Why We Do What We Do
Much of our health and happiness is rooted in our own behaviors: whether we exercise and eat right, whether we make choices as optimists or as pessimists, whether we stay motivated to reach our goals or stick to the status quo. But even the best conscious intentions don’t always translate into the behavior we want. Increasingly, neuroscientists are starting to see—and even manipulate—the brain activity responsible for turning thoughts and feelings into actions. This work raises the tantalizing possibility that we could find more precise therapies for conditions like mood disorders and anxiety, compulsive behaviors, and addiction.

Institute Professor Ann Graybiel, PhD ’71, is at the forefront of this research, having devoted much of a career now in its fifth decade to understanding a seemingly humble set of brain structures called the basal ganglia. Once known only for helping to control movement, this region deep within the brain is now believed to play fundamental roles in how we learn, process emotions, make decisions, and adopt habits. And that shift in thinking is due in no small part to the research done in Graybiel’s lab.
Her work has already yielded insights into patterns of brain activity associated with movement disorders and psychiatric diseases. Recent studies using light to control individual brain cells, for instance, show how shutting off some of this activity can control habit formation or pessimistic decision-making. Although this technique, known as optogenetics, is still just a research tool, she is convinced that such technological advances hold therapeutic promise—and that learning about these deep patterns in the brain will also be important for everyone who wonders: What makes me do what I do?
“This is truly important for everyday life, and it’s truly important at a social and societal level,” says Graybiel, an investigator at MIT’s McGovern Institute for Brain Research and a member of the Department of Brain and Cognitive Sciences. “We human beings need to understand this stuff about ourselves.”
A new brain architecture
The study of the brain has long been stymied by the meagerness of the techniques available to address grand questions about the nature of thoughts, memories, and decisions. Today, the field is enjoying a renaissance driven by technologies that offer new ways to study patterns of communication between cells and regions of the brain. It’s yielding some stunning breakthroughs in the ability to manipulate complex behaviors. Graybiel’s early fundamental insights into the basic architecture of the brain were among those that laid the groundwork for these breakthroughs.
Graybiel was born in Boston in 1942 but raised in Pensacola, Florida, where ninth-grade girls of her era studied sewing but not science. After boarding school in Washington, D.C., she studied chemistry and biology at Harvard and headed to MIT, whose psychology department, led by Hans-Lukas Teuber, was a magnet for pioneers in the field of neuroscience.
By then—the late 1960s—scientists were performing landmark experiments that began to map out how the systems governing vision and touch were organized in the brain. “There were so few techniques to study the brain,” Graybiel says, “but it was a very exciting time.” Scientists were beginning to measure electrical signals in animals’ brain cells to map the organization of the neocortex, the folded outer rind of the brain that is the seat of higher functions like perception and conscious thought.
When she joined the MIT faculty two years after receiving her PhD in 1971, Graybiel specialized in studying the brain’s anatomy. She was well equipped for that task by her training under the great neuroanatomist Walle Nauta, who developed special stains that could be applied to human or animal brain tissue to trace how brain fibers were connected. It was “aesthetically pleasing work,” she says. “The brain just happens to be very beautiful. It doesn’t need to be, but it’s just extraordinarily beautiful.”
Most stains were designed to show the physical properties of cells, but Graybiel developed novel stains that revealed the locations of chemicals that cells use to communicate, creating a map of chemical activity.
This strategy turned out to be useful in exposing the organization of the brain. In some areas that organization had been easy to see: the neocortex was fascinating, for instance, because it held a layer cake of precisely ordered neurons that hinted at the complexity of its functions. But other regions seemed chaotic at first glance. “It’s just fabulous,” Graybiel says of the neocortex. “Then you look underneath that, and there’s this huge ball of neurons that aren’t elegantly organized apparently; it’s very humble-looking, but it’s huge.” This giant glop of brain tissue was the striatum, part of the basal ganglia, which was seen as a more primitive area of the brain.
When she applied her chemical stains to the seemingly homogeneous mass of the striatum, however, an organizing principle suddenly came into view. The striatum’s cells were arranged into chemically distinct compartments, which Graybiel dubbed striosomes. This insight revealed a new way to understand the brain’s anatomy: through chemistry rather than the shape or orientation of cells. Paul Glimcher, a neurobiologist at New York University who has been inspired by her work, calls Graybiel’s exploration of the striatum’s structure “the last of the heroic neuroanatomical projects” in classical brain anatomy.
Deciphering a mosaic
The striatum turned out to be much more interesting than people thought, and Graybiel has spent her career seeking to understand it and the neural circuits for which it serves as a hub. When she began her research, the striatum was known to be involved in movement disorders like Parkinson’s disease, which is caused by the death of brain cells that supply dopamine to that part of the brain. Since then, it has been linked to a fascinating array of brain functions, including motivation, reward, habit formation, and decision-making.
For Graybiel, the organization that she uncovered in the striatum is the key to understanding how it works. “If you could imagine the most beautiful mosaic … that’s the way the striatum is,” she says, “only it’s in 3-D.” The “tiles” of this mosaic are chemically distinct striosomes. Individual striosomes and their surrounding matrices of cells seem to make up separate groups of tiles or modules connected to distinct parts of the brain.
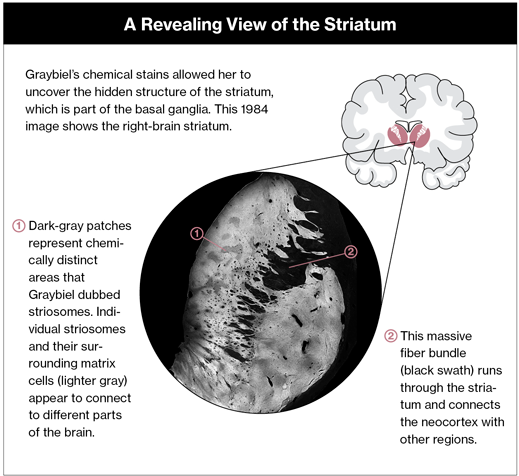
It’s clear that the striatum contains information hubs connecting areas located above it, in the neocortex, with regions lying under it, which govern emotion and mood. In recent years Graybiel’s lab has produced key findings that illuminate the communication between these regions and the role this communication plays in determining behavior. The striatum’s modular architecture, she believes, is a very different way of organizing information from the one seen in the layered cortex. She has come to see it as a learning device: it gathers information from other brain regions so that we can learn to quickly choose which behaviors to carry out, eventually acting instinctively.
Some parts of the striatum are involved in learning, planning, anticipating rewards, and making value judgments about whether something is positive or negative. Other parts allow us to form habits. These seem to involve a different kind of brain functioning, in which we’re not actively anticipating and judging but automatically playing out a previously learned script.
Studies in Graybiel’s lab explore both of these processes and how they interact. One, led by research scientist Ken-ichi Amemori, investigated an area of the cortex that appears to communicate with the striatum and is associated with anxiety and depression. When animals faced a task that produced combinations of negative and positive results (an annoying puff of air and a food reward), stimulating that area made them more likely to avoid the negative outcome even if it meant missing out on the reward, reflecting a tendency to make pessimistic decisions. The researchers were able to block this tendency with an antianxiety drug. Amemori’s research suggests that an independent brain circuit governs this pessimistic decision-making, and he is now investigating a different circuit that may control decisions made on the assumption of a positive outcome, challenging the conventional view that assessing costs and benefits is a single unified process.
Graybiel thinks such findings could identify brain circuits that handle highly emotional decisions hinging on judgments about whether an outcome will be good or bad. “A lot of our emotional lives are very rich, but we have to make decisions that are sometimes ‘feel it in your gut’ decisions,” she says. In other words, the complex emotions and perceptions must coalesce into a simple yes or no. She wants to understand what motivates this decision-making, and why this emotional evaluation goes awry in certain psychiatric disorders.
Another study revealed the role dopamine plays in anticipating how far we are from distant rewards. By examining rats running a maze, grad student Mark Howe, PhD ’13, found that the amount of dopamine released in the striatum slowly rose as the rats approached their goal. These dopamine “ramps” were steeper when a larger reward was expected or when the goal was farther away; they may help maintain motivation to reach a goal.
From decisions to habits
Goals, motivations, and values help us choose behaviors, but habitual behaviors are fundamentally different. The first few times a person smokes a cigarette, Graybiel says, “everything is very volitional—they’re trying it out.” But if it becomes a habit, every step of smoking—sliding the cigarette from its package, lighting it, and inhaling—becomes ingrained. “Even if you really don’t want to, it’s hard to not do,” she says.
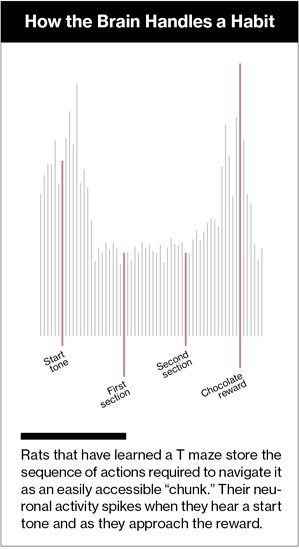
The same phenomenon occurs in lab animals. Her team trains rats to run a simple T-shaped maze, rewarding them when they turn right or left in response to one of two audible tones. Eventually the activity becomes habitual: the rats keep turning right or left even if the reward is taken away or made unpleasant (the chocolate treat that usually greets them is laced with a bad-tasting chemical). When rats first learn the maze, neurons in the striatum fire throughout the run. But as the route becomes habitual, the pattern changes, with neuronal activity mainly spiking at the beginning and end of the task. This suggests that habits are linked sequences of behaviors, stored as easily accessed “chunks” that can be performed without thinking through each step.
Graybiel’s lab has also been at the forefront of research that uses optogenetics to study complex behaviors. With this technique, scientists can use light exposure to precisely shut off or stimulate parts of the brain in freely moving animals, opening up new ways of studying behavior. As a postdoc, Kyle Smith, who is now an assistant professor at Dartmouth College, led research showing that he could break a maze-running habit in rats if he switched off a part of the brain called the infralimbic cortex—one of the areas of the cortex that talk to the striatum—as the rats ran. The study suggests that even the most automatic tasks are under the immediate control of decision-making centers in the brain. A more recent study by Smith and Graybiel found that shutting off the infralimbic cortex can prevent the rats from adopting a habit in the first place.
A study that Graybiel and colleagues published last year in Science looks at the negative side of habit formation: compulsive behaviors. A team led by postdoc Eric Burguière studied an animal model of obsessive-compulsive disorder using mice engineered to lack a gene involved in cell communication in the striatum. By repeatedly following a tone with a water drop on the face, which makes the mice reflexively groom themselves, they conditioned the mice to groom every time they heard the tone. Both the engineered mice and normal mice adopted the habit of grooming after the tone, but the normal mice learned to wait until the water drop actually hit, while the “compulsive” mice groomed as soon as they heard it. When the researchers used optogenetics to silence light-sensitive cells in the cortex that communicate with the striatum, the OCD mice stopped grooming compulsively, sparing their efforts until the water drop touched them.
Now, Graybiel says, “we want to know what it is that initiates this process of becoming repetitive.” She believes it will be possible to develop new ways to treat disorders in these brain systems. For instance, devices currently used for deep brain stimulation could be refined and combined with devices that release drugs, or an approach like optogenetics could be used to safely modulate specific brain circuits.
An ambitious agenda
Graybiel has received science’s top honors—including the 2001 National Medal of Science, the nation’s highest science and technology award—and shared the 2012 Kavli Prize in Neuroscience. In 2002, she was awarded MIT’s James R. Killian Faculty Achievement Award, which recognizes extraordinary professional accomplishment by faculty members. She’s also been recognized for her Parkinson’s disease research; among other distinctions, she was named the Harold S. Diamond Professor by the National Parkinson Foundation in 2006.
Decades after her seminal discoveries about the brain’s anatomy, she now finds herself at the leading edge of research linking brain activity to behavior. She’s sought out collaborators like Karl Deisseroth, a Stanford University bioengineer who pioneered optogenetics, in order to master new tools. Deisseroth says her studies using optogenetics to study behavior “are landmark and help to define the field.”
Most labs specialize in a limited set of techniques—some use molecular biology and genetics, some use imaging, some analyze brain waves, others record electrical signals in the brain. Graybiel and her colleagues have done all of these as they pursue an exceptionally diverse array of research projects. “She’s always reinventing herself,” says Glimcher of NYU. Her foray into animal physiology, which links brain activity to specific behaviors, is extraordinarily ambitious for someone in midcareer, he says; it’s “a bitterly hard discipline” that requires housing animals and training them to perform complex tasks, engineering highly sensitive electrical recording devices, and analyzing piles of data. But Graybiel, he adds with a laugh, “is just not a normal person.”
Graybiel’s ambition is a driving force in her large and active laboratory, but she’s also known for her down-to-earth warmth. Glimcher recalls the first time he was listed as an equal with Graybiel and several other heavyweights on a neuroscience conference program, though he was younger and felt a bit like a “warm-up band.” The other speakers, he says, were reserved, but she put her hand on his arm and said, “I just love your papers. Let’s sit together at lunch and talk about neuroscience.”
While her earlier work studying the beauty and logic of the brain’s anatomy was satisfying, what most excites her is the prospect of actually helping people. (That kind of work seems to run in her family: her father was both a cardiologist and a research scientist, and her brother is also a physician.) “In the end, we’re very, very hopeful that these sorts of findings are going to have a pretty direct helpful influence in the clinic,” she says. A collaboration between her lab and researchers in New Zealand and Japan, for instance, found in a postmortem study that the brains of people with Huntington’s disease who suffered from mood disorders have unusually severe degeneration in their striosomes, suggesting a direct relationship between these structures and mood regulation. And untangling the different steps of habit formation could lead to new ways to treat OCD or the compulsive behaviors that sometimes accompany autism.
Though she has more than her share of laurels to rest on, Graybiel is more inclined to look ahead. “Part of my time I’m thinking about what we’re doing now, but part of my time I’m thinking, ‘Oh, we could do this,’” she says. “I feel like we’re just getting going.”
Keep Reading
Most Popular
Large language models can do jaw-dropping things. But nobody knows exactly why.
And that's a problem. Figuring it out is one of the biggest scientific puzzles of our time and a crucial step towards controlling more powerful future models.
The problem with plug-in hybrids? Their drivers.
Plug-in hybrids are often sold as a transition to EVs, but new data from Europe shows we’re still underestimating the emissions they produce.
Google DeepMind’s new generative model makes Super Mario–like games from scratch
Genie learns how to control games by watching hours and hours of video. It could help train next-gen robots too.
How scientists traced a mysterious covid case back to six toilets
When wastewater surveillance turns into a hunt for a single infected individual, the ethics get tricky.
Stay connected
Get the latest updates from
MIT Technology Review
Discover special offers, top stories, upcoming events, and more.