Cancer? This researcher says he can see it in your blood
Cancer researcher Bert Vogelstein has won plenty of prizes for his work on the genetic mutations that cause cancer. What he always wanted, though, was a way to spot those mutations early on, when cancer can more easily be treated.
Now a group of investors is putting $110 million behind Vogelstein’s dream and creating a company called Thrive Earlier Detection. It’s the latest entry in a costly race to develop the first screening tests able to spot a range of cancers from a simple blood draw. A competing company, Grail, has raised more than $1 billion to develop a general-purpose cancer detection test.
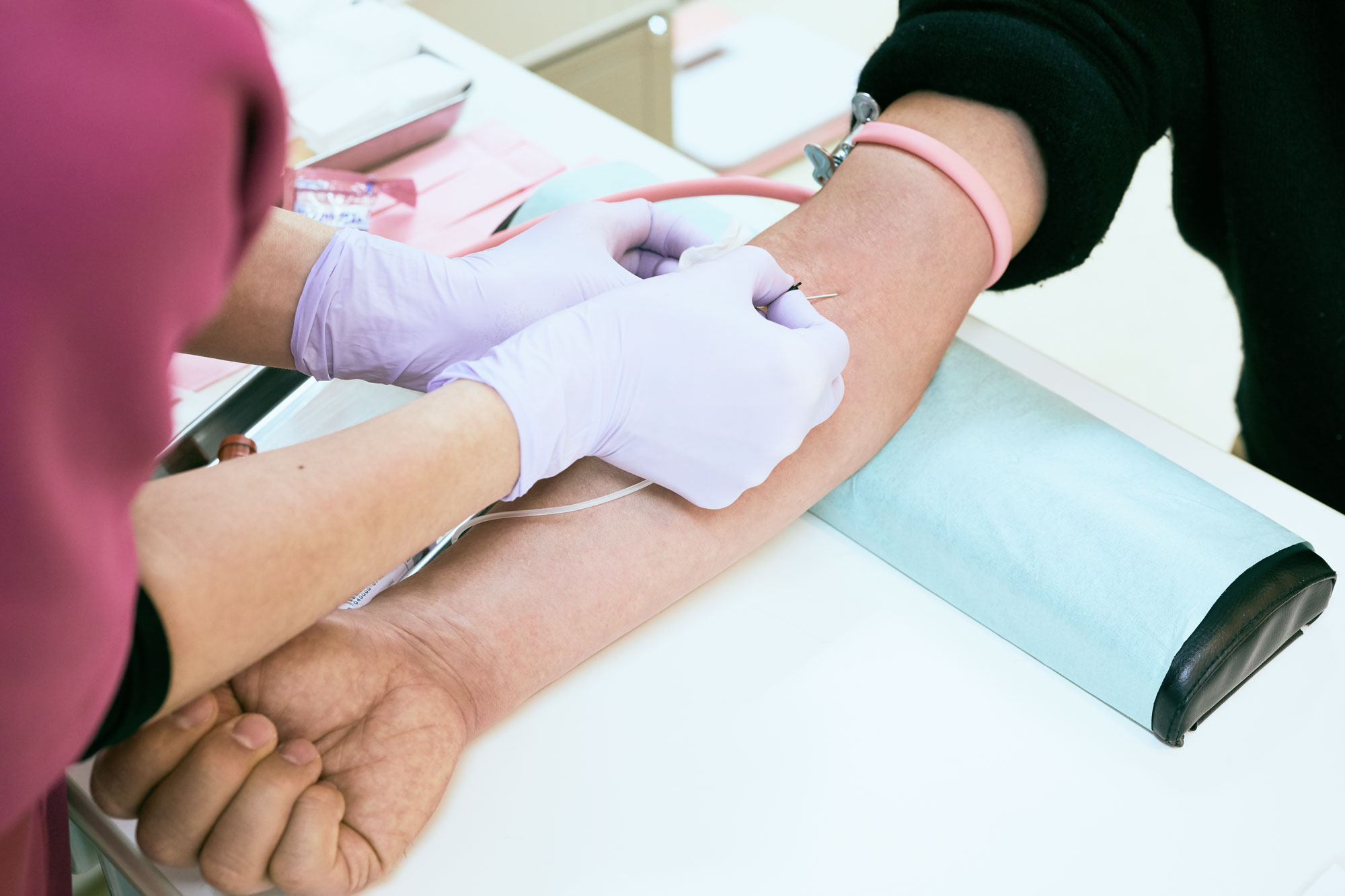
The blood-screening concept, called a “liquid biopsy,” owes much to early efforts by Vogelstein and his Johns Hopkins University colleague Kenneth Kinzler. It relies on the fact that tumors release mutation-riddled DNA and distinctive proteins into the bloodstream. These can sometimes be detected even in someone with no outward signs of cancer.
Along with oncologist Nickolas Papadopoulos, the Hopkins researchers last year described a blood test that looked at 16 genes and eight proteins and was able to spot several cancers, including those of the stomach, ovaries, and liver—though in the blood of people already known to be sick. Now the group is wrapping up a study undertaken with Geisinger, a large health system in Pennsylvania, on 10,000 apparently healthy women aged 65 to 75, to see how often the test finds otherwise hidden cancer in their bodies. Women who test positive are placed in a whole-body PET scanner to search for tumors.

Doctors at Geisinger say they have found cancers this way, and treated them. But it will be another year before they release their findings. “We have some anecdotes but are cognizant of overselling the technology. It’s too early to say if the promise is going to hold up,” says Adam Buchanan, a doctor at Geisinger helping to lead the project.
Screening tests are judged by how many real cancers they find and how many false alarms they set off. For this one to work out, Vogelstein says, it will need to spot perhaps 20% of actual cancers. “I almost always get the same response: it’s ‘Why can’t you detect 70% of them?’” says Vogelstein. In reality, though, finding a fifth of lurking cancers at a stage when they might be cured could do more than any new drugs, which typically extend life just a few weeks.
The trickier issue is when a test falsely indicates cancer is present. For any screening test aimed at the masses, such false positives need to be kept to no more than 1 in 100, says Vogelstein. “If you detect a lot of false positives, you are going to do follow-up tests that are unneeded,” says Vogelstein. That, notes Buchanan, carries both “emotional and financial costs.”
The Hopkins testing idea was being commercialized by a company called PapGene, which is now changing its name to Thrive as it gets an infusion of $110 million from a group of elite investors that include Third Rock, a venture firm, and the insurer Blue Cross Blue Shield. The rebooted company will be led by Steven Kafka, formerly with Foundation Medicine, a DNA testing company in Cambridge, Massachusetts.
Bernie Marcus, a wealthy cofounder of Home Depot, who funded the study at Geisinger with a $15 million grant (and has committed $35 million more to follow an additional 40,000 people) won’t have a role or financial stake in Thrive, according to Vogelstein: “He just wanted a blood test for cancer developed during his lifetime. He doesn’t care how it is done, so long as it is done well.”
Thrive will need to gather enough data to convince insurers to pay for the tests, which Kafka says could cost around $500. To do that, it isn’t expecting to have to prove the test saves lives. Even the value of widely used cancer screening tests, like mammography, remain in debate even after decades. Instead, Thrive is likely to try to market a test based on different measures, such as showing that cancers can get found earlier than usual.
“If you can show you can detect cancers earlier, it’s a reasonable extrapolation that you can save lives,” says Vogelstein. “To actually prove it, though, requires hundreds of thousands, or millions, of patients. You don’t want to wait that long.”
Deep Dive
Biotechnology and health
How scientists traced a mysterious covid case back to six toilets
When wastewater surveillance turns into a hunt for a single infected individual, the ethics get tricky.
An AI-driven “factory of drugs” claims to have hit a big milestone
Insilico is part of a wave of companies betting on AI as the "next amazing revolution" in biology
The quest to legitimize longevity medicine
Longevity clinics offer a mix of services that largely cater to the wealthy. Now there’s a push to establish their work as a credible medical field.
There is a new most expensive drug in the world. Price tag: $4.25 million
But will the latest gene therapy suffer the curse of the costliest drug?
Stay connected
Get the latest updates from
MIT Technology Review
Discover special offers, top stories, upcoming events, and more.