The Practical Activist
On a freezing day in March, Linda Griffith, a biological engineer at MIT, joined thousands of women in a march on the National Mall in Washington, D.C., to raise awareness about endometriosis. In that disease, tissue from the uterine lining grows outside the uterus, often causing pain and infertility. Griffith marched with women from around the country, including Amber Wilson, a 30-year-old from Tennessee who said she’d endured years of unexplained pain until finally undergoing surgery last fall; the growth associated with the disease had acted as a “glue,” her surgeon told her, pinning her fallopian tubes to her ovaries and her ovaries to her pelvis. Griffith, too, suffers from endometriosis and has undergone nine surgeries, including an emergency procedure the week of September 11, 2001. She shares the mission of calling attention to a disease that affects one in 10 women of childbearing age.
Yet her activism has largely taken a different form. In 2009, she became co-director, along with surgeon Keith Isaacson, of a new Center for Gynepathology Research at MIT. One of the goals, she says, is to frame endometriosis—and other “below-the-waist” women’s health issues—in terms of technical puzzles that will appeal to male and female researchers alike. If you say you’re “curled up on the floor in a fetal position” because of menstrual pain, a lot of male researchers will act like “deer in the headlights,” she says. But if you say, “Let’s do a nonnegative matrix factorization”—a kind of mathematical analysis—they will do it. “If there’s a language to talk about the disease at the frontier of science, they will want to talk about it,” she says.
Griffith’s approach has already led to advances in the basic understanding of endometriosis. In February, her lab reported on a group of immune markers that, when elevated together in certain patients, tended to be associated with especially painful cases of the disease. The immune signature her team identified could in theory lead to a minimally invasive test that would allow doctors to diagnose and treat endometriosis in those patients earlier, potentially reducing the damage. That would be a major advance, since a definitive diagnosis currently requires surgery, and women with endometriosis wait, on average, more than a decade before confirming that they have the disease. Griffith’s work is the most exciting paper to come out in this field in a long time, says Hugh Taylor, the chair of the department of obstetrics, gynecology, and reproductive sciences at the Yale School of Medicine.
Given this acclaim, it is remarkable that Griffith, 54, has not specialized in reproductive health for most of her career. She was well known first as a pioneer in tissue engineering, with an emphasis on liver and bone. “I never imagined focusing on ‘women’s stuff,’” she says. When she was growing up in rural Georgia, her family downplayed any distinctions based on gender. She spent time running barefoot outdoors, playing with snakes and frogs. “I did the same chores my brother did,” she says: she mowed the lawn, repaired the car, and helped her father, a mechanical engineer, build a stereo system for the house. “I just got exposed to things in a very natural way,” she says. Griffith also learned to knit, crochet, and sew, and as a child she made most of her own clothes. “I just loved making things,” she says. “And I loved math.” When her older brother learned calculus as a college freshman, he taught her the rudiments; it seemed, she says, like “a natural thing anyone would want to know.”
Griffith went on to study chemical engineering, and after receiving her PhD from the University of California at Berkeley in 1988, she arrived at MIT as a postdoctoral fellow in the laboratory of Robert Langer, now an Institute Professor. At the time, Langer and his colleagues were beginning to make three-dimensional polymer scaffolds to create new tissues and organs. Griffith conducted some of the crucial early studies determining which polymers worked best, says Langer. She focused on liver cells, analyzing which materials supported the optimal rate of growth and did not cause cells to de-differentiate, or lose their mature function. Ultimately, she showed that synthetic materials could support cell proliferation as well as or better than natural polymers like collagen. According to Langer, her work was central to the birth of tissue engineering, which today is a major field that includes the development of tissues and organs for wound healing and reconstruction. (Skin for burn victims and tissue-engineered bladders are two examples.) As a postdoc, she was also “one of the most dynamic people you would ever see,” adds Langer. “She gave unbelievable talks and got people so excited.”
Griffith also helped create the well-known Vacanti mouse, which had cartilage on its back in the shape of an ear. The project was motivated by a plastic surgeon, Joe Upton, who wanted to make shape-specific cartilage for reconstructive procedures in children, says Griffith. Her MGH collaborator Charles Vacanti had “shown you could grow cartilage by combining cells with a degradable suture,” but “it was just blobs.” Griffith’s contribution was to make a porous scaffold in the shape of an ear; it would dissolve slowly while the tissue was growing. “The project really got off the ground when I ran into Dr. Upton at a bed and breakfast in Palo Alto the morning I was going to a wedding,” she says. “He spent two hours convincing me how important it was, and I had a long Catholic wedding to sit through, so I daydreamed all during the ceremony and figured out a way to do it.”
A Bioengineering Force
After joining the MIT faculty in 1991, Griffith continued to break new ground in tissue engineering. Much of her focus was on “how you take a biomaterial and make it interact in very specific ways with cells,” she says. For instance, she found that putting growth factors directly onto the surface of polymer scaffolds, rather than introducing them in soluble form, caused cells to grow more successfully. More recently, in collaboration with an orthopedic surgeon, she has grown bone marrow cells on a scaffold and shown that adding a modified form of epidermal growth factor protects them from inflammation and helps them to proliferate. This work aims to improve outcomes for patients with severe bone injuries—such as a smashed tibia—that today are treated by harvesting bone from the hip. She also pioneered the use of 3-D printing for therapeutic applications. The technology had been applied to inorganic systems like engine parts, but Griffith and colleagues patented methods for using it in tissue engineering and drug delivery. “I was driven by the tremendous need to build objects with control over several levels of architecture,” she says. For example, what if a bone scaffold could be printed in the shape of a patient’s jaw? She also wanted to figure out how to build scaffolds for liver regeneration outside the body, which is tricky given the organ’s complex architecture.

As Griffith’s research progressed, she became convinced that MIT needed a new department devoted to biological engineering. Though many schools offer programs in biomedical engineering, she helped lead the lobbying for a program that would focus on the basic biological interactions of living systems. That faculty campaign led MIT to enroll its first graduate students in biological engineering in the late 1990s. (Douglas Lauffenburger, an engineer and cell biologist who was recruited to MIT to head the new department, would later become Griffith’s husband.) In 2005, MIT began to offer an undergraduate major in the field, the first new major at the Institute in almost 40 years. “She really, as much as anybody, got that whole program started and continues to do a terrific job,” says Langer.
Griffith earned tenure in 1998, just as the new department was getting under way, and in 2006 she won a Mac��Arthur “genius” grant. In other words, she reached a career stage that allowed her to think broadly about her work and perhaps take greater risks. Her personal struggle with endometriosis continued, and Keith Isaacson, who has been her surgeon as well as a friend, prodded her to take on the disease as part of her research. Her sister’s daughter, who suffered from terrible menstrual pain, was eventually diagnosed with endometriosis as well. Though Griffith continued to resist spotlighting her gender, others clearly viewed her as an exemplar. In 2007, she was asked to participate in a panel on women in science at Boston’s Museum of Science. “When they asked me to talk about how my research benefits women, I almost ripped their eyes out,” says Griffith, who recalls thinking, “I work on liver and bone. Everyone has them.” Yet she was also aware that as a woman and a patient with a chronic disease, she had a perspective many of her colleagues did not. During the event, a grad student asked where her research would head in the next few years, and something “just welled up,” she recalls. “I said, ‘I’m going to start a women’s health center and study endometriosis and bring all these great guys and women from MIT to study it,’” she says. “I just blurted it out, and then I was on the hook to do it.”
Targeting Endometriosis
In the corner of Griffith’s lab today sits a red picnic cooler, which her students use to transport samples of endometrial tissue and fluid back from Newton-Wellesley Hospital. That cooler has been put to good use since 2009, when Griffith helped launch the MIT Center for Gynepathology. Elsewhere in her airy Building 16 lab hum large freezers stocked with these samples, gathered from dozens of women with endometriosis. Now, in one major project, she and her colleagues are analyzing the peritoneal fluid of women with the disease, amassing a treasure trove of data. In February, the team found that levels of a group of 13 immune molecules related to inflammation seemed to rise in concert in some women with endometriosis. The women with this immune signature did not necessarily have the most extensive lesions; nor was their disease focused in a particular area, like the ovaries, fallopian tubes, kidneys, or bowel. Yet their disease tended to be especially painful, and they were more likely than other women to have fertility problems. These results, published in Science Translational Medicine in February, were based on women from the Boston area. In May, Griffith and her team presented new work confirming the findings in a composite group that included women in Brazil.

The new results, which they shared at the World Congress on Endometriosis in Brazil, suggest a way to “stratify patients based on molecular markers,” says Yale’s Hugh Taylor. Right now, “we don’t know why some patients have pain and some don’t, some have infertility and some don’t.” In disorders like breast cancer, physicians rely on molecular markers—as well as surgical findings—to predict how the disease might progress and what the most effective therapies will be. Griffith argues that the same should eventually be true of endometriosis. (In a cruel twist, she herself was also diagnosed with breast cancer, though she now has no evidence of the disease.)
The markers that Griffith’s group has identified for endometriosis “still need more vetting” to establish their clinical utility, says Taylor. But ultimately, they might serve as the foundation for a test that could be performed in a gynecologist’s office, rather than an operating room, and could lead to earlier diagnosis and treatment for some women. In theory, earlier treatment might stop the progression of the disease before it leads to internal damage or potentially irreversible scarring, which may cause infertility. Many researchers are working on less invasive tests for endometriosis, says Taylor, but Griffith’s findings have the “greatest potential.”
At the same time, Griffith continues to analyze the complex interplay of other tissues and organ systems. In 2012, the Defense Advanced Research Projects Agency (DARPA) and the National Institutes of Health awarded a group at MIT and another at Harvard more than $30 million to develop a microscale model of interactions among 10 different organ systems, including the liver, gut, and lungs. This “physiome on a chip” project, for which Griffith serves as both a principal investigator and the coördinator of the more than 50 researchers involved, will feature tiny collections of cells organized into three-dimensional structures to model each organ (though the models will not necessarily mimic the structures of the real organs). With small tubes to connect the different structures and pumps to circulate a culture medium, the platform will offer a novel way to eavesdrop on the cross talk between physiological systems. It will allow researchers to study, for instance, how a compound that enters the gut, travels to the liver, and is metabolized there may affect other organs.
Pharmaceutical companies currently test potential drugs for toxicity in human cells before moving on to animal models. This platform offers an intermediate step that might allow them to detect side effects before undertaking animal tests, says Griffith. It might also offer insights that animal models do not, since the cells involved are human. The work represents an effort to build more sophisticated tissue culture models and test hypotheses about how things interact when metabolites, hormones, growth factors, and immune molecules are moving from one organ to the next. When you have higher levels of complexity, she says, “you can always get unintended consequences.”
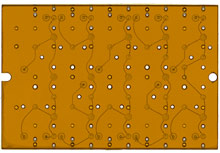
In previous work, Griffith designed a three-dimensional “liver chip�� that can also be used to assess drug toxicity and is now produced commercially by a British company called CN Bio Innovations. A modified version of that chip serves as the basis for the liver component of the DARPA project. Griffith is also collaborating with MIT biological engineer Eric Alm to apply a rich mix of bacteria to the gut component of the platform. That will let researchers study how drugs’ potency can be increased or decreased if they are partially metabolized or altered by gut bacteria before moving on to the liver.
The human physiome on a chip will include a model endometrium, which will provide further insights into how the uterus normally functions and what goes wrong in endometriosis, says Griffith. In the body, cells are “never just sitting in their own media” for extended periods of time, says Teresa Woodruff, director of the Women’s Health Research Institute at Northwestern University. This project creates a more “vibrant and more kinetic system,” she adds, “which is more like the way cells experience the circulatory system.” Griffith and Woodruff are also collaborating to create a chip to model more of the female reproductive system, including the ovaries and vagina. Eventually, they hope to replicate the menstrual cycle on this platform, yielding further insights into disease.
The human physiome on a chip integrates Griffith’s long-standing work on the liver with her more recent drive to conquer endometriosis and other disorders that plague women. She is adamant that her work not be seen as creating a pink ghetto: “I don’t want to make endometriosis a women’s issue,” she says. “I want to make it an MIT issue.” But her ultimate message is also one of empowerment: “I want to show young women that if you have a problem, you can go be an engineer and solve it. In this field, you can gain control over problems that in the outside world you have no control over.”
Keep Reading
Most Popular
Large language models can do jaw-dropping things. But nobody knows exactly why.
And that's a problem. Figuring it out is one of the biggest scientific puzzles of our time and a crucial step towards controlling more powerful future models.
How scientists traced a mysterious covid case back to six toilets
When wastewater surveillance turns into a hunt for a single infected individual, the ethics get tricky.
The problem with plug-in hybrids? Their drivers.
Plug-in hybrids are often sold as a transition to EVs, but new data from Europe shows we’re still underestimating the emissions they produce.
Stay connected
Get the latest updates from
MIT Technology Review
Discover special offers, top stories, upcoming events, and more.