Accelerated Evaporation, Oil Spills, and the Economics of Water Desalination
Here’s an interesting puzzle. Electrorheological fluids are non-conducting oils carrying a sprinkling of dielectric nanoparticles, such as titanium oxide. Applying an electric field to these fluids, forces the nanoparticles together, turning the liquid to solid.
Electrorheological fluids have all kinds of applications in devices such as shock absorbers, brakes and artificial hips where engineers use them to instantly change resistance to movement.
But today Kunquan Lu and pals at Institute of Physics in Beijing, China, say that something strange has been going on in their lab where they study electrorheological fluids for some years.
The base for their electrorheological fluids is silicone oil, a thick viscous fluid that is remarkably robust and stable.
But Lu and co have noticed something strange when they add nanoparticles of titanium oxide to silicone oil–it suddenly begins to evaporate. They’ve found that in just a few days, a significant fraction of the fluid simply disappears.
That’s a huge surprise. Pure silicone oil can be stored for years or even decades without a hint of any evaporation. It’s hard to imagine what might induce this stuff to float off into the air.
Lu and co have been investigating the phenomenon. They’ve tested nanoparticles of different sizes and materials, the density of the suspension and even two different mineral forms of titanium oxide.
It’s easy to imagine that nanoparticles floating at the surface of the liquid increase the surface area and that it is this that allows an increased rate of evaporation.
Lu and co say that’s not what’s going on in their lab. They give two reasons. First, the evaporation rate of pure silicone oil is so low to start off with that even if the nanoparticles doubled the surface area, this couldn’t account for the dramatic evaporation they measure.
Second, if an increase in surface area were responsible, it ought to be possible to increase the evaporation rate using smaller nanoparticles.
But that doesn’t happen. In fact, there seems to be an optimum particle size below which the evaporation rate slows down.
The measurements have thrown up another surprise. Lu and co tested the evaporation rate with two types of titanium oxide nanoparticle called anatase and rutile, which have different crystalline structures. And it is this that turns out to have the biggest impact on evaporation rate.
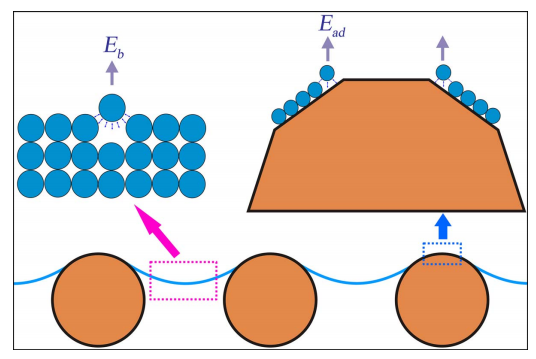
Understanding why is a challenge. Anatase titanium oxide forms crystals with well-defined edges–so the nanoparticles aren’t spherical. Lu and co speculate that the molecules of oil on the surface of a crystal nears these edges are less strongly bound than other molecules.
Consequently, these molecules float away more easily (see figure above). They hypothesis that it’s this process that accelerates evaporation but they say more work is need to characterise exactly what’s going on.
That’s a hugely significant finding. The ability to accelerate the evaporation of oil could have important consequences in all kinds of areas, for example in the way engineers clear up oils spills and other forms of pollution.
Then there is the possibility of repeating this process in water. Enhanced evaporation could dramatically change the economics of desalination, bringing cheap clean water to areas that have previously been without. “A raised evaporation rate will lead to effective and economic treatment of polluted water, or desalination of seawater,” say Lu and co.
However, if Lu and co.’s mechanism is correct, water will be more difficult to conquer than oil. Since it is less viscous, nanoparticles of titanium oxide tend to precipitate out of the liquid. That would make it hard to trigger the mechanism that Lu and co describe.
Nevertheless, it’s not beyond the realms of possibility that chemists will find some handy trick to prevent this, perhaps with other crystals or with coatings of one kind or another.
What is certain is that Lu and co’s work will generate some significant interest. The economics of water purification and of oil clean ups are of the highest importance.
Ref: arxiv.org/abs/1210.6101: Nanoparticle Enhanced Evaporation Of Liquids: A Case Study Of Silicone Oil And Water
Keep Reading
Most Popular
Large language models can do jaw-dropping things. But nobody knows exactly why.
And that's a problem. Figuring it out is one of the biggest scientific puzzles of our time and a crucial step towards controlling more powerful future models.
How scientists traced a mysterious covid case back to six toilets
When wastewater surveillance turns into a hunt for a single infected individual, the ethics get tricky.
The problem with plug-in hybrids? Their drivers.
Plug-in hybrids are often sold as a transition to EVs, but new data from Europe shows we’re still underestimating the emissions they produce.
Stay connected
Get the latest updates from
MIT Technology Review
Discover special offers, top stories, upcoming events, and more.