The record for high-temperature superconductivity has been smashed again

Superconductivity is the weird phenomenon of zero electrical resistance that occurs when some materials are cooled below a critical temperature. The best superconductors have to be cooled with liquid helium or nitrogen to get cold enough (often as low as -250 °C or -480 F) to work. The holy grail for researchers is the idea that a material could be made to superconduct at around 0 °C — so-called room temperature superconductivity. If such a thing was ever discovered it would unleash a wide range of new technologies, including super-fast computers and data transfer.
The history of superconductivity is littered with dubious claims of high-temperature activity that later turn out to be impossible to reproduce. Indeed, physicists have a name for this: USOs, or unidentified superconducting objects.
So new claims of high-temperature superconductivity have to be treated with caution. Having said that, the news today that the record for high-temperature superconductivity has been smashed is worth looking at in more detail.
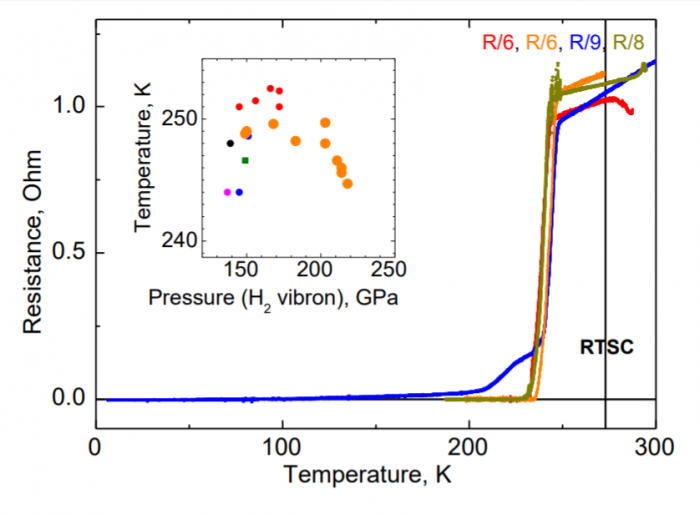
The work comes from the lab of Mikhail Eremets and colleagues at the Max Planck Institute for Chemistry in Mainz, Germany. Eremets and his colleagues say they have observed lanthanum hydride (LaH10) superconducting at the sweltering temperature of 250 K, or –23 °C.
That’s warmer than the current temperature at the North Pole. “Our study makes a leap forward on the road to the room-temperature superconductivity,” say the team. (The caveat is that the sample has to be under huge pressure: 170 gigapascals, or about half the pressure at the center of the Earth.)
Eremets has a fairly impressive pedigree in this field. Longtime readers of this blog will remember when he smashed the previous record for high-temperature superconductivity back in 2014. On that occasion, his team was able to measure superconducting activity in hydrogen sulfide at –80 °C, some 10 degrees warmer than any other material. He later raised that to –70 °C and published the work in Nature to huge acclaim.
But the jaw-dropping surprise for physicists was the nature of the superconducting material.
Superconductivity is well understood in conventional superconductors, which are rigid lattices of positive ions bathed in an ocean of electrons. Electrical resistance occurs when electrons moving through the lattice are slowed down by bumping into it, while superconductivity occurs when the lattice is cooled to a point where it becomes rigid enough for mechanical sound waves, or phonons, to ripple through it. These waves deform the lattice as they travel. And electrons can “surf” on this deformation.
In fact, at low temperature, the electrons bond to each other to form so-called Cooper pairs. And it is these Cooper pairs surfing through the lattice that constitutes superconductivity.
As the temperature increases, the Cooper pairs break apart and the superconductivity stops. This change occurs at what is called the “critical temperature.”
Before 2014, the highest critical temperature for this kind of superconductivity was about 40 K or –230 °C. Indeed, many physicists thought it impossible for this kind of superconductivity to work at higher temperatures.
That’s why Eremets’s announcement was so extraordinary—hydrogen sulfide is a conventional superconductor behaving in a way many people thought impossible.
(In 1986, physicists discovered an entirely different form of superconductivity in ceramic materials at 180 K or –90 °C. This is still not well understood, and little progress has been made in increasing the temperature.)
Eremets’s discovery triggered a feverish bout of theoretical activity to explain how the superconductivity occurs. The consensus is that in hydrogen sulfide, hydrogen ions form a lattice that transports Cooper pairs with zero resistance when the temperature drops below a critical level.
This can happen at high temperature because hydrogen is so light. That means the lattice can vibrate at high speed and therefore at high temperature. But the lattice also has to be held firmly in place, to prevent the vibrations from tearing it apart. That’s why the superconductivity only works at high pressure.
Since then, there has been considerable theoretical and computational work to predict other materials that might superconduct in this way at high temperature. One of the likely candidates has been lanthanum hydride, the one that Eremets and co have been working on.
The discovery that it superconducts at 250 K is a victory not only for Eremets and his team but also for the theoretical methods that predicted it. “This leap, by ~ 50 K, from the previous record of 203 K indicates the real possibility of achieving room temperature superconductivity (that is at 273 K) in the near future at high pressures,” say Eremets and co.
There is still some work ahead, however. Physicists require three separate pieces of evidence to be convinced that superconductivity is actually taking place. The first is the characteristic drop in resistance as the temperature falls. Eremets has this.
The second involves replacing the elements in the sample with heavier isotopes. This makes the lattice vibrate at a different rate and changes the critical temperature accordingly. Eremets and co have this evidence too, having replaced the hydrogen in their samples with deuterium and seen the critical temperature drop to 168 K, just as expected.
The third strand of evidence is called the Meissner effect: a superconductor should expel any magnetic field. It is here that Eremets and co have struggled. Their samples are so small—just a few micrometers across, and sitting inside high-pressure diamond anvil cells—that the researchers have not yet been able to measure this directly, although they do have some other magnetic evidence.
Without this final signature, physicists may withhold their full embrace. But it is surely something the team is working hard to produce.
In the meantime, the work opens up some obvious other avenues to pursue. The computational models suggest that yttrium superhydrides could superconduct at temperatures above 300 K—truly room temperature (although only at pressures more commonly found at the center of the Earth).
So room-temperature superconductors of one form or another may not be far away at all. The question then will be how best to exploit them.
Ref: arxiv.org/abs/1812.01561 : Superconductivity at 250 K in Lanthanum Hydride under High Pressures
Deep Dive
Computing
How ASML took over the chipmaking chessboard
MIT Technology Review sat down with outgoing CTO Martin van den Brink to talk about the company’s rise to dominance and the life and death of Moore’s Law.
Why it’s so hard for China’s chip industry to become self-sufficient
Chip companies from the US and China are developing new materials to reduce reliance on a Japanese monopoly. It won’t be easy.
Stay connected
Get the latest updates from
MIT Technology Review
Discover special offers, top stories, upcoming events, and more.