Scientists Grow 3-D Human Brain Tissues
Scientists at the Institute of Molecular Biotechnology in Vienna, Austria, have grown three-dimensional human brain tissues from stem cells. The tissues form discrete structures that are seen in the developing brain.
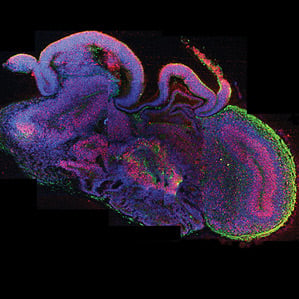
The Vienna researchers found that immature brain cells derived from stem cells self-organize into brain-like tissues in the right culture conditions. The “cerebral organoids,” as the researchers call them, grew to about four millimeters in size and could survive as long as 10 months. For decades, scientists have been able to take cells from animals including humans and grow them in a petri dish, but for the most part this has been done in two dimensions, with the cells grown in a thin layer in petri dishes. But in recent years, researchers have advanced tissue culture techniques so that three-dimensional brain tissue can grow in the lab. The new report from the Austrian team demonstrates that allowing immature brain cells to self-organize yields some of the largest and most complex lab-grown brain tissue, with distinct subregions and signs of functional neurons.
The work, published in Nature on Wednesday, is the latest advance in a field focused on creating more lifelike tissue cultures of neurons and related cells for studying brain function, disease, and repair. With a cultured cell model system that mimics the brain’s natural architecture, researchers would be able to look at how certain diseases occur and screen potential medications for toxicity and efficacy in a more natural setting, says Anja Kunze, a neuroengineer at the University of California, Los Angeles, who has developed three-dimensional brain tissue cultures to study Alzheimer’s disease.
The Austrian researchers coaxed cultured neurons to take on a three-dimensional organization using cell-friendly scaffolding materials in the cultures. The team also let the neuron progenitors control their own fate. “Stem cells have an amazing ability to self-organize,” said study first author Madeline Lancaster at a press briefing on Tuesday. Others groups have also recently seen success in allowing progenitor cells to self-organize, leading to reports of primitive eye structures, liver buds, and more (see “Growing Eyeballs” and “A Rudimentary Liver Is Grown from Stem Cells”).
The brain tissue formed discrete regions found in the early developing human brain, including regions that resemble parts of the cortex, the retina, and structures that produce cerebrospinal fluid. At the press briefing, senior author Juergen Knoblich said that while there have been numerous attempts to model human brain tissue in a culture using human cells, the complex human organ has proved difficult to replicate. Knoblich says the proto-brain resembles the developmental stage of a nine-week-old fetus’s brain.
While Knoblich’s group is focused on developmental questions, other groups are developing three-dimensional brain tissue cultures with the hopes of treating degenerative diseases or brain injury. A group at Georgia Institute of Technology has developed a three-dimensional neural culture to study brain injury, with the goal of identifying biomarkers that could be used to diagnose brain injury and potential drug targets for medications that can repair injured neurons. “It’s important to mimic the cellular architecture of the brain as much as possible because the mechanical response of that tissue is very dependent on its 3-D structure,” says biomedical engineer Michelle LaPlaca of Georgia Tech. Physical insults on cells in a three-dimensional culture will put stress on connections between cells and supporting material known as the extracellular matrix, she says.
Other researchers are developing three-dimensional brain tissue cultures to approach fundamental questions about how the brain works. Utkan Demirci, a biomedical engineer at Harvard Medical School and a 2006 MIT Technology Review Innovator Under 35, reported earlier this year that microfabrication techniques enabled his group to construct three-dimensional neuron cultures. Demirci’s lab is now using electrical recordings and other functional studies to show that there is synaptic activity amongst the neurons. “When you culture these cells in three dimensions, then the arms of the neurons can extend as they do in native tissues and build a circuit,” he says. “Once we show these are functional, we can do a lot of interesting studies with them, including explore brain mapping studies.”
After confirming the success of their methods with mouse stem cells, Knoblich, Lancaster, and colleagues used the methods to study a human developmental genetic disorder that causes microcephaly, a condition in which brain size is markedly reduced and is associated with severe cognitive disabilities. The team worked with a pediatric neurologist to obtain skin cells from a patient with microcephaly. From these cells, the team created induced pluripotent stem cells (see “TR10: Engineered Stem Cells”). The researchers then genetically reprogrammed these cells into primitive neurons and, with a few steps, cultured them into a cerebral organoid in which they were able to glean hints of the origin of the disease.
In the future, the team would like to use the brain tissue system to study schizophrenia and autism—cognitive disorders that are usually diagnosed in adolescents or adults but are thought to begin in early brain development.
Keep Reading
Most Popular
Large language models can do jaw-dropping things. But nobody knows exactly why.
And that's a problem. Figuring it out is one of the biggest scientific puzzles of our time and a crucial step towards controlling more powerful future models.
The problem with plug-in hybrids? Their drivers.
Plug-in hybrids are often sold as a transition to EVs, but new data from Europe shows we’re still underestimating the emissions they produce.
Google DeepMind’s new generative model makes Super Mario–like games from scratch
Genie learns how to control games by watching hours and hours of video. It could help train next-gen robots too.
How scientists traced a mysterious covid case back to six toilets
When wastewater surveillance turns into a hunt for a single infected individual, the ethics get tricky.
Stay connected
Get the latest updates from
MIT Technology Review
Discover special offers, top stories, upcoming events, and more.