Device Lets Cancer Patients Assist in Their Own Breast Reconstruction, at Home
After being diagnosed with breast cancer in December 2015, Cindy Shanker, 67, of Los Gatos, California, got a double mastectomy on the advice of her oncologist. A few months later, her surgeon asked if she wanted to join a clinical trial for an experimental type of tissue expansion for breast reconstruction.
With the rate of double mastectomies on the rise in recent years, more patients than ever are facing the decision of whether to get breast reconstruction following surgery. For decades, that has required injections of saline that expand the tissue around the breast area to create space for an implant. The process can be painful and can take up to two months of weekly appointments in a physician’s office. But a new device promises to be more comfortable, convenient, and time-efficient than the traditional method.
Shanker says she didn’t hesitate to join the Columbia University-led clinical trial testing a handheld, needleless device called AeroForm that uses carbon dioxide instead of saline to slowly expand the breast tissue. The best part, Shanker says, was that she could use the device at home without the need for multiple doctor’s appointments.
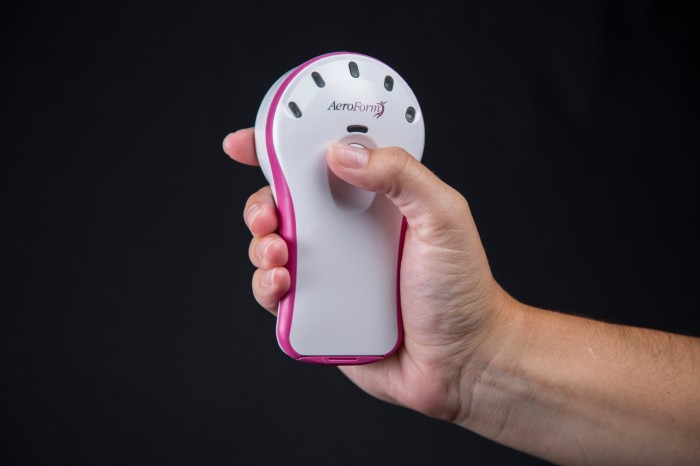
“It gave me a feeling of control over my recovery,” she says.
Jeffrey Ascherman of the Columbia University Department of Surgery, who is principal investigator of the trial, says the device could represent the first major change in breast tissue expansion in 40 years.
Following a mastectomy, a surgeon places a temporary silicon tissue expander underneath the chest muscle. Traditional expanders are filled gradually with injections of saline through a tiny valve mechanism located inside the expander. As the expanders fill, the chest tissue is stretched to make room for a breast implant.
By contrast, the AeroForm device uses carbon dioxide to fill the expander, which is compressed in a cartridge in the device. An internal valve controls the delivery of the gas and limits the amount of gas that is released. Using a wireless dose controller, patients and physicians can release small infusions of carbon dioxide up to three times per day to slowly inflate the expander.
James Appel, a plastic surgeon at Presbyterian Medical Center in North Carolina whose practice was a clinical trial site, says the process is “more gradual” than using saline, which cuts down on discomfort. Though patients of his who used the device don’t have anything to compare it to, they have reported being “very satisfied” with the AeroForm device, he says.
In a Phase 2 trial that enrolled 150 participants across 17 sites in the U.S., women using the AeroForm device completed tissue expansion in an average of 21 days, compared to 46 days for the saline group. AeroForm was deemed as safe as the traditional saline method. In 98 percent of cases, participants reported that the device was easy to use. Those who used the AeroForm device also progressed to breast implantation one month faster on average than the saline group. The results are forthcoming in the December issue of Plastic and Reconstructive Surgery.
AeroForm’s maker, AirXpanders of Palo Alto, California, has filed an application with the U.S. Food and Drug Administration and is awaiting approval. The company hopes to receive a decision by the end of 2016. Meanwhile, the device is in use in Australia, having gained regulatory approval there in 2013.
Ascherman says surgeons have made attempts to improve breast tissue expansion over the years, such as a self-filling device involving a hydrogel that expanded by osmosis, but no other approaches have shown to be better than the saline method.
Not all breast cancer patients who seek mastectomies stand to benefit from the device, though. Appel says the AeroForm trials are not testing the device in women who are smokers, taking steroids, or those who may be prone to infection.
Keep Reading
Most Popular
Large language models can do jaw-dropping things. But nobody knows exactly why.
And that's a problem. Figuring it out is one of the biggest scientific puzzles of our time and a crucial step towards controlling more powerful future models.
How scientists traced a mysterious covid case back to six toilets
When wastewater surveillance turns into a hunt for a single infected individual, the ethics get tricky.
The problem with plug-in hybrids? Their drivers.
Plug-in hybrids are often sold as a transition to EVs, but new data from Europe shows we’re still underestimating the emissions they produce.
Google DeepMind’s new generative model makes Super Mario–like games from scratch
Genie learns how to control games by watching hours and hours of video. It could help train next-gen robots too.
Stay connected
Get the latest updates from
MIT Technology Review
Discover special offers, top stories, upcoming events, and more.