Slow Progress to Better Medicine
As soon as Carina was born on July 25, 2014, it was clear something was wrong. The midwife pointed it out: a lump on the right side of her jaw. At first doctors were unable to make a diagnosis, but in time it was discovered to be a pernicious form of cancer that would, over the first year and a half of Carina’s life, resist eight rounds of chemotherapy and multiple surgeries.
Last October after yet another surgery, when the tumor again began to grow, Carina’s parents, Joe and Christie, found themselves running out of options. Radiation was a possibility, but there was a significant danger of brain damage. Radiologists advised them to first try any experimental drug they could find.

Genetic testing done on the tumor showed an abnormal fusion of two genes. This fusion seemed to be creating growth signals that were leading to cancer, and Carina’s oncologist, Ramamoorthy Nagasubramanian of Nemours Children’s Hospital, found that a medicine designed to interfere with a protein made by that fusion was in clinical trials for adults. He contacted the drug maker, Loxo Oncology, and together they appealed to the U.S. Food and Drug Administration to allow a pediatric trial. In December, Carina became its first patient.
Over the next month, Joe and Christie watched as the tumor shrank. It was the size of a walnut when the treatment started; 28 days later it seemed to be almost completely gone. Though Carina is not cured—she will continue to take the medicine for as long as it works, and at some point it is likely that the cancer will mutate again—today she is an active, curious two-year-old. The treatment “has bought us happy baby time,” says Joe. (To protect their privacy, the family asked that their last name not be disclosed.)
Carina’s case and others like it are the success stories of an approach to medicine that focuses on individual differences in patients’ genes, often in combination with information about their environment, health history, and lifestyle. It’s called precision medicine, a broad term most often used to describe an approach that combines data from many different sources to create a medical portrait of each person and design more effective, targeted treatments. It’s a big shift from our current medical model, which focuses on generic approaches and treatments suitable for as many people as possible, but its potential helped motivate a $215 million investment by the White House to help develop it. That’s also why Memorial Sloan Kettering Cancer Center in New York City, a leader in its field, has sequenced 10,000 tumors since 2014 in an effort to provide insight to oncologists developing cancer therapies individualized for each patient.
Like any big shift, this one will involve a lot of challenges to well-established practices across the medical industry, from regulation to drug development.
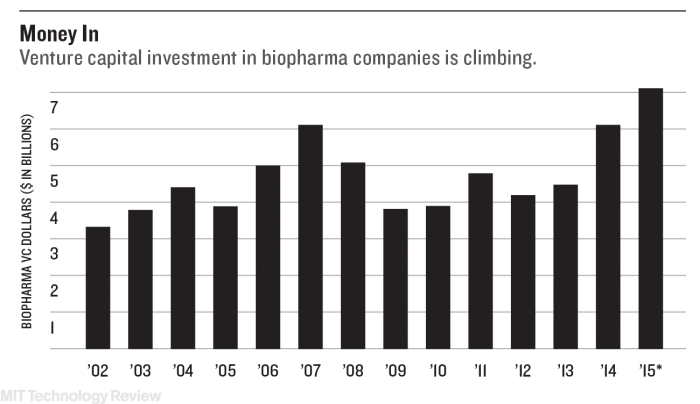
Which parts of the health-care industry will benefit from this disruption? That is the big question of this Business Report.
One group already profiting is technology-driven startups. Venture capitalists invested a record $7 billion in U.S. biopharmaceutical startups last year, according to data from Silicon Valley Bank—much of that driven by enthusiasm over new breakthroughs in genetic medicine and other technologies.
Loxo Oncology, maker of Carina’s medicine, exemplifies the role smaller companies are playing. Loxo founder Joshua Bilenker says the company has benefited from the new availability of public information in sources like the National Institutes of Health’s Cancer Genome Atlas.
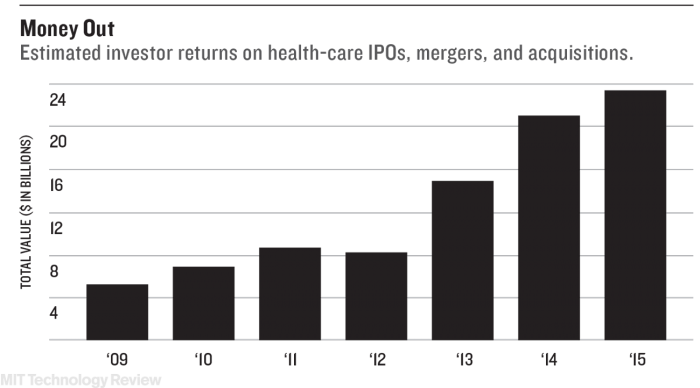
Private and public funding has also been available, with Loxo raising approximately $250 million from investors so far. Bilenker says his company’s focus on relatively simple genetically driven cancers makes it possible to know quickly if a treatment works or not, welcome news among investors. Treatments can also be tested in smaller groups, at a much lower cost than a large pharmaceutical company spends developing a medicine for more widespread illnesses. Blockbuster drugs for common ailments typically require large, expensive randomized trials. The condition Carina has, on the other hand, is unlikely to have more than a few thousand sufferers in the U.S.
The hope is that precision medicine will eventually be used to treat major illnesses. But that will be challenging. Those conditions are complex and may not have a clear genomic component.
With smaller groups of patients to treat, targeted medicines are unlikely to produce blockbuster revenues. Companies like Loxo—which has not yet set a price for its drug—will probably need to charge significant amounts per patient to recoup their investment. The average cost of cancer treatment is currently estimated at $10,000 per month, but after a decade of sharp price inflation in cancer drugs, insurers are questioning those prices. So are patients, who increasingly have to pay a significant portion themselves.
There’s technological risk as well. Editas Medicine is a company focused on using a new form of gene editing called CRISPR/Cas9 to treat medical conditions driven by genomic disorders. Slated to begin clinical trials next year, the company has a lot of complicated science to perfect before it can hope to be effective—everything from gene editing itself to delivering the therapy effectively into enough of the proper cells.
Beyond knowing more about the genome, precision medicine depends on gathering information about environment, lifestyle, and health history as well. Companies like WellDoc and Omada are trying to find ways to use mobile phones and computers to engage with patients, particularly those with chronic diseases like diabetes and hypertension, in order to record health and lifestyle data and help patients and doctors find insights.
Though some of these programs are successful—Omada reports that 64 percent of its participants are still actively using the system after a year—they have not sufficiently captured patients’ attention overall, says Joseph Kvedar, who runs the “connected health” initiative at Partners HealthCare, a leading Boston-area academic health-care system. “We haven’t been able to engage people the way Snapchat, Instagram, or Facebook can,” he says.
Kvedar acknowledges that a reminder of being sick is never going to be as enjoyable as social media, but there remains a great opportunity, he says: “All your device data, mobile data, all your activities that make you unique—if we can track that and your health, plus your genomic predisposition, there is so much we can do.”
Keep Reading
Most Popular
Large language models can do jaw-dropping things. But nobody knows exactly why.
And that's a problem. Figuring it out is one of the biggest scientific puzzles of our time and a crucial step towards controlling more powerful future models.
How scientists traced a mysterious covid case back to six toilets
When wastewater surveillance turns into a hunt for a single infected individual, the ethics get tricky.
The problem with plug-in hybrids? Their drivers.
Plug-in hybrids are often sold as a transition to EVs, but new data from Europe shows we’re still underestimating the emissions they produce.
Stay connected
Get the latest updates from
MIT Technology Review
Discover special offers, top stories, upcoming events, and more.