New Chip Captures Hard-to-Find Tumor Cells
Technologies that analyze cancer cells that circulate through a patient’s bloodstream could provide a less invasive way of monitoring cancer and selecting the best treatments. So Mehmet Toner and collaborators at Massachusetts General Hospital have developed a microfluidics chip that effectively captures these rare cells, which make up just one in a billion cells in blood, in high enough numbers to analyze them for molecular markers. The device also isolated clusters of tumor cells for the first time, which may help shed light on cancer’s ability to spread, or metastasize, from its initial birthplace.
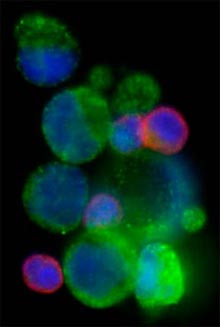
Even before tumors metastasize, they shed a small number of cancer cells into the bloodstream. The ability to simply take a blood sample from a patient and then capture these cells and analyze them for specific molecular markers could provide ways for physicians to select the best drugs for patients. The approach helps doctors avoid the invasive surgeries they now perform to collect tumor tissue for molecular testing.
The technologies are becoming increasingly important as the number of so-called targeted cancer therapies–drugs designed to attack a specific molecular glitch in a patient’s cancer–grows. Physicians need ways to easily assess their patients’ cancers for the molecular markers that suggest a particular treatment will work, as well as for signs the treatment is working or that the cells have evolved resistance to that drug.
“In my mind, this is definitely one of the most important new directions in the field,” says Alison Allan, an oncologist at the University of Western Ontario. “But the major problem in this field is the technology aspect. These cells are rare, and there have been few technologies validated to be sensitive enough, specific enough, and reproducible enough to be useful in a clinical setting.”
A device that uses a filtration-based system has already been approved by the U.S. Food and Drug Administration to monitor circulating cancer cells by counting the number of cells. But scientists are now testing their ability to analyze cells captured from blood in the sophisticated ways that pathologists can do with tissue from biopsies. For that, they need to capture more cells.
Toner’s team had previously developed a microfluidics chip that could isolate enough cells to detect a specific mutation in a gene for the epidermal growth factor receptor. The mutation suggests that patients will respond to drugs designed to inhibit this pathway. (Other researchers have shown that the FDA-approved device can also detect a molecular marker linked to drug response.)
Toner’s group has now developed a new version of the chip that can capture even more cells. The inner surface of the device has a herringbone design, which generates a vortex in the blood flowing through it. This mixing brings the cells in greater contact with antibodies on the surface of the chip. These antibodies bind to molecules specific to cancer cells. The research was published Monday in the Proceedings of the National Academy of Sciences.
Researchers also detected clusters of up to 14 circulating cancer cells, a phenomena that hasn’t been detected with other technologies. While the origins of these clusters aren’t yet clear, Toner speculates they may be linked to the process of metastasis; clumps of cells could lodge in small blood vessels and initiate the formation of secondary tumors.
The chip is made from a transparent plastic, which means that researchers can label the cells with different markers and use automated programs to analyze them. And Toner says the architecture of the new chip is cheaper and easier to manufacture on a large scale. Over the next two years, his team plans to finalize the design and then fabricate thousands of the devices, which will then be tested in clinical trials across the country.
Keep Reading
Most Popular
Large language models can do jaw-dropping things. But nobody knows exactly why.
And that's a problem. Figuring it out is one of the biggest scientific puzzles of our time and a crucial step towards controlling more powerful future models.
The problem with plug-in hybrids? Their drivers.
Plug-in hybrids are often sold as a transition to EVs, but new data from Europe shows we’re still underestimating the emissions they produce.
How scientists traced a mysterious covid case back to six toilets
When wastewater surveillance turns into a hunt for a single infected individual, the ethics get tricky.
Google DeepMind’s new generative model makes Super Mario–like games from scratch
Genie learns how to control games by watching hours and hours of video. It could help train next-gen robots too.
Stay connected
Get the latest updates from
MIT Technology Review
Discover special offers, top stories, upcoming events, and more.