’s a push to establish their work as a credible medical field.
There is a new most expensive drug in the world. Price tag: $4.25 million
But will the latest gene therapy suffer the curse of the costliest drug?
Stay connected
Get the latest updates from
MIT Technology Review
Discover special offers, top stories, upcoming events, and more.