Andrew Lynn
Repairing joints by stimulating regrowth in bone and cartilage.
Andrew Lynn wants to phase out metal joint replacements by coaxing the human body to rebuild damaged bone and cartilage. Lynn, CEO and cofounder of Orthomimetics, in Cambridge, England, developed a biodegradable scaffold that a surgeon can implant into any joint weakened by injury or age.
Microscopic pores draw blood cells and stem cells into the plug-shaped scaffold, which is made of collagen and glycosaminoglycans, the materials in human cartilage. The part of the plug that extends into the bone also contains calcium phosphate, which is found in bone. The stem cells take cues from those materials to grow into bone or cartilage where appropriate, repairing the damage as the plug slowly dissolves. The scaffold is already approved for use in Europe, and Orthomimetics is performing more trials to gain approval from the U.S. Food and Drug Administration, which it anticipates in about two years. Scaffolding designed to regrow tendons and ligaments is also a couple of years away.

Jorge Conde
Offering consumers whole-genome sequencing--and software to interpret it.
Thanks to Jorge Conde, anyone can have his or her genome sequenced and scoured for clues to future health–all for just under $100,000. Conde is the driving force behind Knome, a personal-genomics startup founded in 2007 that is the first to offer whole-genome sequencing directly to consumers. The approach sets Knome apart from other consumer genomics companies, which analyze just a fraction of an individual’s DNA for a few hundred dollars.
Conde, Knome’s cofounder and CEO, thinks that the commercial value in personal genomics will lie less in sequencing itself than in interpretation. So the company has developed software to manage, protect, and analyze genetic data; the software combs online databases for the latest scientific findings that have been validated, ranks their relevance, and then uses them to probe an individual’s DNA sequence for helpful information.
While Knome’s service is still unaffordable for most people, the cost of DNA sequencing is plummeting–from millions of dollars in 2006 to tens of thousands in 2009. Conde believes that when the price of genome sequencing eventually lands within reach of the average consumer, possibly within the next five years, Knome’s whole-genome focus will put it far ahead of other companies.
José Gómez-Márquez
Imagination: Inventor José Gómez-Márquez gets much of his inspiration from his assortment of toys and cheap gadgets.
José Gómez-Márquez’s lab at MIT seems to be part toy store, part machine shop, and part medical center. Plastic toys are scattered across the bench tops, along with a disassembled drugstore pregnancy test, all manner of syringes, and a slew of fake body parts. Coffee filters have been transformed into paper-based diagnostics; a dime-store helicopter provides the design for a new asthma inhaler; even a toilet plunger has been put to use, rigged with tubes and glue to form a makeshift centrifuge.
“Centrifuges break down all the time,” says Gómez-Márquez, spinning the plunger’s wooden handle in his hands. That’s a problem for health-care workers, because even simple medical tests rely on the devices to separate molecules in a blood or urine sample. In rich countries, the broken equipment is quickly repaired or replaced; in the poor countries where Gómez-Márquez often works, finding replacement parts can be impossible, rendering the equipment useless. So he’s tried to use readily available materials to make simple versions that are either easy to fiddle with, disposable, or unlikely to break in the first place. “This one could work even without power,” he says of the plunger-cum-centrifuge.
Gómez-Márquez, a native of Honduras, is a talented tinkerer: “My mother used to say my toys would last only a few days because I would take them apart, saying I had detected a defect,” he recalls. But he is also an inventor on a mission. “When you grow up in a developing country,” he says, “you get the sense that fancy technology is expensive to replace, so it often doesn’t get replaced.”
In his few short years in the field, Gómez-Márquez has gained a reputation among Boston medical-device researchers for his insight into a wide array of design issues. “There are not many people out there that have as broad a view of innovative technology in low-resource settings,” says Kristian Olson, a physician at Massachusetts General Hospital in Boston and leader of the Global Health Initiative at the Center for Integration of Medical and Innovative Technology. “He finds a need and then a technology to fit that need. It’s a remarkable way of approaching technology development for poor people.”
Some might say that Gómez-Márquez was born to improve medical technology. In 1976, when prenatal ultrasound was not available to his mother’s doctor in Honduras (it was just catching on in the United States), the physician mistakenly concluded that she was carrying twins and miscalculated how far along she was in her pregnancy. She was induced to give birth in what was actually her seventh month, and Gómez-Márquez–no twin in sight–was born with the numerous health concerns typical of an underweight and premature infant. He escaped any long-term damage. But thanks to a childhood spent in and out of doctors’ offices, he developed a profound sense of how important health care was, how capricious access to it could be, and how much medical devices could do to improve it.
It helped, too, that he comes from a medical family. His grandfather, a surgeon, worked at both private and public hospitals in Tegucigalpa, the capital of Honduras, where Gómez-Márquez saw firsthand the differences that money made in access to medical services. Poor people, who went to the public hospital, were less likely to get chemotherapy or appropriate prostheses. “People who could afford it would go to Texas or Boston for their health care,” he says.
$$PAGE$$
Gómez-Márquez left Honduras in the late 1990s to attend college in the United States. But his education was sidetracked by Hurricane Mitch, which devastated Honduras in 1998. With his parents in Tegucigalpa no longer able to help pay his tuition, he began working a variety of jobs to support himself–“sometimes the late shift, sometimes regular shifts, sometimes cool stuff, and sometimes humbling stuff.” He eventually ended up at Worcester Polytechnic Institute in Massachusetts. “Quite frankly, it was very hard,” he says. “I wouldn’t recommend that path to anyone.”
What little free time he had was devoted to his real interest–combining aspects of different technologies to make them do new things. In 2005, the fledging inventor joined a team participating in MIT’s IDEAS competition, which aims to develop new technologies or programs that will have a positive impact on the world. His team focused on developing technologies for health care, inspired by a call from the World Health Organization (WHO) for new ways to deliver the measles vaccine; the disease, though nearly eradicated in the United States, still kills 500 people a day worldwide, mostly children. Gómez-Márquez and his coworkers decided to develop a needle-free system for use in poor countries.
Administering injectable vaccines takes training, and his team wanted “a device that would allow any community health-care worker, rather than a trained nurse, to give a dose of vaccine,” Gómez-Márquez says. While needle-free vaccination systems already existed, most relied on aerosolization technology that is cumbersome and requires electricity.
After examining prototypes of new kinds of devices from drug delivery and engineering firms, Gómez-Márquez quickly decided that existing approaches were much too complicated. “The device was sitting in a fancy box in eggshell foam,” he says of one prototype he studied. “If it needs foam to survive a trip to New England, it’s never going to make it to Central America.”
Taking inspiration from the disposable cartridges used in ink-jet printers, Gómez-Márquez’s team designed individual vaporizers preloaded with the correct dose of vaccine; the devices could be used once and then thrown away. The team also developed a way to stabilize the vaccine within the cartridges, eliminating the need for cold storage. Other researchers who’ve worked on vaccine nebulizers have tried to modify asthma nebulizers, which are not disposable or cheap. “If the aerosolizing head broke, the machine had to be sent back,” Gómez-Márquez says. “In our case, because it’s just 10 cents, you can throw the broken one away and pull another one out of the box.” Most nebulizers also require a power source to run the compressor, which converts liquid medicine into vapor. Gómez-Márquez’s team made sure their system can be powered by multiple sources, such as a foot pump, when electricity isn’t available.
The resulting device won an Award for International Technology at the IDEAS competition in 2006. “That gave me the idea that I could really do this for a living,” Gómez-Márquez says. The team is still improving the device, with funding from the U.S. National Institutes of Health.
$$PAGE$$
Gómez-Márquez and a team entered the IDEAS competition again the next year, this time focusing on strategies to improve drug compliance in tuberculosis treatment. Though TB can be treated with antibiotics, the disease killed 1.8 million people in 2007, according to WHO estimates, and 9.3 million new cases were diagnosed the same year. One of the biggest challenges in treating TB is making sure that patients stick with the lengthy course of medication required to clear the infection from the body.
A number of new technologies for monitoring drug compliance are being tested, but most of them rely to some degree on self-reporting; for example, patients may be given a phone number to call when they’ve taken their medicine. “I was the cynic,” says Gómez-Márquez. “Patients had no incentive to tell us if they didn’t take their drugs.” His team developed a simple paper-based test that can be used in conjunction with an incentive program. The test strip is made from layers of coffee filters impregnated with chemicals that react with metabolites of the TB drug in urine, revealing a unique code. The patient texts the code to a central database every day to earn cell-phone minutes. The team launched a trial of the system in Karachi, Pakistan, earlier this year.
Amy Smith, cofounder of the International Development Initiative at MIT, was so impressed with Gómez-Márquez that she hired him to run the Innovations in International Health program, also at MIT, in 2007. He has since built a network of collaborators in countries around the world who can tell him about their populations’ health-care needs. That kind of consultation, which lies at the heart of Gómez-Márquez’s philosophy, reflects a growing trend in the field of appropriate technology: collaborating early on with the people who will use the devices. “José is really at the forefront of that,” says Catherine Klapperich, director of the Biomedical Microdevices and Microenvironments Laboratory at Boston University. “Who is the customer, what do they want, what are they willing to use? You can’t assume the answers–that’s one of the things José teaches his students and his colleagues.”

Gómez-Márquez aims to push this approach even further by encouraging physicians and scientists in poor countries to design their own devices. He is now creating development kits for medical technology–sort of like Erector sets for medical professionals–which will initially be used in Nicaragua. The kits will enable doctors and medical students to devise diagnostics, drug delivery devices, microfluidic chips, and more.
At least, that is the theory. But will busy medical professionals find the kits helpful? Are they too complicated to use or–conversely–too simple to yield useful technology? Gómez-Márquez and his students brought some to Nicaragua over the summer to find out. His hope is that the kits will help a new culture of invention take root. In the long term, it’s this kind of approach that could truly revolutionize both medical care and technology in poor countries.
Michelle Khine
A children’s toy inspires a cheap, easy production method for high-tech diagnostic chips.
In 2006, Michelle Khine arrived at the University of California’s brand-new Merced campus eager to establish her first lab. She was experimenting with tiny liquid-filled channels in hopes of devising chip-based diagnostic tests, a discipline called microfluidics. The trouble was, the specialized equipment that she previously used to make microfluidic chips cost more than $100,000–money that wasn’t immediately available. “I’m a very impatient person,” says Khine, now an assistant professor at the University of California, Irvine. “I wanted to figure out how I could set things up really quickly.”
Racking her brain for a quick-and-dirty way to make microfluidic devices, Khine remembered her favorite childhood toy: Shrinky Dinks, large sheets of thin plastic that can be colored with paint or ink and then shrunk in a hot oven. “I thought if I could print out the [designs] at a certain resolution and then make them shrink, I could make channels the right size for microfluidics,” she says.
To test her idea, she whipped up a channel design in AutoCAD, printed it out on Shrinky Dink material using a laser printer, and stuck the result in a toaster oven. As the plastic shrank, the ink particles on its surface clumped together, forming tiny ridges. That was exactly the effect Khine wanted. When she poured a flexible polymer known as PDMS onto the surface of the cooled Shrinky Dink, the ink ridges created tiny channels in the surface of the polymer as it hardened. She pulled the PDMS away from the Shrinky Dink mold, and voilà: a finished microfluidic device that cost less than a fast-food meal.
Khine began using the chips in her experiments, but she didn’t view her toaster-oven hack as a breakthrough right away. “I thought it would be something to hold me over until we got the proper equipment in place,” she says. But when she published a short paper about her technique, she was floored by the response she got from scientists all over the world. “I had no idea people were going to be so interested,” Khine says.
At the same time, she faced considerable skepticism. How on earth, critics wondered, could you use a toy to make a sophisticated device that’s normally forged from high-grade silicon? “People either love it or they laugh at me,” Khine says. She hastens to point out that Shrinky Dink microfluidics isn’t perfect–minute ink splatters from the printer, for instance, can give rise to slight irregularities in the finished channels.
Still, glitches like these don’t pose a problem for most applications. And Khine has already found a way around a more serious difficulty: PDMS can absorb proteins, throwing off the results of sensitive tests. She has begun to make chips directly out of the Shrinky Dinks by etching the design into the plastic using syringe tips. As the plastic shrinks, the channels become narrower and deeper–perfect for microfluidics. She can even make three-dimensional chips by melting several etched Shrinky Dinks together. The whole process, from design to finished chip, takes only minutes.
Khine plans to use her chips to detect various medical conditions, and she hopes the cheap and portable devices will someday be used to diagnose HIV and other infections at the bedside. She has also found that by growing stem cells in a Shrinky Dink device that contains wells instead of channels, she can coax them to become heart muscle cells. Such a tool might allow researchers trying to grow those cells for tissue transplants to control the process more closely.
Douglas Crawford, associate executive director of the California Institute for Quantitative Biosciences, sees advantages in Khine’s approach. “Michelle’s technique is better, faster, and cheaper–it can put microfluidic prototyping into the hands of every lab,” he says.
Khine recently printed metal patterns on Shrinky Dinks. As the plastic shrinks, the metal buckles to form shallow wells, which Khine thinks may concentrate sunlight; the discovery could help make solar cells more efficient. “We haven’t come close to pushing the limits of this technology yet,” she says.
Erez Lieberman-Aiden
Quantitative tools offer new insights into evolution.
When Erez Lieberman-Aiden started his PhD in applied math in 2003, evolutionary theory couldn’t handle the complex shapes of real-world populations. So he helped it adapt by combining it with specialized mathematical tools. His advances at the Harvard-MIT Division of Health Sciences and Technology have allowed evolutionary biologists to include more variables in their models.
His next challenge: “People talk about the evolution of culture and language, but I found myself wondering whether evolution is really relevant to culture,” Lieberman-Aiden says. Working with Google, he and colleagues are building tools that can scan massive collections of digital texts and quantify how often a word–say, communism–appears in those from a particular era and place. This makes it easy to observe the movement of ideas, culture, and language across time and space.
Recently, Lieberman-Aiden has shifted his research toward genomics. Scientists can determine the sequence of bases in DNA, but they’ve had no way to know the genome’s overall structure. Lieberman-Aiden has codeveloped a method that determines both sequence and structure–revealing, for instance, conformational changes that bring two genes close even though they’re far apart along the length of a chromosome.
Ellis Meng
Micropumps deliver drugs that prevent blindness.
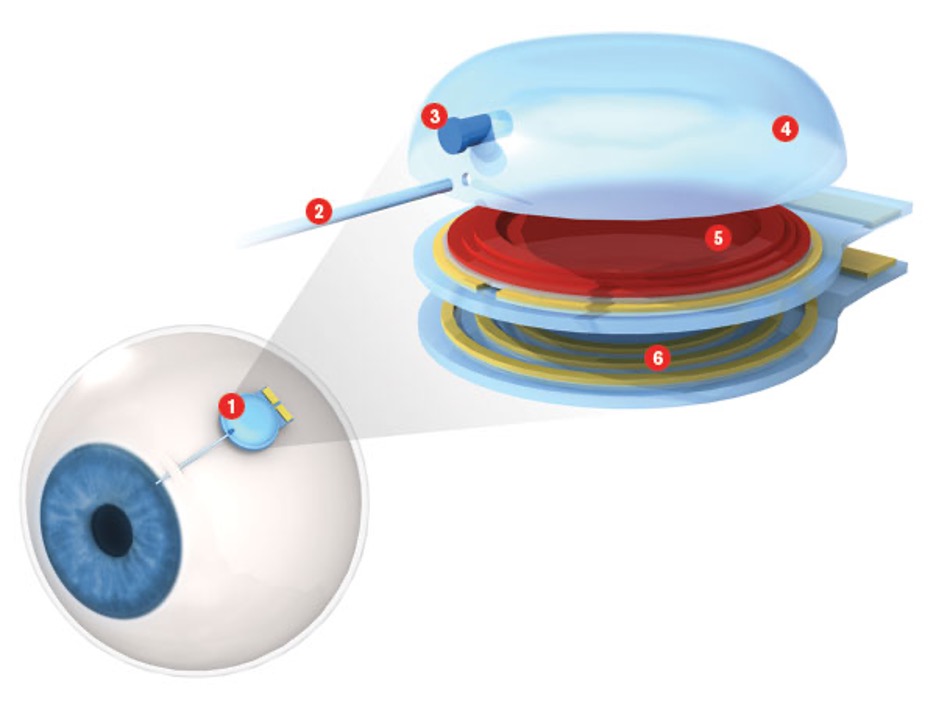
Treating many of the diseases that cause blindness involves frequent, painful injections directly into the eyes, putting patients at risk for infection, cataracts, and torn retinas. Ellis Meng, an assistant professor of biomedical and electrical engineering, has built an implantable pump to deliver medications more safely.
About the size of a watch battery, her device uses a microfluidic pump to push medications from a reservoir through a small tube and into the eye. A surgeon implants the pump and reservoir on the outer surface of the eye; only the tube enters the eye itself. And unlike existing implants that must be replaced periodically as they run out of drugs, Meng’s is refillable. Instead of weekly injections or monthly surgeries, a patient could take just one trip to the operating room, dramatically reducing both pain and risk. Meng is still testing the eye pump in animals but hopes it can be tested in humans within five years.
C. Shad Thaxton
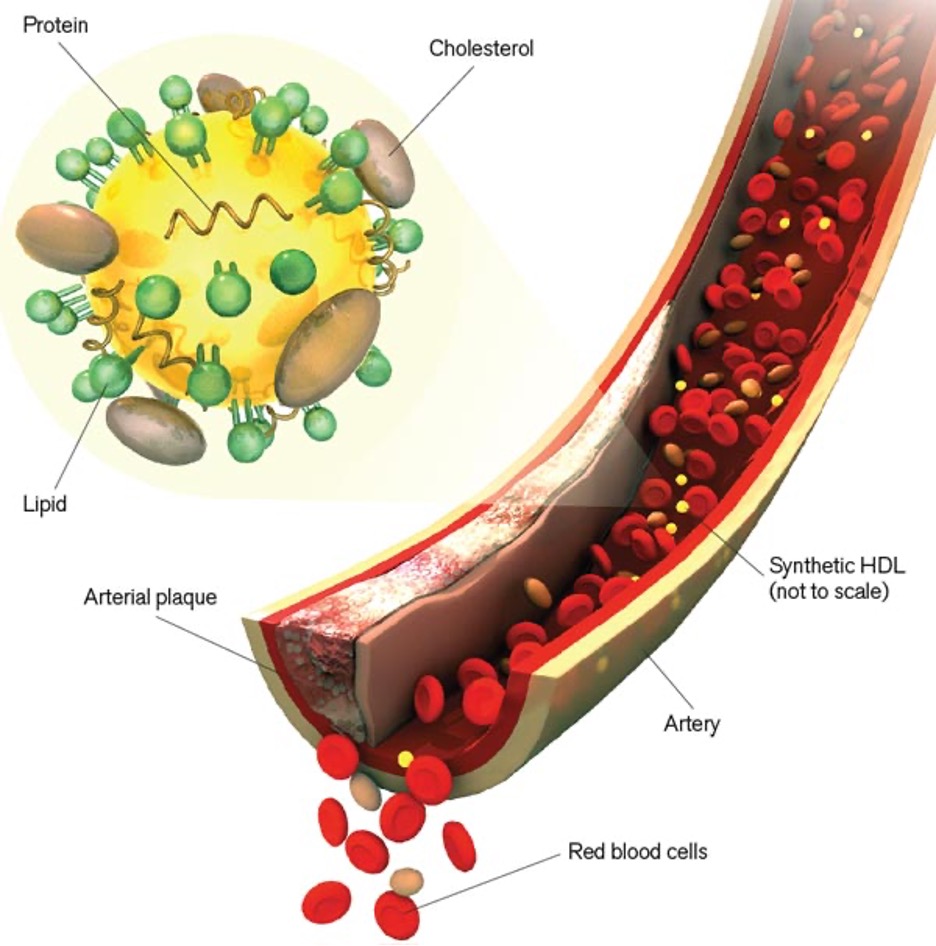
Nanoparticles could treat cardiovascular disease by mimicking “good cholesterol”.
To combat cardiovascular disease, Shad Thaxton, an assistant professor of urology, designed a nanoparticle that may be able to carry cholesterol right out of the body.
Several drugs treat cardiovascular disease by lowering levels of the lipoprotein complex LDL, commonly called “bad cholesterol” because it deposits the cholesterol in blood-vessel walls. But no existing therapies can directly increase HDL, or “good cholesterol,” which carries the sticky molecule through the bloodstream and to the liver for excretion. Thaxton’s nanoparticles mimic HDL. At their heart are gold spheres five nanometers in diameter; these are coated with fat and protein molecules that enable them to bind tightly to cholesterol. The work is in its early stages, but Thaxton envisions synthetic-HDL nanoparticles that will transport cholesterol from blood-vessel plaques to the liver to prevent and treat cardiovascular disease. If proved safe and effective, he says, synthetic HDL could be used to prevent heart attacks and strokes within 10 years.