Adventures on the Intellectual Playground
One of the most thrilling moments in Angela Belcher’s professional life came during a routine visit to the lab in the winter of 2009. Two of her graduate students in MIT’s Department of Materials Science and Engineering were trying to harness biological tools to make materials for a battery electrode. They showed her a petri dish holding a virus they had engineered to bind to materials that it normally wouldn’t have any affinity for—iron phosphate and carbon nanotubes. The virus had neatly assembled the two materials into tiny wires, which would turn out to perform as well as the electrodes in commercial lithium-ion batteries.

To the students, it was a promising result. To her, though, it was something much bigger—the realization of an audacious idea that she had once been discouraged from even pursuing. “When I started out, my dream really was to use genetics, or control of DNA, to make nonbiological devices better than can be made in other ways. It was a longer-term, pie-in-the sky idea,” says Belcher, who says she still gets chills recounting the story. Now her students had actually accomplished that goal: by tinkering with the genes of a virus, they were able to produce a high-performance electrode material. “We had got there faster than I was expecting,” she says.
That lab work on virus-grown batteries led to a paper in Science in 2009 and heaps of media attention. Batteries, however, are just one possible product of the novel toolkit Belcher has pioneered. She engineers viruses, and in some cases yeasts, to act as biological factories that produce inorganic materials with shapes and structures that would be difficult to make otherwise. Pencil-shaped viruses that infect bacteria act as a template or scaffold on which nanoparticles collect. The viruses are capable of assembling highly ordered nanoscale crystals or wires that can be useful in a variety of applications.
Through numerous collaborations, Belcher has applied her tools to some of society’s biggest problems in energy, the environment, and medicine. She has improved solar-cell performance and developed catalysts that can split hydrogen from water for fuel and convert natural gas into industrial chemicals. A company she founded came up with new ways to manufacture materials for touch-screen displays, and her lab has engineered yeast to turn waste carbon dioxide from power plants into floor tiles. A professor of energy in both the materials science and biological engineering departments, she joined MIT’s David H. Koch Institute for Integrative Cancer Research in 2010 and began working to devise diagnostic tools and treatments for cancer as well. More recently, she’s begun developing materials that could purify and separate water from organic waste or oil, and she has started working on lithium-air batteries for electric vehicles as well as energy storage devices called supercapacitors.
A Classic Eureka Moment
Remarkably, she’s accomplished all this by the age of 46—and it all ultimately goes back to the abalone sea snail.
Belcher, who created her own curriculum combining multiple areas of science as an undergrad at the University of California, Santa Barbara, became fascinated by the abalone and chose it as her subject when she pursued a PhD in chemistry at the same school. She had three PhD advisors—a chemist, a molecular biologist, and a physicist—who held weekly meetings to help each other bridge the gaps between their fields. “That’s the way I learned science,” she says. “To me, it’s a completely normal way to approach the world.”
Belcher focused on the way the animal makes its shell. Abalones produce proteins that combine with ions of calcium and carbonate from seawater to form rows of tiny inorganic crystals—one type for an outer shell and one for an extremely strong inner shell. Thinking about this process while she looked out at the ocean from her office one day, Belcher had a classic eureka moment. As her gaze shifted from the window to the periodic table of elements on her wall—placed high so as not to block the view—she wondered whether the shell proteins she had isolated in her PhD work could create useful materials by combining with other elements.
“I thought, wouldn’t it be interesting in semiconductors if you had a protein that would grow one crystal structure versus another and you could do it with genetic control,” Belcher says. “That’s when I set out to think about how to develop that genetic code for semiconductors.”
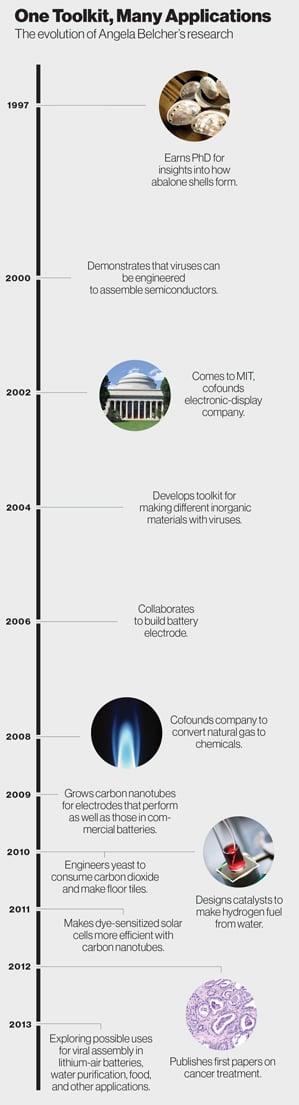
This core nature-inspired insight—that one could use genetics to produce inorganic materials—has led to dozens of scientific publications and a dizzying variety of research projects. Among her accolades are a MacArthur “genius” grant in 2004, a recognition award from the U.S. Army, and this year’s $500,000 Lemelson-MIT Prize. When Barack Obama came to MIT in 2009 and delivered a speech on energy, Belcher briefed the president on her battery and solar research. (She also gave him a small card featuring the periodic table, telling him it might come in handy “if you’re ever in a bind and need to calculate a molecular weight.” Obama’s comeback: “Thank you. I’ll look at it periodically.”)
Scientists respect Belcher for her bold, big-picture ideas and her ability to make real progress in turning them into practical applications. In addition to her academic work, she has cofounded two companies—Cambrios, which makes a material based on silver nanowires that detects finger movements on touch screens, and Siluria Technologies, which has developed a catalytic process to convert natural gas into ethylene that can be used to make materials normally derived from petroleum. “She’s very visionary and very multidisciplinary,” says Seung-Wuk Lee, who worked with Belcher as a grad student and is now a faculty scientist at the Lawrence Berkeley National Laboratory and an associate professor of bioengineering at the University of California, Berkeley. “She’s also kind of practical. She can demonstrate her ideas now, not 10 years from now.”
An Unconventional Approach
Belcher, a seventh-generation Texan, got her first job as a professor of chemistry and biochemistry at the University of Texas, Austin, in 1999. It turned out to be a good time for research in materials science and bioengineering: nanotechnology was a promising emerging field, and life scientists had access to new tools for genetic engineering. She began to work with pencil-shaped bacteriophages, or phages—naturally occurring viruses that infect bacteria. Each virus’s genes contain instructions to produce a protein that coats its surface. Researchers can target a specific kind of molecule by using a phage whose particular surface protein latches onto it—a technique that had typically been used for drug discovery.
The virus proteins don’t bind to inorganic materials in nature, but Belcher wanted to see if they could be engineered to do so. Only a few months after becoming a professor, she wrote her first grant proposal to test the concept she had first hatched in her office in Santa Barbara: that it would be possible to use a virus to bind to a semiconductor and assemble a semiconducting material as the virus invades bacteria. She got only two responses from reviewers. One said it could be interesting but Belcher didn’t have the scientific background to do it; the other simply wrote: “SHE’S INSANE.”
Though upset, Belcher carried on because her experience with the abalone, whose proteins do bind to inorganic materials, gave her confidence that it could work. She spent a few hundred dollars to buy a vial containing a collection of phages engineered with random DNA inserts that code for as many as one billion proteins. Using data from experiments, she resubmitted her grant idea and was funded by the Army. Within a year of the first rejection, she published a paper in Nature demonstrating that viruses could be engineered to produce proteins that bind to the surfaces of semiconductors, a technique that would make it possible to essentially grow materials for electronics. Each virus acts as a template, attracting semiconductor crystals that align with the proteins coating the virus. That groundbreaking proof of concept laid the foundation for all the engineering work she’s done since.
With Belcher’s method, lab researchers expose about one billion virus variants to a material such as a metal or semiconductor, identifying and isolating the proteins that attach to it best. The viruses that produce them are then placed in a solution containing bacteria. They infect the bacteria, which make millions of copies of the virus and its particular DNA sequence. Finally, they are introduced into a solution containing the material of interest. A virus that binds strongly to gold, for example, is put in a solution that contains gold ions. The gold will crystallize along the virus’s surface, conforming to the shape of the protein on its coat.
Using genetic engineering, scientists modify the virus’s DNA sequence to further control how it binds. They can coax it to assemble multiple materials, such as gold and platinum, or they can control the shape of the structures a virus will generate by choosing whether particles bind along its sides or at its tip. As the viruses replicate, they can assemble significant quantities of a desired material. Researchers can also guide the formation of materials by changing the growth conditions, such as the concentration of the virus in a solution. Depending on what’s needed, they can form either randomly oriented structures or a more orderly, repeatable architecture.
The process is water-based and allows scientists to make materials in environmentally benign conditions, at room temperature and under ordinary pressure. That’s a big advantage over traditional fabrication techniques for semiconductors or electronic circuits, which can require complex machinery, toxic materials, and high temperatures.

Disney for Scientists
When Belcher was at the University of Texas, her pioneering research in viral growth of inorganic materials attracted lots of attention within academia. Recruited by a number of places, she took a few trips to MIT, which she calls “Disneyland for scientists and engineers,” and realized it was a great fit. “Every meeting I would go to with faculty, I could see the connection to their work and the people were so excited about their research,” she says. “And it’s not like they’re keeping it close to themselves—they’re collaborating all over campus.” The periodic chart she gave President Obama—one of a stash of cards she had printed to hand out to freshmen—includes a tagline that captures how she felt upon arriving at the Institute: “Welcome to MIT. Now you are in your element.”
One of the first partnerships Belcher formed at MIT was with chemical engineering professor Paula Hammond ’84, PhD ’93. After talking about their research, they decided to work on a project to make sensors that detect biological agents. “She wanted to understand what kinds of science were going on around her and what kinds of developments she could contribute to,” Hammond says. “She was really building her collaborative community.”
Meanwhile, Belcher was improving on the basic science of her virus toolkit and expanding her palette of materials. In experiments, her lab was working through the periodic table and “seeing what we were good at making,” she says. Success with metals and metal oxides led to a collaboration on battery research with Hammond and Yet-Ming Chiang ’80, ScD ’85, a materials scientist who cofounded the battery company A123 Systems. In 2006, the three coauthored a paper in Science describing a viral growth method for making cobalt oxide nanowires, an anode material for a rechargeable lithium-ion battery, on flexible polymer films.
While that was impressive, they had created only half a battery, which needs both an anode and a cathode. Belcher then assembled a “dream team” that included professors Gerbrand Ceder and Michael Stano. They engineered a virus to grow iron phosphate along its surface, forming nanowires to serve as a cathode material.
The group next went a step further, in pursuit of a battery that could be used for cars. Building a cathode for a fast-discharging battery is more difficult than building an anode because such electrodes need to be highly conducting, yet the safe and low-cost materials Belcher’s team was exploring for cathodes are more insulating and don’t conduct well. To address this, Belcher’s group engineered a gene that compelled the virus to latch onto carbon nanotubes. While the iron phosphate assembles along the long sides of the virus’s pencil-like shape, the nanotubes attach to its tip, creating a network of electrical contacts that aid the flow of electrons and improve the battery’s power.
That was the work that led to the groundbreaking Science paper of 2009, in which the researchers described how they built a prototype that matched the power and energy capacity of the best batteries at that time. Because the electrodes could assemble on polymer films, which act as the electrolyte, the technology could lead to thin, flexible batteries or ones that take the shape of their container. Susan Hockfield, then MIT’s president, demonstrated the prototype, which lights a small LED bulb, at a Washington press conference with President Obama on the importance of federal funds for energy research.
Always intensely focused on research that could have a broad impact, Belcher also brainstormed with Hammond about how they could improve solar power. They chose dye-sensitized solar cells, in which the active layer is made of dye-covered titanium dioxide. Although such cells are inexpensive, they don’t convert light to electricity efficiently enough for use on rooftops or in utility-scale applications. But the work on getting viruses to incorporate carbon nanotubes into battery cathodes provided a pathway for making a big jump in efficiency.
In 2011, Belcher’s lab engineered a virus that assembles carbon nanotubes along its length in an orderly fashion. Then the virus grows an outer layer of titanium dioxide around the nanotubes. As in the battery work, the nanotubes create tiny wires for electrical current in the solar cell. Adding the virus-built material to the cell boosts efficiency by more than 30 percent. Her lab has also begun a project to try the same thing with solar cells made of silicon, the dominant material in the industry. “That’s how we approach it: is there any way we can apply biology in a new way?” she says.
Branching Out
As Belcher racked up accomplishments in energy, colleagues at MIT, including drug delivery pioneer Robert Langer, encouraged her to apply her expertise in nanoscience to cancer. At first, she was reluctant and a bit intimidated. More familiar with battery electrodes than cancer cells, she wasn’t sure what contribution she could make. But she ultimately threw herself into it, going through cancer tutorials with colleagues and again collaborating with other researchers, including MIT professor Sangeeta Bhatia, SM ’93, PhD ’97. “We had to learn everything from scratch,” Belcher says of her lab’s cancer work. “We could’ve never gone into that on our own.”
Now she and Bhatia are working on a surgical probe to locate very small tumors. One method their labs are developing uses a virus engineered to bind to cancer cells and carbon nanotubes. A liquid containing viruses already bound to nanotubes would be injected into the bloodstream or abdominal cavity to seek out tumors. When the viruses attached to a tumor, the nanotubes would glow under infrared light, allowing doctors to view the tumor with a specialized camera. With current methods, it’s often challenging to image ovarian tumors smaller than one centimeter in diameter without surgery. But this system has isolated tumors one millimeter in diameter in tests with animals. Still experimental, the technique could be most useful for pinpointing tumors that are difficult to find and for which early detection can be most helpful—such as ovarian and pancreatic tumors, she says. Belcher’s team is also exploring ways to deliver drugs using viruses engineered to attach to cancer cells.
Mixing It Up
Belcher’s genetic toolkit has proved versatile, but great science doesn’t fully explain her ability to work across so many fields. In her lab, she creates an environment where experts from different disciplines—chemists, molecular biologists, physicists, and mechanical engineers—approach problems in unique ways. Berkeley’s Lee, for instance, joined Belcher’s lab with a background in polymer chemistry but says that by working with others in the lab, he learned enough materials science and bioengineering to investigate virus-built semiconductors. “She has a pretty uncanny ability to pick a great set of people for her lab at the boundaries of a lot of different disciplines where a lot of interesting science typically takes place,” says Eric Krauland, PhD ’07, the director of antibody discovery and optimization at the biotech company Adimab and a former PhD student in Belcher’s lab. And she’s not afraid to hire people who know more than she does in a particular area. “They’re teaching me,” she says. “It’s really a collaboration—I just get the big office.”
Belcher likes to call MIT an “intellectual playground” because of the ample opportunities to share ideas on cutting-edge research. “It’s not driven by money or the next paper. It’s driven by ‘Wow, let’s see what we can do together,’” she says. “That’s why it’s fun—that’s why it doesn’t seem like a job at all.”
If a common thread runs through Belcher’s work, it’s a belief that technology and engineering can help address societal problems. In a speech she made when she accepted the Lemelson-MIT prize in July, she urged high-school students who had won prizes from the Lemelson Foundation to try to make the world a better place. Using some of the prize money, she intends to expand an outreach program she’s used to kindle school-age children’s interest in science through hands-on experimentation. She visits schools and museums to host talks with students ranging from kindergarten to high school, leading them in experiments such as isolating DNA from their cheeks. Belcher has fond memories of tinkering, building things in the garage, and—despite being dyslexic—spending hours at the library reading books about medicine when she was a young girl. She hopes to share this passion for science with her sons, who are seven and three.
Belcher understands the importance of starting early: she herself became fascinated with the origins of life and the microscopic world when she was barely in elementary school. That same childlike fascination—and a desire to make a difference—is still what fuels her today. “I love solving problems that are important to the planet,” she says. “Every single day I wake up knowing that there’s something interesting happening in the lab.”
Keep Reading
Most Popular
Large language models can do jaw-dropping things. But nobody knows exactly why.
And that's a problem. Figuring it out is one of the biggest scientific puzzles of our time and a crucial step towards controlling more powerful future models.
How scientists traced a mysterious covid case back to six toilets
When wastewater surveillance turns into a hunt for a single infected individual, the ethics get tricky.
The problem with plug-in hybrids? Their drivers.
Plug-in hybrids are often sold as a transition to EVs, but new data from Europe shows we’re still underestimating the emissions they produce.
Google DeepMind’s new generative model makes Super Mario–like games from scratch
Genie learns how to control games by watching hours and hours of video. It could help train next-gen robots too.
Stay connected
Get the latest updates from
MIT Technology Review
Discover special offers, top stories, upcoming events, and more.