Detecting the Danger Signs of Glaucoma
Researchers at Purdue University have engineered a tiny, wireless eye implant that monitors glaucoma by continuously measuring intraocular pressure–a primary risk factor for the disease. Scientists say that such an implant would help ophthalmologists catch and treat problematic spikes in pressure that would otherwise go unnoticed.
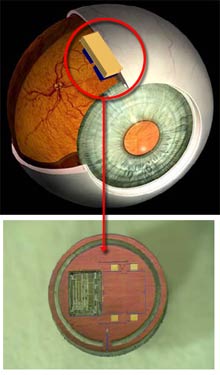
“People with glaucoma go to the doctor every six months to check their eye pressure,” says Pedro Irazoqui, an assistant professor of biomedical engineering at Purdue, who is leading the research. “But the problem is that pressure doesn’t spike within months: it spikes within hours, sometimes minutes. Once there’s a spike, there’s a limited amount of time you have to act before it does permanent damage to the optic nerve. We wanted to design a device that monitors pressure continuously, so that if there is a spike, we’d be guaranteed to catch it.”
Glaucoma is the second leading cause of blindness in the world after cataract. According to the Glaucoma Research Foundation, three million Americans have the disease, although only half know it. There are no outward symptoms other than perhaps a gradual loss of peripheral vision.
The disease itself is caused by a buildup of intraocular fluid, called aqueous humor, which drains from the pupil through a system of canals. In healthy people, this drainage system keeps eye pressure within a normal range–about 10 to 21 millimeters of mercury. People with glaucoma tend to have clogged canals, which causes a buildup in fluid, which in turn creates pressure within the eye that exceeds 21 millimeters of mercury. This elevated pressure, if left untreated, can permanently damage the optic nerve, leading to compromised vision and, in some cases, blindness.
If the disease is caught early enough, ophthalmologists can give patients drugs to relieve the pressure and stave off further damage. But regular examinations in a doctor’s office are inadequate for charting a glaucoma patient’s progress. “It’s not uncommon that a patient doesn’t have a spike in the office, and all of a sudden, you see glaucoma peaking,” says Shakeel Shareef, a glaucoma specialist and associate professor at the University of Rochester Eye Institute, who was not involved in the research. According to Shareef, a continuous monitoring device, like those used to measure an irregular heartbeat, would be a welcome tool.
Toward that end, Irazoqui and his colleagues have designed a tiny microchip, to be implanted between two layers of the eye. The sensor is designed to measure intraocular pressure and wirelessly transmit the data to a nearby computer. A doctor can then access the data and review it for possible warning signs. At present, Irazoqui’s team has engineered a prototype of the sensor, although he and his colleagues have yet to test it on animals. The researchers plan to present the details of their research later this month at the Engineering in Medicine and Biology Society’s conference in Lyon, France.
Other groups are also developing continuous monitoring devices for glaucoma, including a team based in Lausanne, Switzerland, that’s working to embed wireless microchip sensors in soft contact lenses. The researchers have developed and tested a wired version in humans and are now working on a wireless version.
One of the major obstacles in creating this type of device is designing a tiny but highly functional chip that uses very little power. Irazoqui’s group has overcome this problem in part by designing the sensor to run on nanowatts rather than on microwatts. “We use a million times less power, which means we can get a million times more on the circuit,” says Irazoqui, who declined to release further details until he presents the research later this month. The researchers will begin testing the implant in animals by December.
Shareef says that such a sensor, once available to patients and doctors, will not only provide better diagnosis for glaucoma, but it may also hold patients accountable for keeping track of their health. “You may have patients come in, and their pressure is wonderful in the office, but glaucoma is progressing rapidly,” says Shareef. “It may be that patients are not taking their medications, and before they go to the doctor’s, they take meds for two days, and the pressure will go down temporarily. But with a device like this, you can tell if they’re cheating themselves.”
Keep Reading
Most Popular
Large language models can do jaw-dropping things. But nobody knows exactly why.
And that's a problem. Figuring it out is one of the biggest scientific puzzles of our time and a crucial step towards controlling more powerful future models.
How scientists traced a mysterious covid case back to six toilets
When wastewater surveillance turns into a hunt for a single infected individual, the ethics get tricky.
The problem with plug-in hybrids? Their drivers.
Plug-in hybrids are often sold as a transition to EVs, but new data from Europe shows we’re still underestimating the emissions they produce.
Stay connected
Get the latest updates from
MIT Technology Review
Discover special offers, top stories, upcoming events, and more.