What It’s Like to See Again with an Artificial Retina
Elias Konstantopoulos gets spotty glimpses of the world each day for about four hours, or for however long he leaves his Argus II retina prosthesis turned on. The 74-year-old Maryland resident lost his sight from a progressive retinal disease over 30 years ago, but is able to perceive some things when he turns on the bionic vision system.
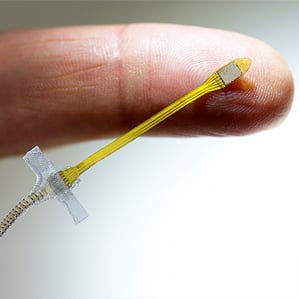
“I can see if you are in front of me, and if you try to go away,” he says. “Or, if I look at a big tree with the system on I can maybe see some darkness and if it’s bright outside and I move my head to the left or right I can see different shadows that tell me there is something there. There’s no way to tell what it is,” says Konstantopoulos.
A spectacle-mounted camera captures image data for Konstantopoulos; that data is then processed by a mini-computer carried on a strap and sent to a neuron-stimulating array of 60 electrodes that was implanted on one of his retinas in 2009.
Nearly 70 people around the world have undergone the three-hour surgery for the retinal implant, which was developed by California’s Second Sight and approved for use in Europe in 2011 and in the U.S. earlier this year (see “Bionic Eye Implant Approved for U.S. Patients”). It is the first vision-restoring implant sold to patients.
Currently, the system is only approved for patients with retinitis pigmentosa, a degenerative eye condition that strikes around one in 5,000 people worldwide, but it’s possible the Argus II and other artificial retinas in development could work for those with age-related macular degeneration, which affects one in 2,000 people in developed countries. In these conditions, the photoreceptor cells of the eye (commonly called rods and cones) are lost, but the rest of the neuronal pathway that communicates visual information to the brain is often still viable. Artificial retinas depend on this remaining circuitry, so cannot work for all forms of blindness.
Many groups around the world are working on bionic vision systems to replace lost photoreceptors. Most use a camera that communicates to an implanted chip, but vary in the number of electrodes in the chip and how deep the chip is placed inside the retina. Yet others eschew the camera for light-sensitive diodes in the chip.
German company Retina Implant, for example, recently completed human tests with its 1,500-pixel implant that does not depend on a camera but instead directly harvests light and transmits that data to remaining neurons (see “Microchip Restores Vision”). A photodiode array replaces the photoreceptors.

Some people with artificial retinas can read large letters, see slow-moving cars, or identify tableware. Other patients experience no benefit. The variation can be ascribed in some cases to the exact placement of the neuron-stimulating array in the tissue-paper-thin retina as well as the state of the remaining neurons and pathways in each individual’s eye. How well people can learn to use the device and retrain their brain is also important.
“Patients will scan their environment and use their memory to reconstruct what they are seeing,” says Raymond Iezzi, a clinician-scientist who performs retinal surgeries at Mayo Clinic in Rochester, Minnesota. He likens the scanning strategy to dragging a brush across a painting. “Patients have to fill in the blanks to connect visual input to a lexicon of nouns, and this requires a significant amount of cognitive ability,” says Iezzi.
At its best, the current level of vision is very pixelated. What’s seen are bursts of light called phosphenes. “It’s not truly naturalist vision,” says Iezzi. Second Sight says the level of visual acuity with its Argus II is 20/1,260 and Retina Implant says the best visual acuity gained with its device is 20/1,000. For comparison, normal vision is 20/20 and the threshold of legal blindness in the U.S. is 20/200 (which indicates that a person can see an object from 20 feet away that a normal-sighted person can see from 200 feet away).
“It’s not restoring vision like you and I think of, it’s restoring mobility,” says Stephen Rose, chief research officer for the Foundation Fighting Blindness. “They provide contrast so that someone can see a difference in light and dark to the point where they can tell how to walk through a doorway,” he says. “This is very much the beginning.”
“Retina prostheses are at the stage cochlear implants were 30 years ago,” says Anthony Burkitt, director of Bionic Vision Australia, a consortium of researchers developing a retinal implant system. “That technology went from being an aid for lip reading to the point now where children with a cochlear implant can go through normal school and even use mobile phones,” says Burkitt. “With retinal implants, we now know it has clinical benefit to patients, and I think we are going to see this technology develop very rapidly over the next decade.”
Experts agree that one way to improve the vision bestowed by these systems is to add more stimulating electrodes. Second Sight, for example, is planning on moving from 60 to 240 electrodes in a future model.
But thousands of pixels will likely be required for facial recognition and other detailed visual tasks, and many artificial retina technologies will have trouble getting to such large numbers of pixels because they depend on wires, says Daniel Palanker, a biophysicist at Stanford University. Wires are used to connect a power supply to electrodes, which requires a surgical procedure to lay the connection through the eyeball. To avoid this limitation, Palanker and colleagues are developing a wireless system that transmits image data captured by a video camera to a photovoltaic chip in the eye. Instead of transmitting visible light to the chip, his system uses near-infrared light that is beamed to flexible arrays of small pixels in the retina. The team has tested the system in blind rats and is now working with a company to test the device in patients.
But even thousands of pixels are a long way from one million, “which is roughly what we have in the natural eye,” says Shawn Kelly, an electrical engineer at Carnegie Mellon University in Pittsburgh. “And even at that, there is a lot of processing that the retina does that we are going to be skipping with an artificial retina.”
When photoreceptors detect light, they convert that information into chemical signals that stimulate other neuron types that process movement, color, and other details from the signals and then pass the information to the optic nerve, which connects to the brain. Depending on where the stimulating arrays are placed, artificial retinas will leave out one or more layers of information processing.
“The way we are interfacing with the nervous system is functional, but it’s not natural,” says Kelly. “I think it’s going to be a long time to develop ways to get better vision, and I don’t think it will ever be entirely natural.”
Still, the brain is remarkably adaptable and can learn to process even foreign information. “We count on brain plasticity to relearn this new language of stimulation and provide reasonable vision,” says Palanker. “The reassurance that this is a reasonable assumption comes from the field of cochlear implants.”
People with artificial retinas do achieve better vision over time. Tim Reddish, a 55-year-old Nottingham, England, resident who lost his vision due to retinitis pigmentosa, was implanted with a Retina Implant device in November and has achieved impressive results in the intervening months. He anticipates his abilities will continue to improve. In the lab setting, Reddish, a gold-medal-winning former paralympian swimmer, says he can identify cutlery and glassware, and even read time from a high-contrast clock. Outside, he says, he can detect the lines of buildings with glass doors and the headlights of slow moving cars at night. He hopes that come summer, a brighter U.K. sun and continued practice will help him see more.
Visual acuity is not the only challenge for these devices. The hard, silicon-based chips are placed in an extremely delicate tissue that is part of a fluid-filled moving orb. Chips can slip out of place. Furthermore, the implanted devices need to survive the tough conditions of the body for years without harming their users. “We are not going to know about very long-term safety for years,” says Rose.
Although approaching natural vision is the goal of many researchers developing artificial retinas, some are considering a wider world of light. “Some cameras work in wavelengths even humans don’t see, so potentially you could have blind patients using [a Second Sight system] to see things that others can’t,” says Brian Mech, vice president of business development with Second Sight. That company is also working on an idea that would forgo retinal stimulation for direct stimulation of the visual cortex, the brain region that processes images. “Instead of only being able to treat outer retinal degenerations, we could essentially treat blindness due to any cause,” says Mech.
In the meantime, researchers will continue to tweak the systems, trying to restore more vision to those who have lost it, and patients like Konstantopoulos are eager to see their progress. “Even that shadow I can see in front of me, whether it’s a person or anything else, is something from nothing,” says Konstantopoulos. “It makes you feel and hope that something better is coming up soon.”
Deep Dive
Humans and technology
Building a more reliable supply chain
Rapidly advancing technologies are building the modern supply chain, making transparent, collaborative, and data-driven systems a reality.
Building a data-driven health-care ecosystem
Harnessing data to improve the equity, affordability, and quality of the health care system.
Let’s not make the same mistakes with AI that we made with social media
Social media’s unregulated evolution over the past decade holds a lot of lessons that apply directly to AI companies and technologies.
Stay connected
Get the latest updates from
MIT Technology Review
Discover special offers, top stories, upcoming events, and more.