Green Chemists Synthesise Vanillin From Sawdust
The demand for vanilla flavouring exceeds the supply from natural vanilla pods by as much as ten to one. So there is huge interest in finding cheap and easy ways to synthesise vanillin, the phenolic aldehyde that is the active ingredient in this flavouring.
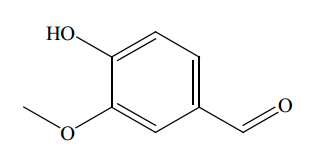
One long-standing approach is to make vanilllin from lignin, a biopolymer found in wood pulp and consequently one of the most common organic polymers on the planet. Second-hand book stores, tend to smell pleasant because the lignin in paper degrades into vanillin. In the 1980s, some 60 per cent of the world’s synthetic vanillin came from lignin waste produced by a single paper mill in Canada.
There is a problem with this approach, however. The commercial process for oxidising lignin relies on a mixture of sodium hydroxide and sodium sulphide, called white liquor, which is a strong base and highly corrosive. The by-products of this process later have to be neutralised with strong acids. Consequently, the production of vanillin in this way is so environmentally damaging that it is rarely used today.
Other approaches involving organic solvents are also avoided because these solvents are highly flammable and toxic and pose significant environmental risks.
Instead, chemists tend to synthesise vanillin using a greener but more expensive process based on the petrochemical material, guaiacol.
Nevertheless, there is widespread interest in finding an environmentally-friendly way to make vanillin from lignin because of the potential to be so cheap.
Today, Ahmad Shamsuri and D K Abdullah at University Putra Malaysia say they’ve discovered an environmentally-friendly way to oxidise lignin into vanillin that has the potential to change the economics of vanillin synthesis.
Their method is based on the relatively new chemistry of ionic liquids, salts that exist in a liquid state. These liquids consist of anions and cations that, while bound, are also able to move freely in a liquid state.
Most ionic liquids are hugely toxic. But in recent years, chemists have begin to play around with versions with anions and cations that are much less reactive and so less damaging.
One of these is 1,3-dimethylimidazolium methylsulphate– a relatively benign substance that is liquid at room temperature
Shamsuri and Abdullah began with lignin that they took from a local saw mill that processed rubber trees. They dissolved the lignin in the 1,3-dimethylimidazolium methylsulphate and then bubbled oxygen through the liquid. Finally, they then separated the vanillin and any other oxidation by-products by filtering the mixture.
The results of their analysis of this filtrate are interesting. Shamsuri and Abdullah used both Fourier transform infrared analysis and high pressure liquid chromatography to identify the presence of vanillin in this stuff.
That’s important because it shows the potential of ionic liquids to replace environmentally damaging processes with much greener ones. Indeed, the green chemistry movement is quietly changing the way the chemical industry does it its work and in the process making giant strides in helping to preserve the planet. It is one of the great untold stories of modern science.
Perhaps the future production of vanillin will be just a small part of this.
Ref: arxiv.org/abs/1306.2442: A Preliminary Study Of Oxidation Of Lignin From Rubber Wood To Vanillin In Ionic Liquid Medium
Keep Reading
Most Popular
Large language models can do jaw-dropping things. But nobody knows exactly why.
And that's a problem. Figuring it out is one of the biggest scientific puzzles of our time and a crucial step towards controlling more powerful future models.
The problem with plug-in hybrids? Their drivers.
Plug-in hybrids are often sold as a transition to EVs, but new data from Europe shows we’re still underestimating the emissions they produce.
How scientists traced a mysterious covid case back to six toilets
When wastewater surveillance turns into a hunt for a single infected individual, the ethics get tricky.
Google DeepMind’s new generative model makes Super Mario–like games from scratch
Genie learns how to control games by watching hours and hours of video. It could help train next-gen robots too.
Stay connected
Get the latest updates from
MIT Technology Review
Discover special offers, top stories, upcoming events, and more.