Paper Gets a Nano Makeover
By splitting up wood pulp into cellulose nanofibers and rearranging the fibers into an entangled porous mesh, researchers have made a nanopaper that is stronger than cast iron and tougher than bone. The nanopaper is seven times stronger and two to three times as stretchy as conventional paper. It could be used to make tough packaging material, filters, membranes, and even car and aircraft parts. “Wood pulp is widely available, and there is the potential to produce nanofibers in very large quantities,” says Lars Berglund, a biocomposites researcher at the Royal Institute of Technology, in Stockholm, Sweden, and coauthor of a paper in Biomacromolecules that describes the new material.
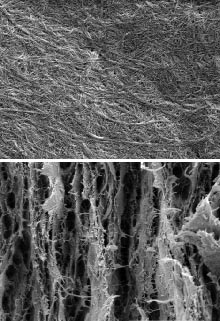
Cellulose, a stiff chain of glucose units, is one of the most abundant natural polymers: it makes up 90 percent of cotton and half of wood. Plant cell walls are made of multiple strands of cellulose bound into fibers typically 5 to 10 nanometers thick. To make regular paper, wood chips are heated with chemicals or mechanical force to create pulp. Aggregated, 30-micrometer-thick bundles of cellulose fibers in the pulp are then intertwined to create sheets.
The new nanopaper is made of much thinner 10-to-40-nanometer-thick fibers. Individual cellulose strands are very robust, Berglund says. “They have properties similar to Kevlar,” he notes. The hydroxyl groups and oxygen molecules on individual nanofibers attach to each other strongly. Even if a sudden impact ruptures the bonds between some of the nanofibers, the defect is small enough that the entire material does not fail. The paper can withstand nearly two-thirds more force than cast iron before it breaks.
The stretchiness comes from the pores in the mesh of nanofibers. When the nanopaper is stretched, there is enough space for the fibers to slip against each other. “You can stretch it up to 10 percent before it fails,” Berglund says. “Conventional paper can stretch a maximum of 3 to 4 percent, then it breaks.”
To make the nanopaper, Berglund and his colleagues first expose wood pulp to enzymes and mechanical force. This separates the pulp into cellulose microfibers. The pulp is passed through a device that uses high-pressure and high velocities in tiny microfluidic channels to create a uniform suspension of nanofibers in water. Finally, the researchers pass the suspension through a filter to create a gel, which they compress to make 100-micrometer-thick sheets.
Berglund says that the dilute, uniform suspension of the nanofibers is critical. It distributes the fibers homogeneously in the paper, making it strong. The porosity of the paper is also crucial. The researchers found that higher porosities led to paper that is stronger–that is, it handles more load per unit area–and tougher, which means that it does not crack easily.
The Swedish work is part of the broader interest in making polymers infused with nanomaterials, such as clay nanoparticles, carbon nanotubes, or graphene, in lieu of using traditional fillers such as glass fibers and carbon black. Compared with carbon nanotubes, cellulose is low cost and abundant. “Cellulose is a fantastic filler because it’s a renewable resource,” says Chris Weder, a professor of macromolecular science and engineering at Case Western Reserve University. “I think it’s a fantastic way to tailor properties of material by way of making nanocomposites.”
Weder says that the researchers will have to test how the nanopaper holds up when wet. He thinks that the mechanical properties of the material might degrade in the presence of water. However, he believes that the new work fills an important knowledge gap. “If you want to understand how nanocomposites behave mechanically, it is helpful to understand how a material formed by the nanofibers themselves would behave.”
Keep Reading
Most Popular
Large language models can do jaw-dropping things. But nobody knows exactly why.
And that's a problem. Figuring it out is one of the biggest scientific puzzles of our time and a crucial step towards controlling more powerful future models.
How scientists traced a mysterious covid case back to six toilets
When wastewater surveillance turns into a hunt for a single infected individual, the ethics get tricky.
The problem with plug-in hybrids? Their drivers.
Plug-in hybrids are often sold as a transition to EVs, but new data from Europe shows we’re still underestimating the emissions they produce.
Stay connected
Get the latest updates from
MIT Technology Review
Discover special offers, top stories, upcoming events, and more.