Neuron Control
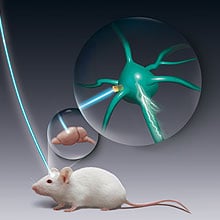
In his psychiatry practice at the Stanford Medical Center, Karl Deisseroth sometimes treats patients who are so severely depressed that they can’t walk, talk, or eat. Intensive treatments, such as electroconvulsive therapy, can literally save such patients’ lives, but often at the cost of memory loss, headaches, and other serious side effects. Deisseroth, who is both a physician and a bioengineer, thinks he has a better way: an elegant new method for controlling neural cells with flashes of light. The technology could one day lead to precisely targeted treatments for psychiatric and neurological disorders; that precision could mean greater effectiveness and fewer side effects.
While scientists know something about the chemical imbalances underlying depression, it’s still unclear exactly which cells, or networks of cells, are responsible for it. In order to identify the circuits involved in such diseases, scientists must be able to turn neurons on and off. Standard methods, such as electrodes that activate neurons with jolts of electricity, are not precise enough for this task, so Deisseroth, postdoc Ed Boyden (now an assistant professor at MIT; see “Engineering the Brain”), and graduate student Feng Zhang developed a neural controller that can activate specific sets of neurons.
They adapted a protein from a green alga to act as an “on switch” that neurons can be genetically engineered to produce (see “Artificially Firing Neurons,” TR35, September/October 2006). When the neuron is exposed to light, the protein triggers electrical activity within the cell that spreads to the next neuron in the circuit. Researchers can thus use light to activate certain neurons and look for specific responses–a twitch of a muscle, increased energy, or a wave of activity in a different part of the brain.
Deisseroth is using this genetic light switch to study the biological basis of depression. Working with a group of rats that show symptoms similar to those seen in depressed humans, researchers in his lab have inserted the switch into neurons in different brain areas implicated in depression. They then use an optical fiber to shine light onto those cells, looking for activity patterns that alleviate the symptoms. Deisseroth says the findings should help scientists develop better antidepressants: if they know exactly which cells to target, they can look for molecules or delivery systems that affect only those cells. “Prozac goes to all the circuits in the brain, rather than just the relevant ones,” he says. “That’s part of the reason it has so many side effects.”
In the last year, Deisseroth has sent his switch to more than 100 research labs. “Folks are applying it to all kinds of animals, including mice, worms, flies, and zebrafish,” he says. Scientists are using this and similar switches to study everything from movement to addiction to appetite. “These technologies allow us to advance from observation to active intervention and control,” says Gero Miesenböck, a neuroscientist at Yale University. By evoking sensations or movements directly, he says, “you can forge a much stronger connection between mental activity and behavior.”
Deisseroth hopes his technology will one day become not just a research tool but a treatment in itself, used alongside therapies that electrically stimulate large areas of the brain to treat depression or Parkinson’s disease. By activating only specific neurons, a specially engineered light switch could limit those therapies’ side effects. Of course, the researchers will need to solve some problems first: they’ll need to find safe gene-therapy methods for delivering the switch to the target cells, as well as a way to shine light deep into the brain. “It’s a long way off,” says Deisseroth. “But the obstacles aren’t insurmountable.” In the meantime, neuroscientists have the use of a powerful new tool in their quest to uncover the secrets of the brain.
Keep Reading
Most Popular
Large language models can do jaw-dropping things. But nobody knows exactly why.
And that's a problem. Figuring it out is one of the biggest scientific puzzles of our time and a crucial step towards controlling more powerful future models.
How scientists traced a mysterious covid case back to six toilets
When wastewater surveillance turns into a hunt for a single infected individual, the ethics get tricky.
The problem with plug-in hybrids? Their drivers.
Plug-in hybrids are often sold as a transition to EVs, but new data from Europe shows we’re still underestimating the emissions they produce.
Google DeepMind’s new generative model makes Super Mario–like games from scratch
Genie learns how to control games by watching hours and hours of video. It could help train next-gen robots too.
Stay connected
Get the latest updates from
MIT Technology Review
Discover special offers, top stories, upcoming events, and more.