The world’s first Gattaca baby tests are finally here

Anxious couples are approaching fertility doctors in the US with requests for a hotly debated new genetic test being called “23andMe, but on embryos.”
The baby-picking test is being offered by a New Jersey startup company, Genomic Prediction, whose plans we first reported on two years ago.
The company says it can use DNA measurements to predict which embryos from an IVF procedure are least likely to end up with any of 11 different common diseases. In the next few weeks it's set to release case studies on its first clients.
Handed report cards on a batch of frozen embryos, parents can use the test results to try to choose the healthiest ones. The grades include risk estimates for diabetes, heart attacks, and five types of cancer.
According to flyers distributed by the company, it will also warn clients about any embryo predicted to become a person who is among the shortest 2% of the population, or who is in the lowest 2% in intelligence.
The test is straight out of the science fiction film Gattaca, a movie that’s one of the inspirations of the startup’s CEO, Laurent Tellier. The company’s other cofounders are testing expert Nathan Treff and Stephen Hsu, a Michigan State University administrator and media pundit.
So far, fertility centers have not leaped at the chance to offer the test, which is new and unproven. Instead, prospective parents are learning about the designer baby reports through word of mouth or news articles and taking the company’s flyer to their doctors.
One such couple recently turned up at New York University’s fertility center in Manhattan, says David Keefe, who is chairman of obstetrics and gynecology there. “Right off the bat it raises all kind of questions about eugenics,” he says.
Keefe, who has seven children, worries that couples who think they can choose kids from a menu could be disappointed. “It’s fraught with parenting issues,” he says. “So many couples just need to feel they have done enough.”
Picking your baby
The company’s project remains at a preliminary stage. While some embryos have been tested by the company, Tellier, the CEO, says he is unsure if any have yet been used to initiate a pregnancy.
The test is carried out on a few cells plucked from a days-old IVF embryo. Then Genomic Prediction measures its DNA at several hundred thousand genetic positions, from which it says it can create a statistical estimate, called a “polygenic score,” of the chance of disease later in life.
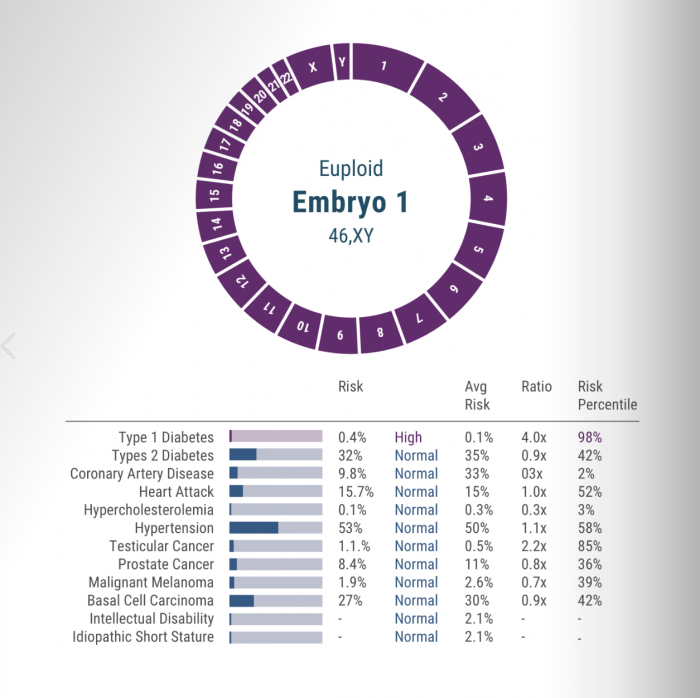
In October, the company pitched the test, which it calls “LifeView,” from a trade-show booth at the annual meeting of fertility doctors in Philadelphia. A promotional banner read: “She has your partner’s ears and smile. Just not their risk of diabetes.”
Criticism of the company from some genetics researchers has been intense.
“It is irresponsible to suggest that the science is at the point where we could reliably predict which embryo to select to minimize the risk of disease. The science simply isn’t there yet,” says Graham Coop, a geneticist at the University of California, Davis, and a frequent critic of the company on Twitter.
The company has raised several million dollars in venture capital from investors including People Fund, Arab Angel, Passport Capital, and Sam Altman, the chairman of Y Combinator and CEO of OpenAI.
At an investor event last April, Genomic Prediction compared itself to 23andMe for IVF clinics and boasted it was preparing for a “massive marketing push.”
Our reporting suggests the company has struggled both to validate its predictions and to interest fertility centers in them. Its customers so far seem to be a scattering of individuals from around the world with specific family health worries. The company declined to name them, citing confidentiality.
The company is expected to soon present its first case reports, describing clients and their embryo test results. One case involves a married gay couple who have begun IVF using donor eggs and plan to employ a surrogate mother. That couple wants a child with a low risk for breast cancer.
How will it be used?
Genomic Prediction thinks it can piggyback on the most common type of “preimplantation” embryo test, which screens days-old embryos for major chromosome abnormalities, called aneuploidies. Such testing has become widespread in fertility centers for older mothers and is already employed in nearly a third of IVF attempts in the US. The new predictions could be added to it.
Fertility centers can also order tests for specific genetic diseases, such as cystic fibrosis, where a gene measurement will give a definite diagnosis of what embryo inherited the problem The new polygenic tests are more like forecasts, estimating risk for common diseases on the basis of variations in hundreds or thousands of genes, each with a small effect.
In a legal disclaimer, the company says it can’t guarantee anything about the resulting child and that the assessment “is NOT a diagnostic test.”
Santiago Munne, an embryo testing expert and entrepreneur, thinks patients already undergoing aneuploidy testing would likely want the add-on test, but that doctors could object if it introduces uncertainty: “For monogenic disease, if the embryo is abnormal, we will tell you, and it is. With a risk score, it may be affected. And some patients will only have embryos with higher risks. Then what?”
As well, he says it won’t be possible with a test to optimize a child for many features at once: “My personal opinion is once you start looking, some embryos will be brighter, some will be taller, some will have longevity, and none will have those qualities all together. And in an IVF cycle, you produce maybe six embryos on average. You won’t be able to get all the traits that you want.”
Despite such inherent limits, there’s a bigger plan afoot. Treff, the startup's chief scientist, believes even fertile couples might begin to undergo IVF just so they can select the best child. “I do believe this is going to be the future … we can start to ... reduce the incidence of disease in humans through IVF,” Treff told an audience at a conference in China last month.
How many people will be willing to go through the trouble of IVF if they don’t need it to have a baby? IVF involves weeks of hormone shots and two medical procedures (one to collect eggs, another to implant the embryos) and typically costs around $15,000. Add to that the company’s fee to test embryos, which is $1,000, plus $400 for each embryo scored.
“If someone is fertile, unless there is a family history of disease, I don’t think that it is going to be popular,” says Munne.
Can you get a smarter baby?
Genomic Prediction has so far won the most attention for the possibility of using genetic scores to pick the most intelligent children from a petri dish. It has tried to distance itself from the controversial concept, but that’s been difficult because Hsu, a cofounder, is frequently in the media discussing the idea.
Hsu told The Guardian this year that “accurate IQ predictors will be possible if not in the next five years, the next 10 years certainly.” He says other countries, or the ultra-wealthy, might be the first to try to boost IQ in their kids this way.
During his talk in China, Treff called improving intelligence via embryo selection an application that “many people think is unethical." In private, Treff tells other scientists he thinks it's doable, but wants to promote the technology for medical purposes only.
For now, the company is limiting itself to alerting parents to embryos it predicts will be the least intelligent, with the highest chance of an IQ which qualifies as “intellectual disability” according to psychiatric manuals.
Some experts see a transparent maneuver to avoid controversy. “They say they’re going to test for the medical condition of intellectual disability, not for the smartest embryos, because they know people are going to object to that,” says Laura Hercher, who trains genetic counselors at Sarah Lawrence College. “They are trying to slide, slide into traits without admitting as much.”
A May report from the Hebrew University of Jerusalem found that trying to pick the tallest or smartest embryos might not work particularly well. Researchers there estimated that using polygenic scores to locate the tallest or smartest child from a batch of sibling embryos would result in an average gain of 2.5 centimeters in height, and less than three IQ points.
“They modeled what everyone is scared of happening,” Treff said of that study. “It’s not what we are doing.”
The predictions, however, could be more effective at helping people avoid children with specific diseases. Treff, during his speech in China, said that a couple choosing between two embryos would see, on average, a 45% reduction in risk for type 1 diabetes. That is a serious disease from which Treff suffers and which runs in families, although it has complex causes. The more embryos there are to choose from, he says, the more the risk will go down.
Demand for the test
Patients and doctors are mostly on their own when it comes to deciding if the tests really work. While federal and state agencies do oversee laboratory accuracy, the oversight is limited to whether analytes like DNA are correctly measured, not what they mean. So Genomic Prediction doesn’t need to prove that the test is useful before selling it. In fact, it could take decades to ascertain if tested kids fare better than others.
And it’s not only whether the test works or not. Uptake will depend on demand from patients and the degree of pushback from doctors and genetic counselors. In the US, tests for gender—that is, picking a boy or a girl embryo—are accepted and relatively routine. But that’s never become the case for choosing eye color, which is also possible. “In terms of eye color, the pressure not to do it, to not offer it, was met with a weak market demand. So it doesn’t exist,” says Hercher.
Genomic Prediction provided a map of 12 fertility clinics it says will order its test, including five in the US and others in Nigeria, Peru, Thailand, and Taiwan.
MIT Technology Review was able to independently locate two IVF clinics where customers have recently requested the embryo predictions. Michael Alper, founder of Boston IVF, one of the world’s largest fertility clinics, says his center was approached by a couple a few weeks ago but he decided the request needs to be weighed by the center’s ethics committee before he would agree to order it.
“This is the first case we have had,” says Alper. “To me it’s a 23andMe type of prediction: there’s a propensity, but how strong? That is the problem. We don’t have any problem testing for cystic fibrosis—that is a lethal disease, it strikes young. But we are not there yet with these other tests. It’s soft; it’s not that predictive.”
At NYU, Keefe says the test raises profound questions. His center is in Midtown Manhattan, just blocks from a hub of finance and legal offices. He says his clientele are typically well-off professionals, “people who have programmed everything” in life and feel “they are in control.” They sometimes even ask out loud if a mere doctor is smart enough to help them.
The case he is working on involves a family that has two children with autism. They now want a child without the condition, and they hope the intelligence feature of the test will help them. Treff says he counseled the family that the Genomic Prediction test wasn’t likely to help—autism can have specific genetic causes that the intelligence prediction isn’t designed to capture.
Yet the family remains interested. They want to do whatever they can to have a healthy kid. Keefe says he’s so far supporting their choice, but he is concerned by all that it implies. “There is potential psychological harm to the kid,” he says. “God forbid the kids ends up with autism after spending this money.”
Deep Dive
Biotechnology and health
How scientists traced a mysterious covid case back to six toilets
When wastewater surveillance turns into a hunt for a single infected individual, the ethics get tricky.
An AI-driven “factory of drugs” claims to have hit a big milestone
Insilico is part of a wave of companies betting on AI as the "next amazing revolution" in biology
The quest to legitimize longevity medicine
Longevity clinics offer a mix of services that largely cater to the wealthy. Now there’s a push to establish their work as a credible medical field.
There is a new most expensive drug in the world. Price tag: $4.25 million
But will the latest gene therapy suffer the curse of the costliest drug?
Stay connected
Get the latest updates from
MIT Technology Review
Discover special offers, top stories, upcoming events, and more.