Shining Light on Madness

At Novartis’s research lab in Cambridge, Massachusetts, a large incubator-like piece of equipment is helping give birth to a new era of psychiatric drug discovery. Inside it, bathed in soft light, lab plates hold living human stem cells; robotic arms systematically squirt nurturing compounds into the plates. Thanks to a series of techniques perfected over the last few years in labs around the world, such stem cells—capable of developing into specialized cell types—can now be created from skin cells. When stem cells derived from people with, say, autism or schizophrenia are grown inside the incubator, Novartis researchers can nudge them to develop into functioning brain cells by precisely varying the chemicals in the cell cultures.
They’re not exactly creating schizophrenic or autistic neurons, because the cells aren’t working within the circuitry of the brain, but for drug-discovery purposes it’s the next best thing. For the first time, researchers have a way to directly examine in molecular detail what’s going wrong in the brain cells of patients with these illnesses. And, critically for the pharmaceutical company, there is now a reliable method of screening for drugs that might help. Do the neurons look different from normal ones? Is there a flaw in the way they form connections? Could drugs possibly correct the abnormalities? The answer to each of these questions is a very preliminary yes.
The technique is so promising that Novartis has resumed trying to discover new psychiatric drugs after essentially abandoning the quest. What’s more, it’s been introduced at a time when knowledge about the genetics behind brain disorders is expanding rapidly and other new tools, including optogenetics and more precise genome editing (see “Neuroscience’s New Toolbox”), are enabling neuroscientists to probe the brain directly. All these developments offer renewed hope that science could finally deliver more effective treatments for the millions of people beset by devastating brain disorders.
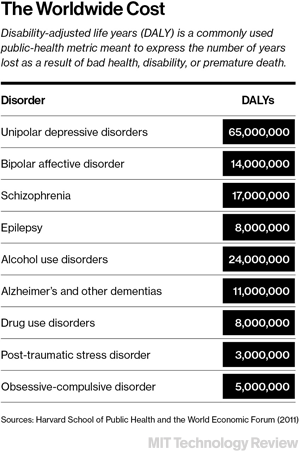
A revival in psychiatric drug development is badly needed: there hasn’t been a breakthrough medicine for any of the common mental illnesses, including schizophrenia, bipolar disorder, or severe depression, in roughly 50 years. From the late 1940s through the 1960s, a series of serendipitous discoveries, beginning with the finding that lithium could help bipolar patients, transformed the treatment of the mentally ill. It became possible to quiet the hallucinations and delusions of schizophrenia and offer a drug to the severely depressed. The sudden availability of pharmacological relief transformed psychiatry and played a role in closing down many of the mammoth mental hospitals of the era. But then, almost as suddenly as it had started, the revolution stalled.
Many of the drugs discovered in the 1950s and 1960s are still the most effective treatments available for schizophrenia, anxiety disorders, and depression. But while these medications have improved the lives of some patients, they are ineffective for others, and they are woefully inadequate in treating many of the worst symptoms. What’s more, the drugs can have severe side effects.
Take schizophrenia, for example. Existing antipsychotic drugs can make the hallucinations and delusions disappear, but they don’t improve the so-called negative symptoms—the disruption of emotions such as pleasure, which can leave people uninterested in communicating or even in living. Existing drugs also have no effect on the way schizophrenia can impair concentration, decision-making, and working memory (critical in such tasks as language comprehension). These debilitating cognitive problems make it impossible for people to work and difficult for them even to make the seemingly simple logical choices involved in everyday life. Insidiously, such symptoms can strike high-performing individuals, often in their late teens. “People don’t understand,” says Guoping Feng, a professor of neuroscience at MIT who studies the neural basis of psychiatric disorders. “They ask, once a patient is given antipsychotic medicine, ‘Why can’t you go to work?’ But [those with schizophrenia] can’t work because they don’t have cognitive functions, they don’t have normal executive functions. And there are no drugs for this.” On top of that are the side effects of antipsychotic medicines, which can include Parkinson’s-like movement disorders, dramatic weight gain, or a potentially deadly loss of white blood cells. In short, the illness destroys many patients’ lives.
Finally, many people with brain disorders are simply not helped at all by available drugs. Antidepressants work well for some people but do nothing for many others, and there are no effective drug treatments for the social disabilities or repetitive behaviors caused by autism.
“We were led down a path that said depression is about being a quart low in serotonin, and schizophrenia means you have a bit too much dopamine on board. But that just isn’t how the brain works. The brain isn’t a bowl of soup.”
Overall, neuropsychiatric illness is a leading cause of disability. According to the National Institute of Mental Health (NIMH) in Rockville, Maryland, 26 percent of American adults suffer from a “diagnosable mental disorder” in any given year. Severe depression, the most common of these disorders, is the leading cause of disability in the U.S. for individuals between 15 and 44. Around 1 percent of the American population suffers from schizophrenia; one in 68 American children is diagnosed with an autism spectrum disorder.
Though the need for better treatments is unquestionable, drug companies had until very recently simply run out of good ideas. The drugs developed in the 1950s and 1960s were discovered by accident, and no one knew how or why they worked. In the subsequent decades, drug researchers reverse-engineered the medications to identify the brain molecules that the drugs acted on, such as dopamine and serotonin. In retrospect, however, scientists now realize that while tweaking the levels of these chemicals addressed some symptoms of psychiatric disorders, it was a crude strategy that ignored the biological mechanisms underlying the illnesses.
“By studying the drugs, we were led down a path that said depression is about being a quart low in serotonin, and schizophrenia means you have a bit too much dopamine on board,” says Thomas Insel, the director of NIMH. “But that just isn’t how the brain works. The brain isn’t a bowl of soup; it’s really a complex network of networks.” Psychiatric illnesses such as schizophrenia, severe depression, and bipolar disorder, Insel says, “are fundamentally disorders of the brain circuits.” It is only in the last few years, he adds, that technologies such as optogenetics have allowed neuroscientists to shift their thinking from “soups to sparks”—electrical impulses—and begin exploring the circuits involved in brain disorders. Still, he says, the search for treatments is slowed by “our profound ignorance of the brain.”

Another obvious impediment to finding better drugs is that there haven’t been reliable ways to screen them. Because researchers have had limited ability to measure how potential psychiatric drugs affect the biology of lab animals, they’ve “concocted” tests based on the way existing drugs affect animal behavior, says Steven Hyman, director of the Stanley Center for Psychiatric Research at the Broad Institute of Harvard and MIT. One conventional assay for antidepressants, for example, is called the “forced swim test.” When rats given the commonly used drug imipramine, which was invented in the 1950s and is still considered one of the most effective medicines for depression, are dropped in a bucket of cold water, they swim longer before giving up. The animals’ propensity to stop struggling has been rationalized as a measure of “behavioral despair,” Hyman says, but there’s actually no proof that the behavior in the test reflects human depression. Though the swim test has been used for 50 years to test antidepressants and is still widely used, all it probably does is select for drugs that mimic the effects of imipramine in allowing a rodent to swim longer, he says. That has led to a series of “me-too drugs.”
The discovery of new psychiatric drugs is “dangerously stalled,” Hyman says: in terms of efficacy, antidepressants “maxed out” in the 1950s and antipsychotics in the 1960s. Though a number of new psychiatric drugs have been marketed in recent decades, says Richard A. Friedman, a professor of clinical psychiatry and director of the psychopharmacology clinic at Weill Cornell Medical College in New York, they are simply molecular knockoffs of older ones. Some of the newer medications are somewhat safer, he says, but essentially, drug companies are just “tweaking the same molecules.” Given the lack of ideas for effective new compounds and the high rate of failure for psychiatric drugs in expensive clinical trials—only about 8 percent succeed, compared with 15 percent for drugs overall—it’s no wonder that, in Friedman’s words, pharmaceutical companies have “gotten cold feet.”
Indeed, in 2011 Novartis announced it was shutting down its center for basic neuroscience research in Basel, Switzerland. The company wasn’t alone in stepping away from the search for psychiatric drugs. Over the last five years, other drug makers, including GlaxoSmithKline and AstraZeneca, have all scaled back efforts and decreased investments in neuroscience and related drug discovery. But Novartis’s move was particularly noteworthy because the discovery of psychiatric drugs plays such a big part in its history. In the 1960s, Basel-based Sandoz, which merged with Ciba-Geigy in 1996 to form Novartis, was instrumental in developing clozapine, still one of the most effective treatments for severe schizophrenia. And Ciba, another Swiss parent of Novartis, had introduced imipramine in the late 1950s.

Now, in Cambridge, Novartis is back on the search. Ricardo Dolmetsch, the company’s global head of neuroscience, is the one tasked with translating what he calls the recent revolutions in genetics and genomic tools into safe and effective medicines. A former professor of neuroscience at Stanford, Dolmetsch joined the company last August and immediately began hiring. Less than a year later, his colleagues are conducting experiments among stacks of plastic moving crates, even as they continue to set up the lab. Though there is a sense of excitement on his team, Dolmetsch’s words are measured: “We now have the tools to give it another shot.”
Jigsaw Puzzle
Faulty genes have a significant role in causing brain disorders. If you have an identical twin with schizophrenia, the likelihood that you will also have the disorder is between 40 and 65 percent; if a sibling has the illness, you have about a 10 percent chance. Statistics are similar for autism and bipolar disorder. While genes are somewhat less of a factor in depression than in the other disorders, they play a critical role there too. But, says Broad’s Hyman, it’s only in recent years that researchers have realized how complex the genetics are. When he was the director of NIMH in the 1990s, Hyman says, it was already clear to him and others that there was no single schizophrenia or autism gene. “But I thought that there were at most 20 or, at the extreme, 100 genes,” he adds. “We were way off.”
So far, researchers have identified hundreds of genetic variants associated with increased risk for schizophrenia, and Hyman guesses the number could go as high as a thousand. Some of the mutations appear to be common, while some rare variants seem to cause the same symptoms as those experienced by individuals with a completely different set of rare mutations. Moreover, different variants seem to confer different degrees of risk, and recent studies have shown that multiple disorders, including schizophrenia and autism, share a number of culpable genes. Hyman calls it an immensely complicated jigsaw puzzle.

Whether you think the extreme genetic complexity of brain disorders portends well for drug discovery depends on whether you’re a pessimist or an optimist, says Pamela Sklar, who directs the department of psychiatric genomics at the Icahn School of Medicine at Mount Sinai in New York. The conventional approach to discovering drugs for diseases with a strong genetic component is to identify the gene causing or playing a prominent role in the illness, and then test compounds against the protein it codes for. That approach is not likely to work for most psychiatric illnesses, given that they are caused by combinations of so many genetic variants. But Sklar obviously leans toward optimism. She suggests that the numerous variants provide more chances to home in on key pathways involved in the disorders, and more opportunities to come up with clever ways to fix them.
The hope is that all those genetic variants will tend to affect common sets of molecular pathways, types of cells, or specific neurocircuits. That could help scientists pinpoint what’s going wrong, and it could also give them new targets for potential treatments. Yet Sklar, who specializes in searching for the genetic causes of schizophrenia and bipolar disorder, acknowledges that despite the rapid advances in genetics over the last few years, large gaps in understanding remain. “We don’t know all the risk factors,” she says, “and with so few pieces of the puzzle, it’s still hard to know how it all hangs together.”
Add to this genetic mystery the fact that the brain has roughly 86 billion neurons and around a quadrillion synapses (the connecting points between neurons), and it becomes obvious how overwhelming it will be to understand the causes of brain disorders. It’s why the ability to take cells from a patient and turn them into neurons in a dish has researchers so excited. Now they have a way to directly examine how genetic variants have affected the neurons of a patient with an illness. You still might not know all the details of the genetics, but at least you can see the results. What’s more, new genome-editing methods make it possible to precisely alter the genes of the stem cells from which the neurons develop, adding a mutation associated with the illness to see how it affects the resulting neurons.
But how do these neurons function in an actual brain, with its immense networks of circuits and connections? How are the genetic mutations implicated in autism and schizophrenia actually affecting those circuits to alter behavior? New research is starting to investigate those questions.

Later this summer, a colony of marmoset monkeys, primates native to South America, will begin living at MIT’s McGovern Institute for Brain Research. Monkeys and people share a highly developed prefrontal cortex, the region near the front of the skull. And, says MIT’s Feng, there’s increasing evidence that many of the intractable defects of schizophrenia and the deficiencies of social communication and behavior found in autism can be traced to this area of the brain.
To begin to unravel what’s going wrong, Feng and his colleagues plan to use genome editing to breed monkeys with precise mutations associated with psychiatric disorders. Initially, the scientists will focus on a rare mutation in a gene called SHANK3. Because it is an unusual example of a single gene that causes clear, autism-like changes in behavior, it’s a simple place to start. Later generations of monkeys could have the multiple mutations found in most forms of autism and schizophrenia.
The monkeys could provide a more reliable way to test psychiatric drugs than rodents, whose brain circuitry is much less similar to ours. The idea is not to create animals with schizophrenia or autism—the complex mixture of aberrant human behaviors can’t be truly replicated even in other primates—but rather to see how the genetic mutations change the circuitry at a molecular level and how the animals’ behavior is altered as a result. “The behavior may not be identical to that in humans,” says Feng, “but at least it’s a readout. It becomes a confirmation that we can correct the circuits and that the changes lead to improvements in behavior.”
Don’t Worry
The mouse cowers in one corner of the maze. Even in the video of the experiment, its anxiety seems palpable as it presses against a wall. A thin fiber-optic thread is connected to the animal’s skull. Suddenly, after a burst of blue light through the fiber, the mouse begins scurrying about, exploring the four branches of the maze with new energy and courage.
The invention of optogenetics has revolutionized the study of neurocircuitry. But even among all the impressive studies using the technology, this mouse experiment, which Kay Tye conducted in 2011 as a postdoc at Stanford, stands out. Tye, now an assistant professor at MIT, showed she could turn anxiety on and off with a flip of a switch. Though she was targeting a part of the brain called the amygdala, which is well known to be involved in fear and anxiety, she was “surprised by just how sudden and robust the change was,” she says: “It was almost instantaneous. I was blown away. It has changed how I think about the brain.”
How can this increasing knowledge of brain circuits and connections—and their role in such feelings as anxiety—be translated into actual therapies? Can researchers find effective and safe ways to intervene?
How can this increasing knowledge of brain circuits and connections—and their role in such feelings as anxiety—be translated into actual therapies? Can researchers find effective, safe ways to intervene in these circuits and connections in patients’ brains—ways to fix what has gone wrong?
Optogenetics, at least in its existing version, doesn’t look like the way to do that. It requires genetic modification of the cells that researchers want to activate and the intrusive use of a fiber-optic thread in the brain. That’s why it’s largely limited to rodents, though a few monkey experiments have been done. Using the technology to directly fix malfunctioning brain circuits in humans is not now practical, and it may never be. But as a research technique, it could give drug researchers what they so desperately need: new molecular targets. Researchers like Tye and Feng believe that their optogenetic experiments can help identify specific types of cells in the circuits underlying certain psychiatric symptoms. Then they will have to spot distinguishing markers on those cells that allow the drug to recognize them. It’s an extremely promising approach; recent results do in fact suggest that there are ways to single out critical cells as targets for drug compounds. But the research is still in its very early days.
One alternative is to try to intervene directly in the circuits, skipping the use of drugs. A standard treatment for Parkinson’s is to implant an electrode array in a patient’s brain to calm tremors. It’s called deep brain stimulation, and researchers at Emory University are attempting to adapt the technology to treat depression, inserting an electrode into a region of the brain called Area 25. Others are using deep brain stimulation to treat obsessive-compulsive disorder with promising results.
There might also be ways to directly affect faulty circuits without resorting to surgery. Amit Etkin, an assistant professor of psychiatry at Stanford, is using a combination of functional magnetic resonance imaging (fMRI) and noninvasive magnetic stimulation to map the circuitry that goes wrong in patients. His goal is to tailor magnetic stimulation, which is already widely used to treat intractable cases of depression, to the specific problems in a patient’s neurocircuitry.
The therapy, which is administered using an electromagnetic coil placed against the scalp, uses magnetic pulses to create an electric current that can increase or decrease brain activity. The commercial version of the technique is designed to target the same small part of the prefrontal cortex in all patients, but by combining it with imaging technology, Etkin hopes to aim the stimulation more precisely to where a patient needs it. It won’t be a miracle cure. The method seems to help only some people. But driving his work, says Etkin, is his frustration at not being able to offer patients more successful options.
Etkin, who also works at a clinic at the Palo Alto VA Hospital for veterans suffering from severe anxiety and depression, uses a variety of tools to help patients, including drugs and psychotherapy as well as magnetic stimulation. The key to making all the approaches more effective, he says, is to learn more about how faulty neural circuits and connections lead to aberrant behavior. In trying to fix those problems, he says, “I try not to be chauvinistic about the [treatment] technology.” Working with patients not only motivated his research on potentially better treatments, he adds, but also teaches him about its practical limitations: “Lots of scientific studies might make sense, but there is a gap with what is doable in the real world. And sometimes the gap is significant.”
Chasing Drugs

At Novartis, Ricardo Dolmetsch is responsible for trying to close that gap between the exploding scientific understanding of brain disorders and the availability of more effective drugs. And he’s realistic about the prognosis: “I hope it will be a story of excitement. But we don’t know yet. It takes a long time.”
Dolmetsch is not your typical drug-industry manager. Less than a year ago, he was still running a lab at Stanford and helping to create a library of neurons from autism patients, to be hosted at the Allen Brain Institute in Seattle. And his Stanford website—he’s officially taking a leave of absence from the university—still reflects his quirky persona. It has links to stories that describe his “impish humor” and his attempts to commute on a pogo stick during his early days at the university.
About a decade ago, his research took a dramatic turn. He had started out at Stanford looking into basic questions about the biochemistry in brain cells, work that was impressive enough to gain him an appointment as an assistant professor. But then, in 2005, his son received a diagnosis of autism. Frustrated by the lack of treatment options, Dolmetsch rebuilt his lab around researching the disorder. Since then, he has helped pioneer methods that take skin cells from individuals with autism, reprogram those cells to become stem cells, and then induce them to develop into neurons in which abnormalities can be studied. It’s that technology, along with the revolution in genetics created in part by fast, cheap DNA sequencing, that he believes will be the linchpin of Novartis’s revived effort to identify new psychiatric drugs.
“It allows you to start with the patient,” says Dolmetsch. While oncologists have long had the ability to biopsy tumors, “you can’t just drill holes in people’s brains and take little bits out,” he notes. “But now we have the capacity to make biopsies out of stem cells.” Pushing the technology even further, the Novartis researchers are gearing up to make organoids, small chunks of brain that form in a petri dish as the neurons mature and lump together into three-dimensional structures. Not only can the researchers look for abnormalities, they can screen compounds in Novartis’s vast library of potential drugs to see how the chemicals affect the neurons.
It’s far from clear how or even whether the growing knowledge of what goes wrong in the brains of those with psychiatric disorders can lead to medicines. But at least now, researchers have the tools they need.
Designing drugs to precisely target circuits in the brain remains a more distant opportunity. “We now have some idea of the kinds of cells and regions of the brain that we need to inhibit or activate to make someone happier or less anxious. We no longer need to treat the brain as one mush of signaling molecules,” Dolmetsch acknowledges. But that still leaves the daunting challenge of developing a drug that selectively activates or inactivates certain types of cells in certain circuits. “How do you do that?” he asks. “It’s not been done before.” He adds: “It’s not where drug discovery is now, though it’s where it is going.”
Dolmetsch joined the pharmaceutical industry because he realized that the science and technology had advanced far enough to create opportunities for developing new psychiatric drugs. He also realized after years of academic research that commercializing a new drug requires the resources, money, and patient populations available to a company like Novartis. It was, he says, “time to walk the walk” after years of talking about potential treatments for autism and schizophrenia.
Still, the failure to find effective new drugs for brain disorders—and the stigma that has grown around the high cost of those failures—is clearly never far from the minds of those in the industry. Companies “have developed some fantastic drugs over the last 10 years or so that were safe and engaged the target,” says Dolmetsch, before delivering the punch line: the drugs were as ineffective as water. While he’s convinced that he and his team have “a better way to discover new drugs,” he also acknowledges that it will be five to eight years before they know whether their strategy based on the new genetic and stem-cell tools is working.
For those trying to discover new medicines, it’s all about finding novel molecular targets that a chemical compound can safely affect in a way that will address the symptoms of a disease. That’s a daunting challenge. And it’s still far from clear how or even whether the growing knowledge of what goes wrong in the brains of those with psychiatric disorders can lead directly to such medicines. But at least now, after decades of dead ends, drug researchers finally have some of the tools they need to begin methodically testing strategies for finding and acting on those targets. When at last we have a better way to chase drugs for such disorders as autism and schizophrenia, says Dolmetsch, “it would be a bit of a crime not to take advantage of it.”
Keep Reading
Most Popular
Large language models can do jaw-dropping things. But nobody knows exactly why.
And that's a problem. Figuring it out is one of the biggest scientific puzzles of our time and a crucial step towards controlling more powerful future models.
The problem with plug-in hybrids? Their drivers.
Plug-in hybrids are often sold as a transition to EVs, but new data from Europe shows we’re still underestimating the emissions they produce.
Google DeepMind’s new generative model makes Super Mario–like games from scratch
Genie learns how to control games by watching hours and hours of video. It could help train next-gen robots too.
How scientists traced a mysterious covid case back to six toilets
When wastewater surveillance turns into a hunt for a single infected individual, the ethics get tricky.
Stay connected
Get the latest updates from
MIT Technology Review
Discover special offers, top stories, upcoming events, and more.