Cody Friesen
Making cheaper, higher-energy batteries to store renewable energy.
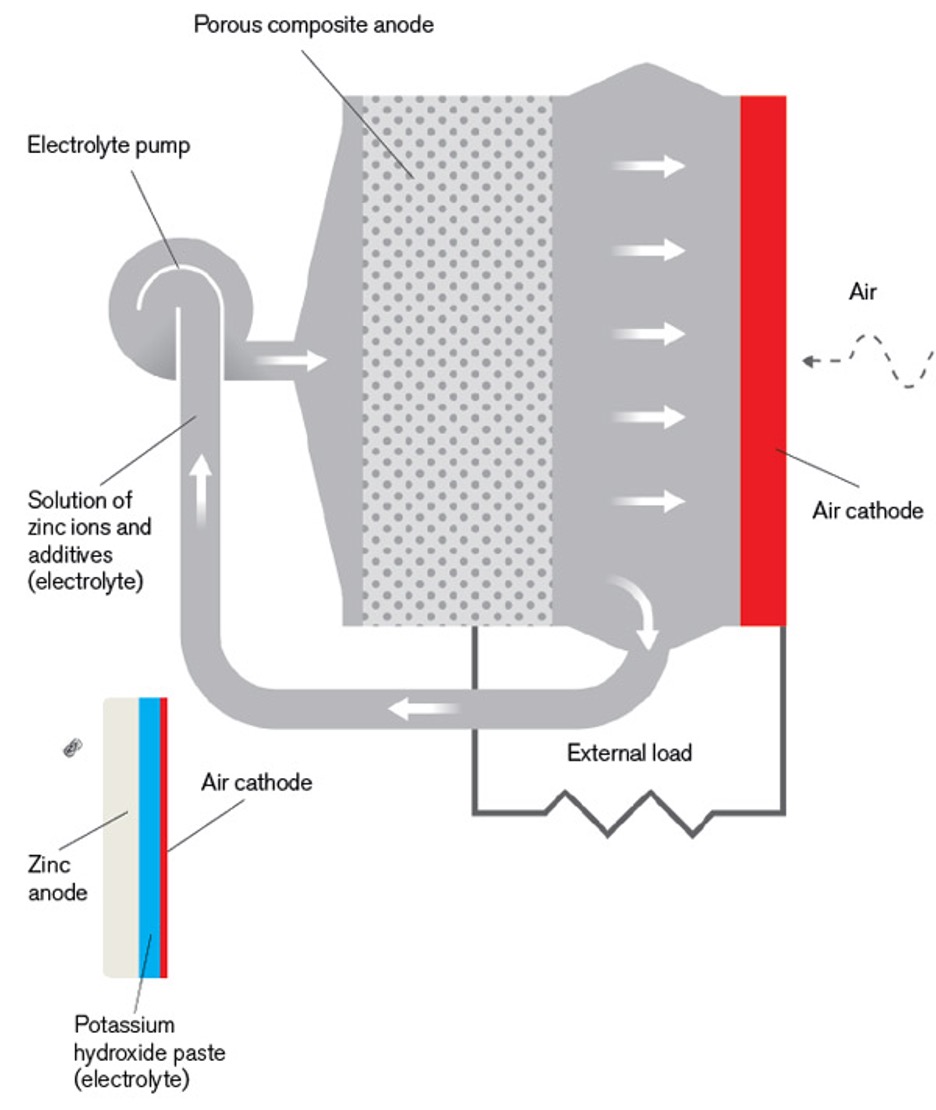
Zinc-air batteries, which use zinc metal as the anode and an alkaline paste as the electrolyte, are simple, inexpensive, nontoxic, and long-lasting. But engineers haven’t been able to figure out how to recharge them. Cody Friesen, an associate professor of materials science at Arizona State University, solved the problem by using a porous electrode and a liquid solution of zinc ions and additives as the electrolyte. He cofounded Fluidic Energy in 2007 to commercialize the design, and outside testing of its commercial prototype is planned for late fall. Within two years, Friesen hopes to be selling batteries that can hold twice as much energy as the lithium-ion batteries used in laptops and, increasingly, in electric cars. He calculates that his rechargeable metal-air batteries could ultimately hold 10 times as much energy as lithium-ion devices at a much lower cost. The first market he’s aiming for is storage of wind- and solar-generated electricity, but the batteries could also be useful in hybrid electric vehicles and portable electronics.
Ranjan Dash
Nanoporous carbon could help power hybrid cars.
Problem: Ultracapacitors, which last longer than conventional batteries and can deliver stronger bursts of power, hold great promise as energy-storage devices for applications such as hybrid electric vehicles. But ultracapacitors typically can’t store as much energy as batteries, so they need to be recharged frequently. That drawback has limited their use.
Solution: As a graduate student in materials science at Drexel University, Ranjan Dash used a novel chemical recipe to engineer nanoscopic pores into the carbon materials used in ultracapacitors. The tiny pores, whose size can be tuned with subnanometer precision, provide more surface area for charged particles to stick to, doubling the amount of energy the ultracapacitors can hold. Dash cofounded Y-Carbon, a startup based in King of Prussia, PA, to commercialize the technique, and he now serves as its chief technology officer. He says that his company has already developed a prototype ultracapacitor. The plan is to partner with other companies to develop this and other applications for the porous material, which Y-Carbon will manufacture. The first ultracapacitor products could be on sale in about two and a half years, Dash says.
Kurt Zenz House
Capturing carbon dioxide through cement production.
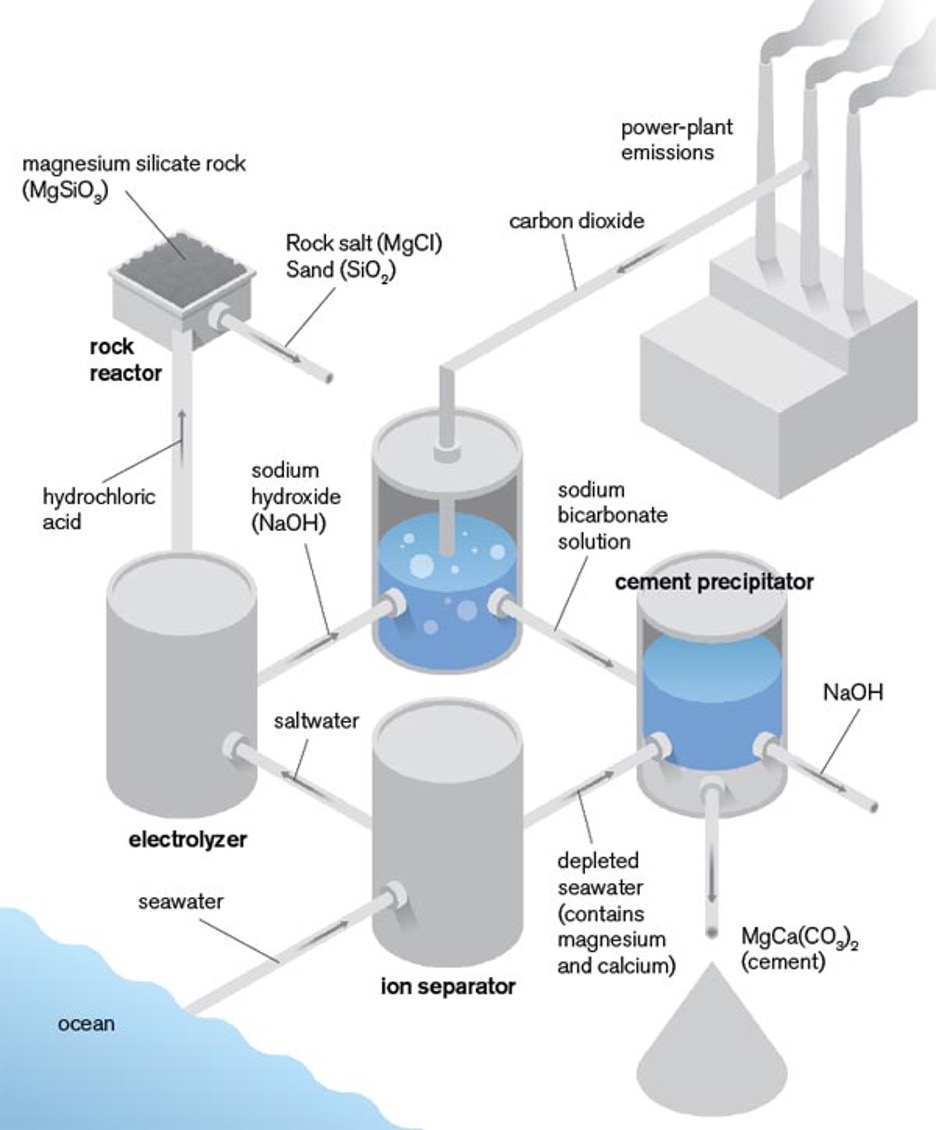
About 5 percent of global carbon dioxide emissions come from manufacturing cement. Kurt Zenz House, a research fellow at MIT and cofounder of a startup called C12 Energy, hopes to turn the problem into a solution. He thinks that the carbon dioxide from industrial smokestacks can be captured for use in cement production–keeping it out of the atmosphere for good.
The key to his approach is that alkaline solutions react with carbon dioxide and trap it in various compounds. For example, lye reacts with carbon dioxide to form baking soda. Combining the baking soda with seawater creates a type of cement, the glue that holds concrete together.
House says that regulations designed to limit greenhouse-gas emissions, such as a carbon tax, could eventually make this process profitable as well as environmentally sound. Meanwhile, he’s researching other ways to store carbon dioxide, including sequestration under the ocean and in geologic reservoirs on land. And at C12, he’s developing technology to reduce the cost of storing carbon dioxide.
Andrew Perlman
Slashing carbon emissions by converting coal into natural gas.
One of the biggest challenges in fighting global warming is reducing carbon dioxide emissions from the burning of coal, a cheap and abundant but dirty fuel. In 2004, serial entrepreneur Andrew Perlman founded GreatPoint Energy to commercialize a process for converting coal into methane (the main component of natural gas), which emits half as much carbon dioxide when burned to generate a given amount of electricity. The carbon dioxide that’s produced in the conversion process is easy to capture for storage underground. GreatPoint, where Perlman is president and CEO, has built a demonstration plant in Somerset, MA, and plans to build its first commercial plant as soon as next year. Meanwhile, Perlman has cofounded additional ventures that are developing, among other things, cheaper desalination plants, antiobesity medicines, drugs that fight diseases of old age, and processes for converting garbage into biofuels and generating electricity by extracting heat from rocks located miles underground.
Cyrus Wadia
Identifying materials that could be unexpectedly useful in solar cells
Solar power simply won’t be able to supply the terawatts of power we need until we identify better materials for solar cells. Silicon, which is used in most photovoltaics, is too expensive; the materials used to make cheaper thin-film solar cells, such as cadmium telluride, are rare–and some are toxic. To uncover other options, Berkeley Lab researcher Cyrus Wadia did an economic analysis of materials that have good electrical properties and can efficiently absorb sunlight. His results point to two previously overlooked materials: iron pyrite and copper sulfide. The analysis shows that the costs of extracting these compounds from existing reserves are low: less than .000002 cents per watt for iron pyrite and .0014 cents for copper sulfide. Wadia has since developed ways to synthesize pure nanoparticles of each. He’s made functional but, so far, low-efficiency solar cells from the copper sulfide nanoparticles and is working on iron pyrite cells. –Katherine Bourzac
