Gene Editing Study in Human Embryos Points toward Clinical Trials
A team of U.S.-based scientists who genetically edited scores of human embryos to remove a disease gene say they’ve laid the foundation for trying to create the first gene-modified people.
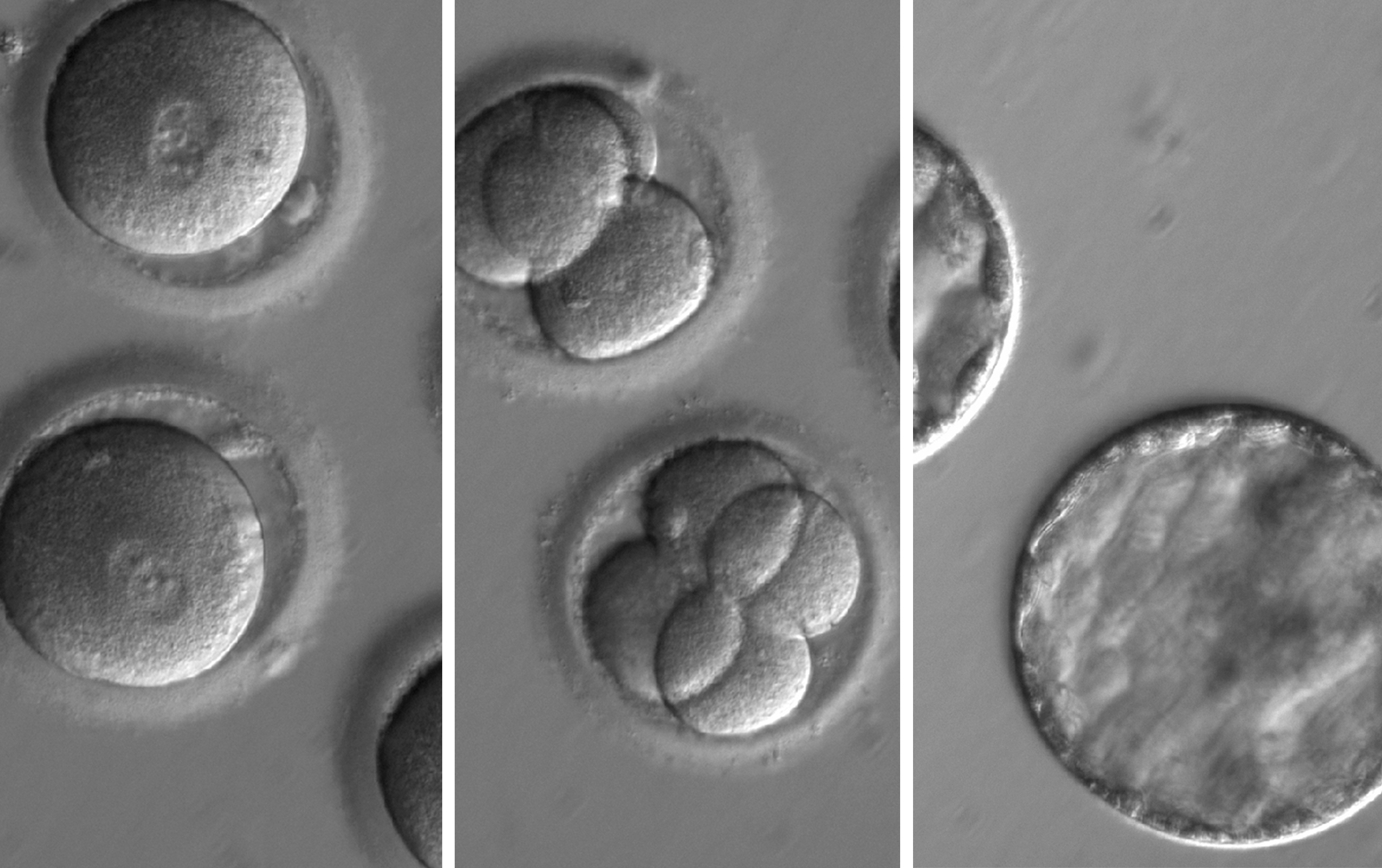
Shoukhrat Mitalipov and colleagues at Oregon Health and Science University successfully used the gene editing tool CRISPR-Cas9 to fix a disease-causing DNA error in dozens of early-stage human embryos.
The embryo editing project, the largest of its kind ever undertaken, was first described last week by MIT Technology Review.
According to a detailed report published today in Nature, the scientists demonstrated it was possible to correct a mutation in a gene called MYBPC3 that causes a deadly familial heart condition known as hypertrophic cardiomyopathy.
The scientists said the procedure was surprisingly effective and that clinical trials could eventually be considered. "With this particular mutation, we’ve already done the groundwork, so we’re probably much closer to clinical applications," Mitalipov said during a conference call with journalists. "Clinical trials would mean actually implanting some of these embryos with the goal of establishing pregnancy and monitoring births of children and hopefully following up with children."
The dramatic advance will deepen the debate over whether to use editing technology to intervene in the human gene pool. Critics have raised concerns that modifying embryos creates unknown risks to children and would be a slippery slope toward genetically enhanced “designer babies.”
The medical rationale for doing germ-line modification—that is, altering an embryo—is to repair a genetic defect even before birth, ensuring that a trait isn’t passed on to future generations. "This would completely eradicate the gene mutation and this disease from the lineage of a family," Mitalipov says.
Previously, three reports of genome editing experiments in human embryos have emerged from China. But Mitalipov’s work was much more extensive, involving the creation of about 150 human embryos, far more than any previous study.
In each case, eggs obtained from healthy donors were fertilized with sperm from a single man carrying the cardiomyopathy mutation.
Mitalipov says he believes gene editing could be used as a preventive tool to lower a newborn’s lifetime risk of a wide range of diseases, including cancer. For instance, CRISPR could be injected in embryos to fix mutations in the BRCA gene which are associated with a high risk of breast and ovarian cancer.
Some people whose families are afflicted by inherited disease say they wouldn’t hesitate to employ the technology, were it available. Lisa Salberg, 49, who was diagnosed with the same genetic heart condition at age 12, says she if she were having children now she would choose gene editing “in a minute.”
Salberg, who is the founder of the Hypertrophic Cardiomyopathy Association, says her father had the disease and that her daughter also inherited it. “I was told at age 12 that I could die suddenly and that there was nothing anyone could do about it,” she says. “I’ve never known healthy.”
Skeptics of germ-line editing argue that modifying genes in embryos is unnecessary. That is because women undergoing in-vitro fertilization can opt for pre-implantation genetic diagnosis, a test carried out on IVF embryos to determine if they are affected by a gene mutation.
In the case of the MYBPC3 gene error, known as an autosomal dominant disorder, half a couple’s embryos would be carriers of the disease, but half would not. Doctors can therefore test and transfer only unaffected embryos.
The existence of the testing option means that gene editing of embryos has "a very narrow window of opportunities," says Lluís Montoliu, a genome researcher at the National Centre of Biotechnology in Madrid, Spain. For women ages 35 to 40, pre-implantation diagnosis was used in about one out of four IVF attempts in the U.S. last year, according to the Society for Assisted Reproductive Technology.
However, Paula Amato, one of the study authors and a professor of obstetrics and gynecology at Oregon Health and Science University, says repairing embryos would increase the number of healthy ones and, potentially, result in quicker success getting pregnant.
In their study, the researchers found the efficiency of the editing process was greatest when CRISPR was injected into eggs along with sperm. When that was done, 42 out of 58 edited embryos were found to be free of the hypertrophic cardiomyopathy mutation.
In other words, CRISPR editing increased the number of healthy embryos from the expected 50 percent to about 72 percent. The remaining 16 embryos suffered unwanted genetic insertions or deletions, demonstrating that the editing technique requires further improvement.
Repairing genes early in life could also generate huge savings for society and the medical system. Salberg can name six relatives, including a 19-year-old great uncle and 36-year-old sister, that died early from the disease. She has already had five defibrillators implanted, suffered a stroke, lost part of her vision, and six months ago had a heart transplant. "You start doing the math, and CRISPR would be way cheaper," she says. "If you told me we could go back to 1967 and my mother could do CRISPR editing to take out the [hypertrophic cardiomyopathy] mutation so I wouldn’t have it, I’d have a very different life in so many ways."
Hypertrophic cardiomyopathy affects an estimated one in 500 people and can lead to sudden heart failure in people who look outwardly healthy.
But germline modification will undoubtedly face challenges moving beyond the lab, particularly in the U.S. Although the concept has been sanctioned by scientific bodies in limited circumstances, the National Institutes of Health does not fund research on human embryos and the U.S. Congress has forbidden the Food and Drug Administration from approving any clinical trials.
"There is a long road ahead, particularly if you want to do this study in a regulated way. It’s unclear at this point when we would be able to move on," Mitalipov says. "We would be supportive to moving this technology to other countries."
The scientists say their research held several surprises. One was that the editing process occurred in an unusual fashion. Embryos injected with CRISPR appeared to use the healthy maternal gene to guide the correction of the mutation but ignored a DNA template provided by scientists.
Mitalipov argues this means that the procedure should not actually be called gene editing, but that it is more of an assisted form of natural DNA repair. The argument means that children born from such embryos might not, strictly speaking, be considered genetically modified.
Keep Reading
Most Popular
Large language models can do jaw-dropping things. But nobody knows exactly why.
And that's a problem. Figuring it out is one of the biggest scientific puzzles of our time and a crucial step towards controlling more powerful future models.
How scientists traced a mysterious covid case back to six toilets
When wastewater surveillance turns into a hunt for a single infected individual, the ethics get tricky.
The problem with plug-in hybrids? Their drivers.
Plug-in hybrids are often sold as a transition to EVs, but new data from Europe shows we’re still underestimating the emissions they produce.
Stay connected
Get the latest updates from
MIT Technology Review
Discover special offers, top stories, upcoming events, and more.