Clinics Offering Unproven Stem Cell Therapies Are Proliferating Across the U.S.
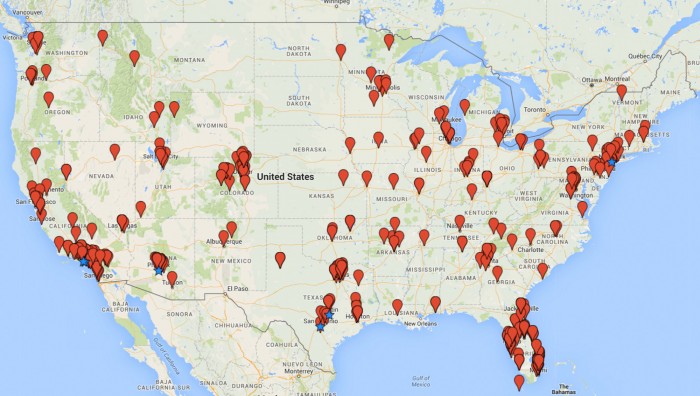
At least 351 U.S. businesses are offering unproven stem cell interventions in clinics across the country, claiming to treat everything from arthritis to Alzheimer’s, according to a paper published Thursday in the journal Cell Stem Cell. The finding casts light on an emerging area of medical treatment that has received little attention from the U.S. Food and Drug Administration.
“It is hard to do a project like this and think the FDA is doing a good job regulating the marketplace,” says Leigh Turner, a bioethicist at the University of Minnesota who performed the study along with Paul Knoepfler, a stem cell researcher at UC Davis.
Concern over unregulated stem cell therapies is not new, but the issue was previously thought to be mostly confined to patients who traveled abroad for treatments unavailable or forbidden domestically. These renegade treatments can be ineffective and dangerous. Just last week the New England Journal of Medicine described an incident in which surgeons discovered a sticky mass of foreign cells in someone’s spine after he got injections of fetal stem cells in several different countries.
Knoepfler and Turner’s new study shows that 570 clinics throughout the country are offering similar treatments, with perhaps tens of thousands of patients a year undergoing stem cell procedures. “If there were just one or two fly-by-night operations this wouldn’t be a problem,” Turner says. “But it seems to be a massive breakdown in regulations.”
FDA regulations are designed to address which types of cell-based therapies require agency approval and which do not. One of the biggest restrictions is on so-called non-homologous stem cell therapy: doctors can’t take cells from one tissue and place them in an unrelated organ. Sucking fat stem cells out of a patient’s abdomen and then injecting them into the brain to treat Alzheimer’s would almost certainly fall into this category.
Yet that’s exactly what some of these clinics are doing. Their therapies often make use of fat stem cells, perhaps because the procedure sounds so easy. Doctors use liposuction to obtain fat tissue from a patient. Then they liquefy it with enzymes, separate the cells in a centrifuge, and inject them back into the patient. The details vary, but the clinics often say the process is FDA compliant.
The FDA might disagree. Last year it released a draft guidance, which is not yet legally binding, saying this process alters the cells too much and therefore should undergo FDA approval just like drugs.
Aaron Levine, who studies public policy at the Georgia Institute of Technology, says that Knoepfler and Turner’s paper “should serve as a wake-up call,” or at least “raise the profile of this problem to the FDA.”
But while many researchers say the FDA should regulate these procedures, doctors working in the clinics often think it’s unnecessary. “I think that’s a huge mistake,” says Drew DeMann, the director of Manhattan Spine and Sports Medicine. “I don’t think these are really drugs. It’s just using tissue from our own body in different locations.” DeMann considers stem cells from bone marrow and fat tissue an “indispensable” tool for treating sports injuries, orthopedic conditions, and pain at his clinic. “There is no indication that they are not safe,” he says.
Knoepfler and Turner found that orthopedics was the area most commonly advertised by stem cell clinics, with over 300 businesses offering therapies. DeMann acknowledges that the effectiveness of these treatments should receive more study, but he also says, “I don’t think it is up to the clinicians to wait until there is an overwhelming degree of evidence.”
Some clinicians have more mixed feelings about stem cell regulations. “We need to do something about regulating these clinics,” says Duncan Ross, the founder of Chimera Labs, a stem cell clinic in Miami. His concern is that improper handling by inexperienced lab techs could contaminate the cells, a problem he claims to have solved in his own lab.
Currently, he is attempting to treat chronic obstructive pulmonary disease (COPD) by injecting fat stem cells into patients’ bloodstreams. That seems to conflict with the FDA’s rules against taking one kind of cell and putting it in a different organ. “That’s true, it’s non-homologous, but so what?” Ross says. “It’s never hurt anybody yet.”
But Turner worries about the clinics that promote therapies for diseases like Alzheimer’s, which 27 clinics were found to do. “If someone has a terminal illness and they begin looking on the Internet for a glimmer of hope, they are going to find a great many businesses that look professional and compelling and appear to have links to credible researchers and preclinical studies,” he says. That can lend a dangerous allure to treatments that have no proven track record of being effective—or even safe.
Despite the vast number of active stem cell clinics, the FDA has issued only a handful of letters warning them that their procedures must undergo FDA approval as biological drugs. Knoepfler says that creates “a disconnect between the magnitude of the stem cell industry and the action taken to [regulate] it.” Stem cell research has come a long way in the past 10 years, but Knoepfler says he wishes less attention were paid to the unproven and controversial therapies that athletes and celebrities receive. “I am really optimistic about the field overall, and we will see a fair number of clinical trials finish up over the decade,” he says. “We are just not there yet.”
Keep Reading
Most Popular
Large language models can do jaw-dropping things. But nobody knows exactly why.
And that's a problem. Figuring it out is one of the biggest scientific puzzles of our time and a crucial step towards controlling more powerful future models.
How scientists traced a mysterious covid case back to six toilets
When wastewater surveillance turns into a hunt for a single infected individual, the ethics get tricky.
The problem with plug-in hybrids? Their drivers.
Plug-in hybrids are often sold as a transition to EVs, but new data from Europe shows we’re still underestimating the emissions they produce.
Stay connected
Get the latest updates from
MIT Technology Review
Discover special offers, top stories, upcoming events, and more.