First Self-Assembling Diamond Quantum Devices Unveiled
One of the great goals of applied physics is to make quantum information processing a robust and common technique. To achieve this, physicists will need a simple way of storing and manipulating quantum information, preferably at room temperature.
There is no shortage of possible quantum storage devices but one sits head and shoulders above most others: a nitrogen atom that has replaced a carbon atom in a diamond lattice, an arrangement known as a nitrogen-vacancy centre.
Today, an international team of physicists say they’ve used biological self-assembly techniques to make diamond-based prototypes of the quantum information storage devices of this type. That’s a development that has the potential to profoundly influence the future of computing.
The key to all this is nitrogen-vacancy centres in diamond which behave like single atoms. They can store photons, emit them again and interact with other nitrogen-vacancy centres nearby. In fact, their photon storage ability is legendary, holding them, and the information the carry, for periods stretching to milliseconds. At room temperature.
That’s significantly longer and more robust than other quantum information storage devices.
When two nitrogen vacancy centres are close enough to together to interact, they can also process quantum information. But this is only possible at distances of less than 10 nanometres or so and therein lies the problem.
The conventional way of making nitrogen vacancy centres is to fire nitrogen ions into a diamond crystal and hope they embed themselves into the structure. But that can only be done at a resolution measured in tens of nanometres. So adjacent nitrogen-vacancy centres are always too far apart to interact.
Clearly some other way of bringing these centres together is desperately needed. Enter Andreas Albrecht at the University of Ulm in Germany and a few pals who have come up with a better way to do it using the self-assembling technique of molecular origami.
Their idea is conceptually simple. For some time now, biologists have been creating complicated three dimensional structures out of biomolecules. Their technique is based on the highly selective reactivity of biomolecules such as DNA. These can be designed to fit together and to other things, like self-assembling Lego bricks, to form three dimensional shapes that are nanoscopic in scale.
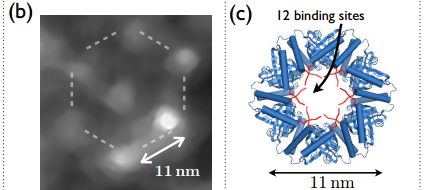
Albrecht and co simply modified a well known ring-shaped protein called SP1 so that it binds to diamond. In fact, they created 12 binding sites on this ring allowing it to hold six nanodiamonds in hexagonal formation.
They then used a laser to generate nanodiamonds just 5 nanometres across by blasting them off a larger crystal. They placed the resulting crystals in a liquid which they poured onto a layer of the modified SP1 rings.
When the nanodiamonds bonded with the rings, they formed hexagonal structures in which each nanodiamond is just a few nanometres from its neighbours. That’s exactly the kind of distance required for nitrogen vacancy centres to interact.
They go on to show theoretically that this kind of arrangement should allow for some serious quantum information processing.
Of course, there are challenges ahead. One is to repeat this method with nanodiamonds that actually contain nitrogen vacancies and then to experimentally show that they do indeed interact. Clearly, that must be high on the list of future priorities.
In the meantime, this kind of self-assembly is a powerful new approach that is full of promise. Biologists have been able to create a rich tapestry of shapes and structures using molecular origami.
There’s no reason why Albrecht and co can’t do the same to create vastly more complex arrangements of nitrogen vacancy centres.
These might well become the beating hearts of quantum chips of the future.
Ref: arxiv.org/abs/1301.1871: Self-Assembling Hybrid Diamond-Biological Quantum Devices
Keep Reading
Most Popular
Large language models can do jaw-dropping things. But nobody knows exactly why.
And that's a problem. Figuring it out is one of the biggest scientific puzzles of our time and a crucial step towards controlling more powerful future models.
The problem with plug-in hybrids? Their drivers.
Plug-in hybrids are often sold as a transition to EVs, but new data from Europe shows we’re still underestimating the emissions they produce.
How scientists traced a mysterious covid case back to six toilets
When wastewater surveillance turns into a hunt for a single infected individual, the ethics get tricky.
Google DeepMind’s new generative model makes Super Mario–like games from scratch
Genie learns how to control games by watching hours and hours of video. It could help train next-gen robots too.
Stay connected
Get the latest updates from
MIT Technology Review
Discover special offers, top stories, upcoming events, and more.