Brain Scan for Alzheimer’s
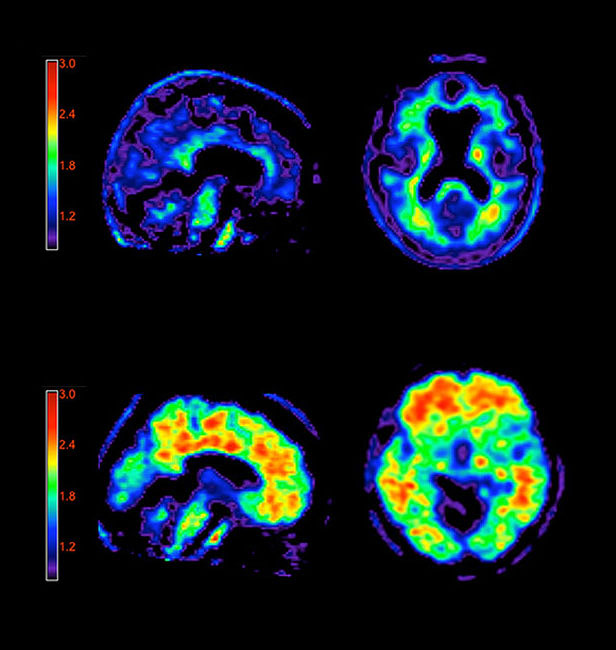
Beginning next month, doctors can use a brain scan to better diagnose Alzheimer’s. The U.S. Food and Drug Administration recently approved a fluorescent dye that binds to amyloid plaques, a physical hallmark of the disease, as a diagnostic tool.
Currently, doctors cannot be certain whether or not a patient’s brain is riddled with amyloid plaques until after the patient’s death. But now, clinicians can use a weakly radioactive dye to search for the presence of plaques in a living patient. The dye binds to the starchy amyloid that builds up in the brains of Alzheimer’s patients and can be visualized in a PET scan. The FDA approved the scan as a method for estimating plaque content in the brains of people exhibiting cognitive decline. The presence or absence of plaques in a patient with forgetfulness, confusion, or other signs of neurological trouble could help doctors with their diagnoses.
Although scientists do not know whether or not the plaques are the cause of Alzheimer’s, the plaques are strongly correlated with symptoms of dementia. Elderly people with the plaques have a more rapid mental decline than the elderly who don’t have the plaques. And even though doctors cannot treat Alzheimer’s, diagnostic tools like the plaque-marking dyes will help them identify patients with the disease, as well as those who do not have it but exhibit signs of mental decline due to other problems.
“A positive scan would add more weight to our bedside diagnosis,” says Liana Apostolova, an Alzheimer’s specialist at the University of California, Los Angeles. She says the technique could be useful in diagnosing patients with unusual forms of the disease and patients whose symptoms may be caused by other conditions, such as drug side effects or depression. “If a patient’s cognitive impairment is purely due to depression, then a negative amyloid scan would help ascertain that,” Apostolova says.
The new dye and other plaque-marking dyes have been used in clinical research for years, but Eli Lily, whose subsidiary Avid Radiopharmaceuticals produces the diagnostic agent, is the first to bring the technique to the doctor’s office. Some experts are still concerned that the clinical use of the dye might be a bit premature, since the presence of the plaques isn’t enough to confirm an Alzheimer’s diagnosis. In fact, the amyloid deposits are often found in older people with normal mental ability and could be associated with other neurologic conditions. For now, the Avid dye is only approved to be used to rule out Alzheimer’s (if no plaques are detected, a doctor can be confident the patient doesn’t have the disease).
“I think it puts the clinician in a bind,” says Ronald Petersen, director of the Mayo Clinic’s Alzheimer’s Disease Research Center. If a scan is negative, then a doctor can tell a patient that amyloid is not contributing, but if a scan is positive, what should a clinician do? “The FDA nor the company says anything about the meaning of a positive scan,” says Petersen. “The data is not out there.”
Petersen and others agree, however, that plaque-detecting diagnostics are critical for Alzheimer’s research. The dyes enable researchers to determine which participants carry plaques and thus design better trials for plaque-attacking drugs. Within the trials, plaque tracers can be used to monitor the amount of plaque in a participant’s brain during the course of treatment.
The tracers can also help the field elucidate the causes and disease mechanisms of Alzheimer’s, which will be key to treating or preventing the disease. Researchers can study people who have amyloid plaques and monitor their cognitive abilities over time. From the pool of cognitively normal elderly with a positive scan, knowing who “develops Alzheimer’s disease and who doesn’t will help us explore disease susceptibility and resistance, identify all genetic risk factors and the early metabolic changes, so we can then target these specific processes with drugs,” Apostolova says. “Diagnoses can be helpful, but we want therapeutics.”
More than five million people in the United States have Alzheimer’s, and that number is expected to double in coming years, according to the U.S. Department of Health and Human Services. By their early 70s, as many as 30 percent of people have amyloid plaques in the brain, says Michael Weiner, principal investigator of the Alzheimer’s Disease Neuroimaging Initiative, which tests imaging techniques as well as biomarkers for Alzheimer’s that could be found in the blood or cerebrospinal fluid.
The new dye-based scans “are going to create greater public consciousness of the disease, and that will have a big impact,” says Weiner.
Keep Reading
Most Popular
Large language models can do jaw-dropping things. But nobody knows exactly why.
And that's a problem. Figuring it out is one of the biggest scientific puzzles of our time and a crucial step towards controlling more powerful future models.
How scientists traced a mysterious covid case back to six toilets
When wastewater surveillance turns into a hunt for a single infected individual, the ethics get tricky.
The problem with plug-in hybrids? Their drivers.
Plug-in hybrids are often sold as a transition to EVs, but new data from Europe shows we’re still underestimating the emissions they produce.
Stay connected
Get the latest updates from
MIT Technology Review
Discover special offers, top stories, upcoming events, and more.