A slice of lab life
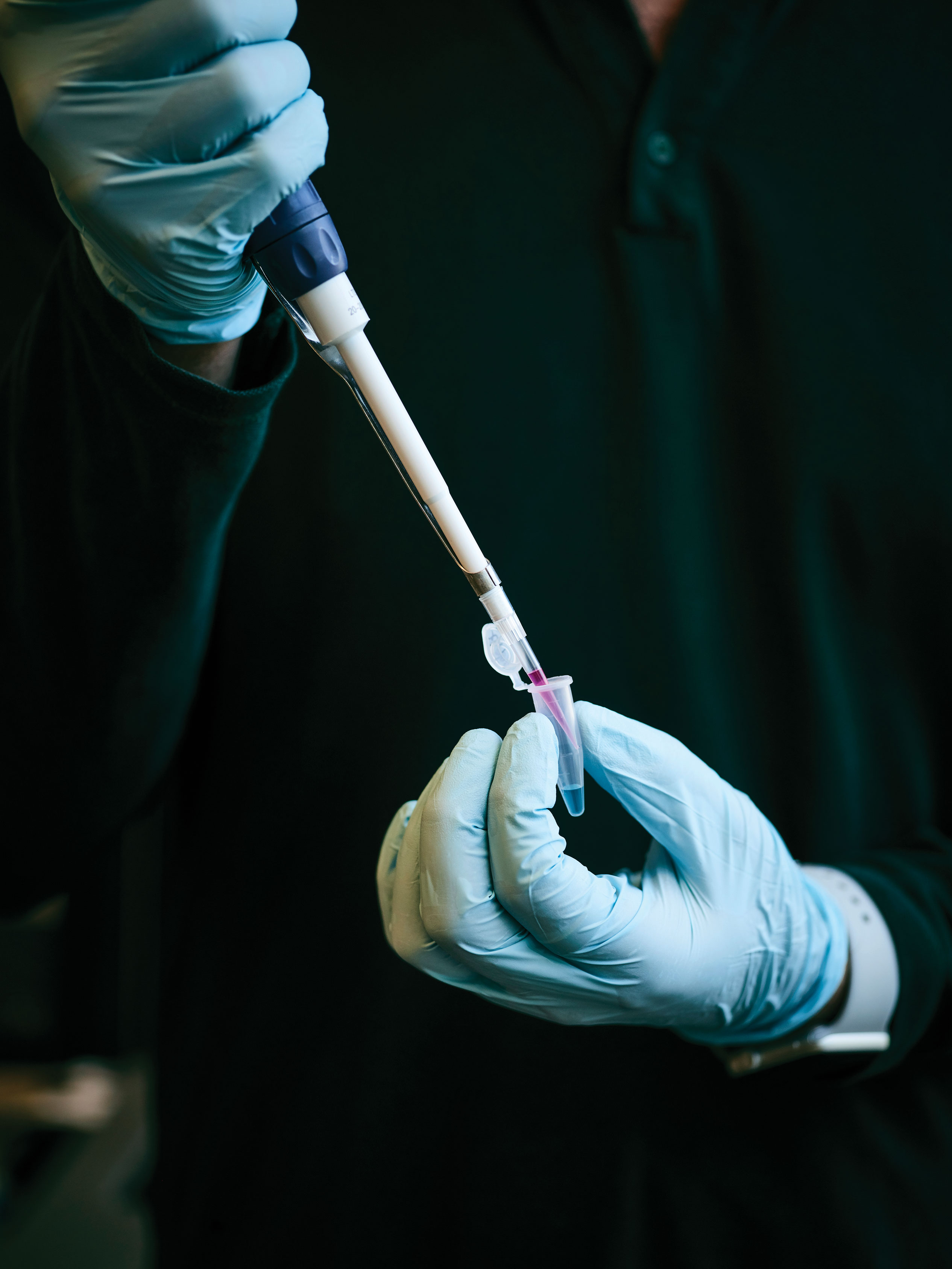
Colin Buss, a graduate researcher at MIT’s Koch Institute for Integrative Cancer Research, says it’s not too much of an exaggeration to call the BD LSRFortessa his best friend. “We’ve spent many a Saturday afternoon together,” Buss says of the flow cytometer, which is used for counting and sorting cells in a solution. After he places a tube of cell samples on the device and a metal nozzle sucks in the liquid, the machinery separates the individual cells and analyzes them by exposing them to a variety of lasers. Fortessa can churn through a thousand cells per second, saving researchers like Buss countless hours of peering into a microscope. No wonder they’re friends.
Even when using techniques at the cutting edge of his field, Buss, like so many of the 6,900 other grad students across campus, puts in long hours in the lab carrying out seemingly routine tasks. But the potential of his research is far from mundane. The technology he’s testing by examining all those cells could lead to better treatments for sepsis, a condition in which the body’s overreaction to an infection damages its own tissues and organs. Each year, some 250,000 Americans die from sepsis, which is estimated to kill more than five million people annually worldwide.
Buss ended up befriending Fortessa at MIT after being inspired by the work of bioengineering professor Sangeeta Bhatia, SM ’93, PhD ’97, who was developing cancer diagnostics and therapeutics that made frequent use of nanoparticles as a delivery technology. Buss had studied chemical engineering at Cornell and initially considered becoming a doctor, but he realized he was more interested in medical research. “I feel like I can have a greater breadth of impact developing a new therapeutic technique rather than treating patients directly,” he says. Eager to help expand Bhatia’s nanoparticle technology into new areas of application, like targeting cells of the immune system, he joined her Laboratory for Multiscale Regenerative Technologies at the Koch Institute five years ago. His research there as a doctoral student has pushed him to tap into his inner engineer, he says, to “come up with a solution to some really interesting question.”
The question that now has his attention is whether the immune system’s natural response to infection can be fine-tuned with a bit of nanotech-enabled gene manipulation. So today, he’s using Fortessa to analyze samples of white blood cells he collected from mice. Last night, he had several mice inhale airborne bacteria, which caused them to develop an infection in three to four hours. Early this morning, he injected them with nanoparticles containing small interfering RNA (siRNA). And if all went as planned, Buss says, introducing that genetic material into their bloodstream should help regulate their immune systems’ response to the infection.

Mice’s bodies react to infections the same way humans’ do. When the immune cells constantly circulating in the bloodstream detect the presence of an intruder, such as a virus, bacteria, or fungus, they kick the body’s immune response into gear and recruit many platoons of defender cells—including several of the five varieties of white blood cells—to join them at the site of the infection. Buss is studying monocytes, a type of white blood cells that often do their job a little too well, recruiting too many other cells and creating too much inflammation. While a certain amount of localized inflammation at the infection site is crucial to destroy the invading pathogen, the prolonged and widespread inflammation that occurs in sepsis can lead to organ damage and failure. “With infections in sensitive organs like the lungs and heart, your immune response can want to throw everything it has at the bacteria,” Buss says. “But too much inflammation and too little focus on regulating the rest of your body can actually kill you before the infection does.”
“Your immune response can want to throw everything it has at the bacteria. But too much inflammation ... can actually kill you before the infection does.”
The siRNA-carrying nanoparticles, which Buss designed, are meant to control that inflammation. An engineered molecular coating helps them survive the swim through the bloodstream and allows them to dock on the monocytes’ membranes to unload their cargo. The siRNA then slips into the cells’ DNA replication process, blocking the production of a signaling protein that acts as a chemical lighthouse to recruit other monocytes to the infection site. The challenge lies in making sure that the bundles of siRNA don’t interfere with other varieties of immune cells or shut down the monocytes completely. “We still want the monocytes to react to the infection,” Buss says. “We just don’t want them to overreact.”
Today’s experiment is designed to test whether the nanoparticles reached their intended destination. So after isolating the white blood cells from the blood samples, he color-codes them. Each kind of white blood cell has a unique combination of surface receptors, loading docks used to bind and interact with other cells. Buss tags the cells with a variety of dyes designed to attach only to a specific receptor, which allows him to sort them by cell type. (Monocytes, for example, will emit blue, white, and yellow light in Fortessa.) Buss has also dyed his nanoparticles pink. So if the delivery was successful, the monocytes should also glow pink when he runs them through Fortessa’s lasers. And by counting the number of other white blood cells also emitting pink, Buss will be able to determine how many received unintended siRNA parcels.

To color-code the white blood cells, Buss uses a propipette, an instrument that looks like a hot-glue gun, to dispense dye into his solutions. His motions are rhythmic—stick the propipette into a new sterile tip, suck up the solution, dispense the solution, flick the used tip into a bio-waste bucket, and repeat. Creating dozens of samples—including control samples to properly calibrate Fortessa—is repetitive work, but Buss says he has “found his groove.”
In his years working in the lab, Buss says, he has seen the growing promise of “tiny technologies” to create more precise and tunable effects in the biological processes of living things. Technologies like siRNA in nanoparticles allow researchers to home in on one behavior in one specific group of cells, accuracy that has long been out of reach in drug therapies for cancer and infectious diseases. “siRNA therapeutics give us the ability to control cells at the level of individual genes—something that has traditionally been difficult with small-molecule drugs,” he says. “Hence why a lot of drugs have multiple adverse side effects.”
Buss hopes that his nanoparticle experiments, including previous work in which he targeted cancer cells, will be his thesis project. “The biggest difference between cancer work and infectious-disease work is the time scale,” he says. “With cancer, you inoculate the mice and work over the course of a month. With infectious diseases, everything has to happen in 24 hours.”
With the clock ticking on his siRNA experiment, Buss carries his dyed samples down three flights of stairs in a Styrofoam box of ice to run them through Fortessa. As the white blood cells chug through the maze of lasers inside, small dots begin to appear in a scatter plot on the monitor. He leans forward and examines the many plots as the cells begin to cluster.
“Do you see that?” he says, flipping between the plots of blue, white, and yellow—the dye colors corresponding to the receptors on the monocytes—and the plot corresponding to the pink tag on the siRNA parcels. The dots are clustering in the same place on all of them, which means his parcels found the monocytes and unloaded their cargo. He will have to examine the results for the other white blood cell types to see if they, too, received the siRNA parcels. And hundreds of repetitions of this same experiment lie ahead. But for now, he gazes at the scatter plot telling him that his nanoparticles found their target. “That one is beautiful,” he says.
Keep Reading
Most Popular
Large language models can do jaw-dropping things. But nobody knows exactly why.
And that's a problem. Figuring it out is one of the biggest scientific puzzles of our time and a crucial step towards controlling more powerful future models.
How scientists traced a mysterious covid case back to six toilets
When wastewater surveillance turns into a hunt for a single infected individual, the ethics get tricky.
The problem with plug-in hybrids? Their drivers.
Plug-in hybrids are often sold as a transition to EVs, but new data from Europe shows we’re still underestimating the emissions they produce.
Stay connected
Get the latest updates from
MIT Technology Review
Discover special offers, top stories, upcoming events, and more.