3-D Chip Promises Better Brain Control
Optogenetics, which pairs light-sensitive genes with a light source to selectively switch brain cells on or off, has shown promise as a research tool and a potential therapy. But the technology mostly delivers light to one spot, whereas brain activity usually involves complex sequences of activation in different locations. A new device takes optogenetics into three dimensions, with the ability to send patterns of light to neurons at various coördinates in the brain.
“In the coming few years, there will be tons of devices like this,” said Ilker Ozden, a research assistant professor in the Nanophotonics and Neuroengineering Laboratory at Brown University. Ozden was not involved in the research but is one of several researchers working on similar technologies.
“The brain is a 3-D object,” says Ed Boyden, lead author of the paper and head of the Synthetic Biology Group at MIT’s Media Lab.
Most optogenetics studies to date have used just one or two optic fibers to turn a piece of brain tissue on or off. Labs are now looking for ways to expand their level of control. While using a single light is like playing one note, Boyden says, a 3-D probe makes it possible “to play the brain like a piano.”
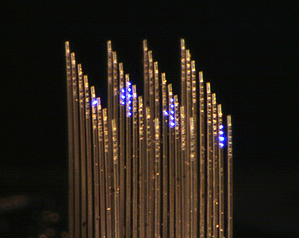
Two years ago, Boyden’s team, in collaboration with Clifton Fonstad’s lab at MIT, created a linear optogenetics probe with many parallel passages for light called waveguides, allowing light to be directed to any of several different points along the length of the probe.
The new device, described in the November 20 issue of Optics Letters, takes the technology further by creating an implantable, microfabricated block of these tiny prongs, each of which can emit light at many different depths. Boyden’s team used a laser light source and an array of tiny mirrors, which turn the light away or toward the individual waveguides, to create a pattern in time and space, while microlenses focus the light down the waveguides into the brain. Mirrors at the ends of the waveguides send the light out into cells.
The paper doesn’t demonstrate its use in the brain, but Boyden says his team is currently implanting the device into the brains of living mice and activating neurons in patterns.
The goal is to boost the amount of information and control that current optogenetics systems deliver. “With a 3-D array, there are a variety of things you can do that weren’t possible,” Boyden says. For instance, the technology could be used to systematically pinpoint the exact region of cells responsible for a specific behavior, or to create randomly generated patterns to study their effect.
It could also give much more control and flexibility to optogenetics-based neural prostheses, which Ozden says must deliver patterns of stimulation to different areas of the brain to be really effective. Boyden says that another long-term goal is to understand the specific sequences of brain activity that underlie disease processes or the brain’s response to therapies. These patterns could then be used as a way to test the efficacy of a drug, or to develop better interventions for disease.
Keep Reading
Most Popular
Large language models can do jaw-dropping things. But nobody knows exactly why.
And that's a problem. Figuring it out is one of the biggest scientific puzzles of our time and a crucial step towards controlling more powerful future models.
The problem with plug-in hybrids? Their drivers.
Plug-in hybrids are often sold as a transition to EVs, but new data from Europe shows we’re still underestimating the emissions they produce.
Google DeepMind’s new generative model makes Super Mario–like games from scratch
Genie learns how to control games by watching hours and hours of video. It could help train next-gen robots too.
How scientists traced a mysterious covid case back to six toilets
When wastewater surveillance turns into a hunt for a single infected individual, the ethics get tricky.
Stay connected
Get the latest updates from
MIT Technology Review
Discover special offers, top stories, upcoming events, and more.