Radical New DNA Sequencer Finally Gets into Researchers’ Hands
One day in 1989, biophysicist David Deamer pulled his car off California’s Interstate 5 to hurriedly scribble down an idea. In a mental flash, he had pictured a strand of DNA threading its way through a microscopic pore. Grabbing a pen and a yellow pad, he sketched out a radical new way to study the molecule of life.

Twenty-five years later, the idea is now being commercialized as a gene sequencing machine that’s no larger than a smartphone, and whose effects might eventually be similarly transformative.
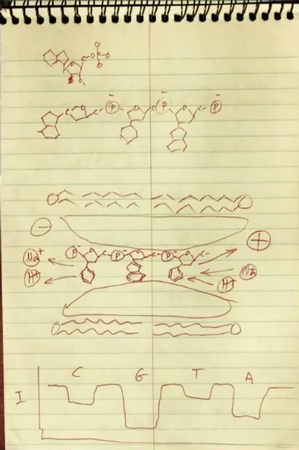
Early versions of the instrument, called the MinION, have been reaching scientific labs over the past few months after long delays (see “10 Breakthrough Technologies 2012: Nanopore Sequencing”). It’s built by a U.K. company, Oxford Nanopore, that has raised $292 million and spent 10 years developing Deamer’s idea into a DNA sequencer unlike any other now available. It is four inches long and gets its power from a USB port on a computer. Unlike other commercial sequencing machines, which can be the size of a refrigerator and require jugs of pricey chemicals, this one measures DNA directly as the molecule is drawn through a tiny pore suspended in a membrane. Changes in electrical current are used to read off the chain of genetic letters, A, G, C, and T.
Scientists with early access to prototypes of the first commercial “nanopore” sequencer say it’s glitchy and error-prone but may still be the way scientists study DNA in the future. After testing it, Mick Watson, a bioinformatics researcher at the Roslin Institute, in Scotland, says nanopore sequencing is a “disruptive technology that could, potentially, dominate the sequencing market for years to come.”
Although researchers say the device is still desperately inaccurate, it can already carry out some unheard-of scientific feats. And then there’s its size. A sequencer this small might one day let police read off a genome from a spot of blood at a crime scene, or permit doctors to pinpoint viruses in the midst of an epidemic. One scientist this month tweeted a picture of the sequencer on his dining room table, decoding DNA.
The MinION is the result of some very high-stakes R&D by Oxford, a 200-person company that’s long has had its eye on the expanding market for high-speed DNA sequencers. Cracking that market won’t be easy. About 90 percent of DNA data is produced on sequencing machines from a single company, Illumina of San Diego (see “50 Smartest Companies: Illumina”). Its sequencers are so good that most of its competitors have ended up in Chapter 11 or retreated in ignominy.
But now some big companies are betting that nanopores could be the technology to break Illumina’s lucrative monopoly. Roche, which made a failed attempt to acquire Illumina in 2012, this year spent $125 million to buy Genia Technologies, a small nanopore company based in California, and invested in another, Stratos Genomics. Hitachi is also working on nanopore technology, as are startups like Electronic Biosciences.
Deamer says the idea of nanopore sequencing occurred to him in 1989, just three years after the first automated DNA sequencers were introduced. He had been trying to build artificial cells, spherical blobs of fat that could pump molecules in and out through microscopic pores the way real cells do. His flash of insight was that a molecule passing through one of these pores—especially a long molecule like DNA—would continuously change the blob’s electrical properties. That would create a signal you could measure.
It took another 25 years before the MinION was developed. That’s because the technical problems were so daunting. Each DNA letter is only about half a nanometer from the next and some differ by just an atom or two, so they’re hard to tell apart. And how could you pull the string of DNA letters through the pore, suspended in a layer one-100,000th as thick as a hair—much less at a rate of 30 letters a second, as the MinION does?
“There were a lot of smart people saying this is physically not possible to do,” says Jeffery Schloss, head of the division of genome sciences at the National Human Genome Research Institute, in Bethesda, Maryland. “Well, it definitely is possible. The question now is if it’s good enough to be used in a practical way.”
Oxford has disappointed its fans before. When it first announced the MinION, in 2012, expectations soared off the charts (see “Why a Portable DNA Device Could Yield Better Data”). But when the promised machines failed to appear, some started to wonder if the sequencer was vaporware.
By this spring, Oxford had the bugs worked out—enough, at any rate, to start mailing out beta versions of the nanopore sequencer to 500 hand-picked labs it is collaborating with. Another early creator of the technology, Mark Akeson, who works alongside Deamer in the bioengineering department at the University of California, Santa Cruz, says since June he’s received two updated versions, a sign of how quickly Oxford is scrambling to improve the device.
To the technology’s original inventors, the arrival of any commercial nanopore sequencer is a milestone. “The idea they can FedEx a 100-gram machine that actually works is pretty amazing,” says Akeson. While both he and Deamer have extensive financial ties to Oxford, they say it’s obvious to everyone that the MinION is just the start. “It’s doing okay,” says Deamer. “The question is accuracy, but improvements are coming, believe me.”
Several scientists using the device say it correctly reads only 60 to 85 percent of DNA letters. That’s the bad news. (Illumina’s machines, some of which can cost $1 million, are more than 99.9 percent accurate.) Yet nanopore sequencing is so different that even a machine that’s error-prone might be a boon to science.
One reason is that today’s fastest sequencers decode DNA after it’s been shredded into tiny bits, reading off just 150 letters at a time. Those bits then have to be puzzled back together to create a genome. Even with a supercomputer, the puzzle often can’t be solved—there can be too many repeated sequences or parts that go missing.
Nanopore sequencing may help because it produces what scientists call “long reads.” For instance, Akeson says this summer his lab read across a continuous strand of human DNA that was 79,000 letters long. That’s probably a record. Like having the edges of a puzzle, long reads make it much easier to reassemble a genome, especially of a species never studied before.
The eventual commercial price of the sequencer isn’t known. Nick Loman, a researcher at the University of Birmingham, said his lab has paid about $1,000 each to get hold of the machines, though disposable cells containing the matrix of pores (about 2,000 of them) would cost extra. He said he hoped the price would stay “very cheap” in the future.
Last week, Oxford’s chief technology officer, Clive Brown, said further instruments would be announced soon. One, dubbed PromethION, will use as many as 100,000 pores in parallel and could compete with Illumina’s top-of-the line sequencing system, which was introduced early this year (see “Does Illumina Have the First $1,000 Genome?”) to labs interested in sequencing hundreds of thousands of human genomes for medical research.
Keep Reading
Most Popular
Large language models can do jaw-dropping things. But nobody knows exactly why.
And that's a problem. Figuring it out is one of the biggest scientific puzzles of our time and a crucial step towards controlling more powerful future models.
How scientists traced a mysterious covid case back to six toilets
When wastewater surveillance turns into a hunt for a single infected individual, the ethics get tricky.
The problem with plug-in hybrids? Their drivers.
Plug-in hybrids are often sold as a transition to EVs, but new data from Europe shows we’re still underestimating the emissions they produce.
Stay connected
Get the latest updates from
MIT Technology Review
Discover special offers, top stories, upcoming events, and more.