Stem Cells from a Diabetes Patient
A series of breakthroughs in cloning technology over the last year and a half are stoking hopes that cells could be used as treatments for patients with chronic, debilitating diseases such as diabetes and Parkinson’s.
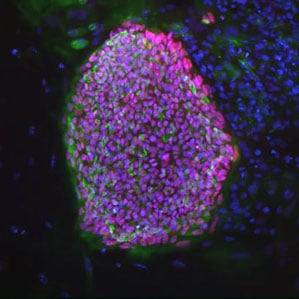
In January 2013, researchers at the Oregon Health and Science University reported that they had successfully created embryonic stem cells from a human embryo formed when the nucleus of one person’s cell was transferred into another person’s egg that had its original nucleus removed (see “Human Embryonic Stem Cells Cloned”). That was the first time stem cells had been made from such a cloned embryo, and the advance provides a potential route by which scientists could create various kinds of replacement cells based on a patient’s own genome. Many other research teams are pursuing another method of creating stem cells from a patient’s own cells, but some believe cells made with the cloning technique could be more likely to develop into a wide variety of cell types.
In the most recent advance for the cloning-based approach, a new report describes stem cells produced by cloning a skin cell from a woman with type 1 diabetes. The researchers were then able to turn those stem cells into insulin-producing cells resembling the beta cells that are lost in that disease. The immune system attacks these pancreatic cells, leaving patients unable to properly regulate their blood sugar levels.
Susan Solomon, a coauthor of the new study and cofounder of the New York Stem Cell Foundation (NYSCF), told reporters the results are an “important step forward in our quest to develop healthy patient-specific stem cells to be used to replace cells that are diseased or dead.”
The ultimate idea is to treat diabetes with insulin-producing cells made from a patient’s own cells and a donated egg. Currently, insulin-producing cells harvested from a cadaver are transplanted into some diabetes patients. But patients treated this way must take immunosuppressing drugs, and the number of cadaver cells is limited.
The cloned cells are thought to be better accepted by the immune system. But given that the body attacks its own beta cells, how can researchers prevent the immune destruction of the transplants? “It’s very difficult,” says Solomon. “We are acutely aware of the need to address both sides of the problem.”
There are also regulatory issues surrounding the cloning method. Lead researcher and coauthor Dieter Egli began the research at Harvard University but moved it to the New York institution because Massachusetts restrictions on egg donation prevented the work from progressing.
Egg supply is another challenge. The cloning worked about 10 percent of the time, report the authors, and not all of the embryos led to viable stem-cell lines. “When you think about wider application of this technology for patients with diabetes, cardiovascular disease, [and others], you are talking about hundreds of millions of people,” says Robert Lanza, a stem-cell pioneer at Advanced Cell Technology and coauthor of a recent cloning report. “When you start talking about numbers like that, it’s just not going to be practical to use these cells in that patient-specific way.”
Instead, he thinks patients could be better served by a bank of therapeutic cells that are created to fit a variety of immune types. “So you try to match tissue types, like with organ transplant now,” he says.
The NYSCF group is comparing the cloning-derived insulin-producing cells with similar cells derived from another type of patient-specific stem cell: induced pluripotent stem cells. Producing IPS cells does not require a donated egg; instead, they are made from an adult’s cell that has been reprogrammed by genetic factors (see “Engineered Stem Cells”). That technology was the basis for the 2012 Nobel Prize in medicine. While it may be surprising that researchers are still pursuing the cloning-derived stem cells when a less controversial option is available, experts say it’s not clear which cell type performs better. “Let science takes its course and determine what is best,” says Lanza.
Keep Reading
Most Popular
Large language models can do jaw-dropping things. But nobody knows exactly why.
And that's a problem. Figuring it out is one of the biggest scientific puzzles of our time and a crucial step towards controlling more powerful future models.
How scientists traced a mysterious covid case back to six toilets
When wastewater surveillance turns into a hunt for a single infected individual, the ethics get tricky.
The problem with plug-in hybrids? Their drivers.
Plug-in hybrids are often sold as a transition to EVs, but new data from Europe shows we’re still underestimating the emissions they produce.
Stay connected
Get the latest updates from
MIT Technology Review
Discover special offers, top stories, upcoming events, and more.