Retinal Transplant Restores Vision in Mice
The prospect of restoring vision in people who have been blinded by disease is now on the verge of being a real possibility, thanks to the first successful transplant of retinal cells in mice.
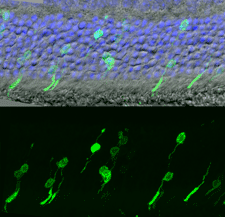
This is the first time that vision has been restored by transplanting light-sensitive photoreceptor cells, says Robert MacLaren, a clinician scientist at Institute of Ophthalmology and an eye surgeon at the Moorfields Eye Hospital, both in London, England. MacLaren, a member of the team that carried out the research, describes the results as a milestone as significant for vision as the first heart transplant was for cardiology.
Robert Lanza, vice president of research and scientific development at Advanced Cell Technology, a startup in Worcester, MA, agrees. He considers it a “huge” step toward treating people blinded by disease. “This is a major advance,” he says.
This is not the first time that cells have been transplanted from healthy eyes into those that are diseased. Research carried out by Advanced Cell Technology has shown that human epithelial cells–cells that nourish the photoreceptors in the retina–can be transplanted to halt the effects of degenerative eye disease.
But to have any hope of treating people already blinded by diseases such as macular degeneration and retinitis pigmentosa, the photoreceptor cells must be replaced, says MacLaren.
The latest research, funded by the UK’s Medical Research Council and published today in the journal Nature,suggests that this is now possible. “This is incredibly significant for patients that are already blind,” says Lanza.
In the study, the researchers harvested stem cells that were in the process of turning into light-sensitive photoreceptor cells and implanted them in the eyes of mice that had been bred to suffer from retinal degeneration. Subsequent tests on the mice revealed that the cells had successfully integrated into the retinas, forming functional connections with the existing host’s retinal cells. “These cells make enough connections in the host retina to restore a light response,” says MacLaren.
Previous efforts to do this have failed. According to MacLaren, the trick seemed to be to use stem cells that had stopped dividing and were beginning to actually turn into photoreceptors just before transplanting them into the host.
While it remains to be seen if this mouse experiment applies to humans, researchers say that creating supplies of transplantable cells in the lab should not be a problem. It’s already possible to make human embryonic stem cells turn into photoreceptors, says Lanza. “So this study is suggesting that we already have the raw materials we need in the lab.”
What’s more, there is evidence to suggest that stem cell-like cells exist within adult eyes, says MacLaren. The hope is that they can be harvested from the patient and reimplanted once they have differentiated into photoreceptors.
The eye is largely ignored by the immune system, so the hope is that patients receiving transplants will not need to take immunosuppressant drugs. But if stem cells can indeed be harvested from the patient, then the risk of rejection would be reduced even more, says MacLaren.
Keep Reading
Most Popular
Large language models can do jaw-dropping things. But nobody knows exactly why.
And that's a problem. Figuring it out is one of the biggest scientific puzzles of our time and a crucial step towards controlling more powerful future models.
How scientists traced a mysterious covid case back to six toilets
When wastewater surveillance turns into a hunt for a single infected individual, the ethics get tricky.
The problem with plug-in hybrids? Their drivers.
Plug-in hybrids are often sold as a transition to EVs, but new data from Europe shows we’re still underestimating the emissions they produce.
Stay connected
Get the latest updates from
MIT Technology Review
Discover special offers, top stories, upcoming events, and more.