When Will Gene Therapy Come to the U.S.?
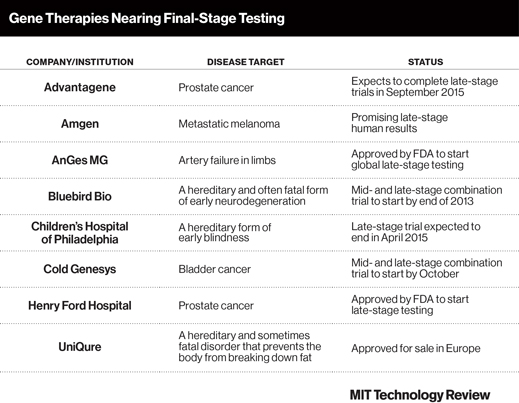
Though many gene therapies have been tested in patients around the world in hopes of curing hereditary diseases, few governments have approved their sale, and none has been approved in the United States. That could change in coming years as several therapies enter advanced trials.

A big step forward already came in November 2012, when the European Medicines Agency gave the Dutch biotech startup UniQure permission to sell its treatment. That approval came as a relief to many in the field, who had been waiting for a break in the clouds hanging over the technology since failed and fatal trials in the 1990s. “You see a resurgence in terms of investors, and in truth, a number of problems have been solved,” says Katherine High, a medical researcher at Children’s Hospital of Philadelphia, who is overseeing a late-stage clinical trial for a different gene therapy.
Still, experts say it is likely to be a few years before a treatment is approved in the U.S. With its European approval in hand, UniQure may have good chance of also getting the first U.S. approval, but the company says it has not yet submitted an application to the FDA.
Like most gene therapies, UniQure’s treatment uses a modified virus to deliver a working copy of a gene to patients who lack a healthy version. In this case, the gene is needed for the body to break down fats; without it, patients can develop painful and even fatal inflammation of the pancreas. UniQure uses a modified version of a virus that most of us already carry. The choice of virus used to deliver a gene therapy depends in part on where the treatment needs to go in the body and whether the viruses are intended to replicate themselves. Some viruses, for instance, are designed to spread throughout the body to kill cancer cells.
There are several groups that could be the first to develop a U.S.-approved gene therapy (see table). High’s team is one; they are enrolling patients in a late-stage trial of a treatment for a disorder that causes blindness at an early age. The patients in this trial have previously been given the gene therapy in one eye, and now the other will be tested.
In the experimental treatment, doctors inject a virus-borne gene just behind a patient’s retina. The treatment improved some patients’ vision to the point that they were no longer legally blind. Some patients have been stable for nearly six years. The trial is scheduled to end in April 2015.
Another possibility comes from Massachusetts-based Bluebird Bio, which has published results from patients who have seemingly been cured of a genetic blood disease (see “Gene Therapy Combats Hereditary Blood Disease”). The company is about to start testing its approach in a hereditary neurological disorder that is often fatal in young boys.
In a different form, gene therapy could also become an option for cancer treatment. At a meeting this summer, Amgen announced that it had met its goals for an advanced test of a gene therapy for melanoma that has spread from the skin to other parts of the body. The Amgen treatment, which was engineered from a virus that normally causes cold sores, takes a two-pronged approach to fighting cancer. The virus selectively infects cancer cells, where it replicates until the cell bursts. While growing inside the cell, the virus also produces a protein that rouses the immune system. When the cell explodes, immune cells are attracted to the tumor site to fight the disease.
In a test in patients with late-stage melanoma, 26 percent of patients whose cancer had spread saw a partial or complete tumor response for at least six months. In 11 percent of patients, the cancer completely disappeared, which suggests that the therapy spreads throughout the body, targeting tumors that weren’t initially injected. Overall survival rates for cancer patients in the trials are expected to be reported in the first half of 2014.
The completion of these clinical trials is no guarantee that the technology will become widely used. However, companies and hospitals are also starting to build manufacturing plants capable of producing large amounts of virus-based cures. UniQure, for example, announced in July that it is building a 55,000-square-foot plant in Massachusetts to manufacture commercial-grade gene therapy products. It expects the facility to be operational by early 2015.
Keep Reading
Most Popular
Large language models can do jaw-dropping things. But nobody knows exactly why.
And that's a problem. Figuring it out is one of the biggest scientific puzzles of our time and a crucial step towards controlling more powerful future models.
How scientists traced a mysterious covid case back to six toilets
When wastewater surveillance turns into a hunt for a single infected individual, the ethics get tricky.
The problem with plug-in hybrids? Their drivers.
Plug-in hybrids are often sold as a transition to EVs, but new data from Europe shows we’re still underestimating the emissions they produce.
Google DeepMind’s new generative model makes Super Mario–like games from scratch
Genie learns how to control games by watching hours and hours of video. It could help train next-gen robots too.
Stay connected
Get the latest updates from
MIT Technology Review
Discover special offers, top stories, upcoming events, and more.