
Cody Daniel ’11 was a high school student looking for intricate pranks to play on his friends when he stumbled upon the history of hacks at MIT. “I found the subversive culture of MIT very exciting, and I loved the character of the students,” he says. Now, having studied mechanical engineering as one of those students, he is subverting the status quo in a far more serious way—bringing high-volume 3D imaging into the field of pathology, where it has the potential to yield unprecedented scientific insights.
It was through a late-night, quirky conversation at a Steer Roast party at MIT’s Senior House that Daniel first met Todd Huffman, with whom he would later join forces to start 3Scan, a biotechnology company that is changing the way we understand human biology. “He and I got to talking about future technologies, neuroscience, and brain uploading—all these weird, out-there concepts,” he says.
When they met, Huffman was working in the lab of the late Bruce McCormick, a computer scientist and founder of the Brain Networks Lab at Texas A&M University. McCormick had pioneered knife-edge scanning microscope (KESM) technology, which captures high-resolution images of thinly sliced layers of tissue, making it possible to create digital 3D images of cellular structures. The two realized that with Daniel’s engineering and instrumentation background, they could commercialize McCormick’s prototype and use it to automate much of the painstaking work pathologists typically do when they examine samples of organs, tissues, and fluids to study and diagnose disease.
Pathology typically involves cutting tissue samples by hand, placing each sample between two pieces of glass, and studying it under a microscope. The method has remained largely unchanged for more than 150 years. A human can typically process about 12 sample slices per hour.

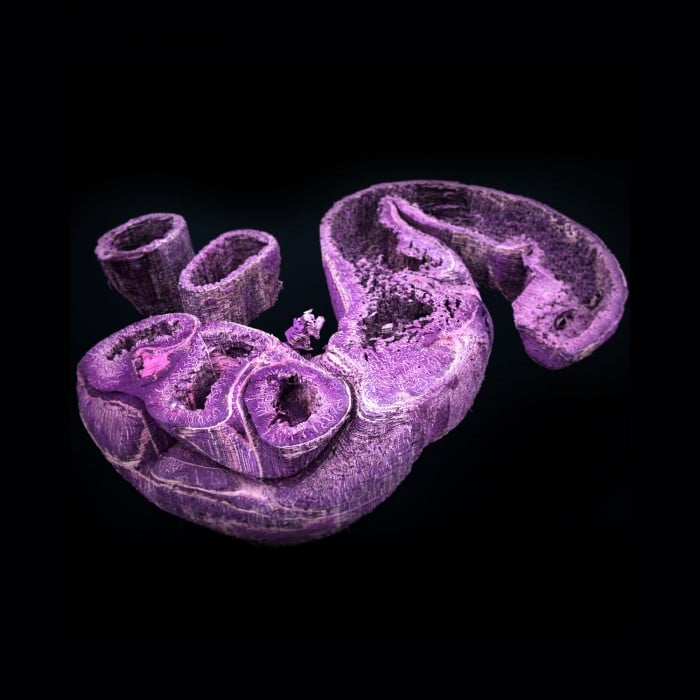
Left: Human muscle tissue; Right: Murine gut
3Scan speeds this process up considerably. Its KESM tool uses an automated diamond knife to cut samples at 1,000 slices per hour while simultaneously scanning an image of each slice, and then layering those scans to create a 3D tissue model with micron-scale resolution, like that of a CT scan.
3Scan’s platform has the potential to illuminate the mechanisms by which biological processes become abnormal, which could improve diagnostics, Daniel says. “There’s only so much tissue one person can see in their lifetime, and if we can build something that looks at pathology across many different demographics, across many different cases and diseases, we can get better insights,” he reasons. “There are also reports that show that pathologists achieve consensus on a case 80% of the time. That’s a pretty high success rate—unless it’s your diagnosis, in which case it’s very scary.”
Once 3Scan’s tools do the imaging and initial analytics, the data is sent back to pathologists, who explore and translate the findings. “We like to imagine the pathologist as the conductor of an orchestra of robots that can go out there and image vast fields of biology,” Daniel says. “The pathologist plays a crucial role in being the informed human perspective, differentiating what is pathologic versus normal within that biology.”
Keep Reading
Most Popular
Large language models can do jaw-dropping things. But nobody knows exactly why.
And that's a problem. Figuring it out is one of the biggest scientific puzzles of our time and a crucial step towards controlling more powerful future models.
How scientists traced a mysterious covid case back to six toilets
When wastewater surveillance turns into a hunt for a single infected individual, the ethics get tricky.
The problem with plug-in hybrids? Their drivers.
Plug-in hybrids are often sold as a transition to EVs, but new data from Europe shows we’re still underestimating the emissions they produce.
Stay connected
Get the latest updates from
MIT Technology Review
Discover special offers, top stories, upcoming events, and more.