Adam Cohen
Making molecules motionless.
How do you get a molecule to stop jiggling long enough to get a good look at it? It’s a problem that has vexed biologists for years. But Adam Cohen, who worked on it for the last four years as a graduate student at Stanford University, has solved it.
Tiny objects, such as cells, viruses, and proteins, naturally flitter about in solution. It’s called Brownian motion, and Albert Einstein explained it more than a century ago: particles in solution are invisibly bumped by water molecules. Researchers have been largely powerless to stop the jiggling of particles smaller than 100 nanometers, making it difficult to directly observe, for example, a single protein in its natural environment.
By developing a method for gently stopping the motion of nanoparticles, Cohen has given scientists a way to more easily discern differences between individual molecules and determine how those differences affect biological processes. Cohen points out that biological molecules like proteins exhibit “lots of variability.” That variability–say, how an odd protein is misshaped–can have important medical implications.
Optical tweezers, invented in the mid-1980s, use lasers to trap objects in solution by exerting a force on them. This technique works well with objects larger than 100 nanometers, but Cohen knew that a laser trained on something as small as a protein would “cook it rather than trap it.” So he turned to electrokinetic forces.
Cohen traps his molecules using a microfluidic chamber lined with electrodes. The microfluidic device is mounted under a standard fluorescence microscope that sends images of the targeted nanoparticle to a PC, which quickly determines the exact location of the molecule and applies voltages to the tiny chamber. These precisely calibrated voltages create minuscule drifts in the fluid that cancel out the Brownian motion of the molecule. “It’s like balancing a broom pole on your palm,” Cohen explains. “If you keep adjusting your palm, you can keep it balanced.”
Cohen begins work as an assistant professor of physical chemistry at Harvard University this fall. Now that his instrument works, he says, he is anticipating “the real excitement of making measurements on biological molecules.”
Erik Bakkers
Combining semiconductors.
Silicon chips have revolutionized electronics, but for certain purposes, such as radio frequency transmission, chips made from compound semiconductors like gallium arsenide or indium phosphide work much better. Erik Bakkers of Philips Research Laboratories in Eindhoven, the Netherlands, has found a way to mix semiconductors on a single chip.
Different semiconductors are normally incompatible, partly because they expand at different rates when heated. Combining them thus leads to physical strain that reduces performance. Bakkers solved the problem by building circuits out of nanowires. Because the point of contact between the different semiconductors is small–just a few tens of nanometers–there is no strain.
To grow a nanowire, Bakkers places a gold nanoparticle on top of a silicon wafer. Then he exposes the wafer to a vapor of, say, gallium arsenide; the nanoparticle catalyzes the growth of a gallium arsenide nanowire.
This technique opens up possibilities for multipurpose chips that could be used in wireless devices and other applications. It could also make it easier for engineers to take advantage of the inherent properties of compound semiconductors to create highly efficient LEDs, faster transistors, optical interconnects to rapidly shunt data around chips, or fast, highly sensitive biosensors to detect diseases.
Ju Li
Modeling designer materials.
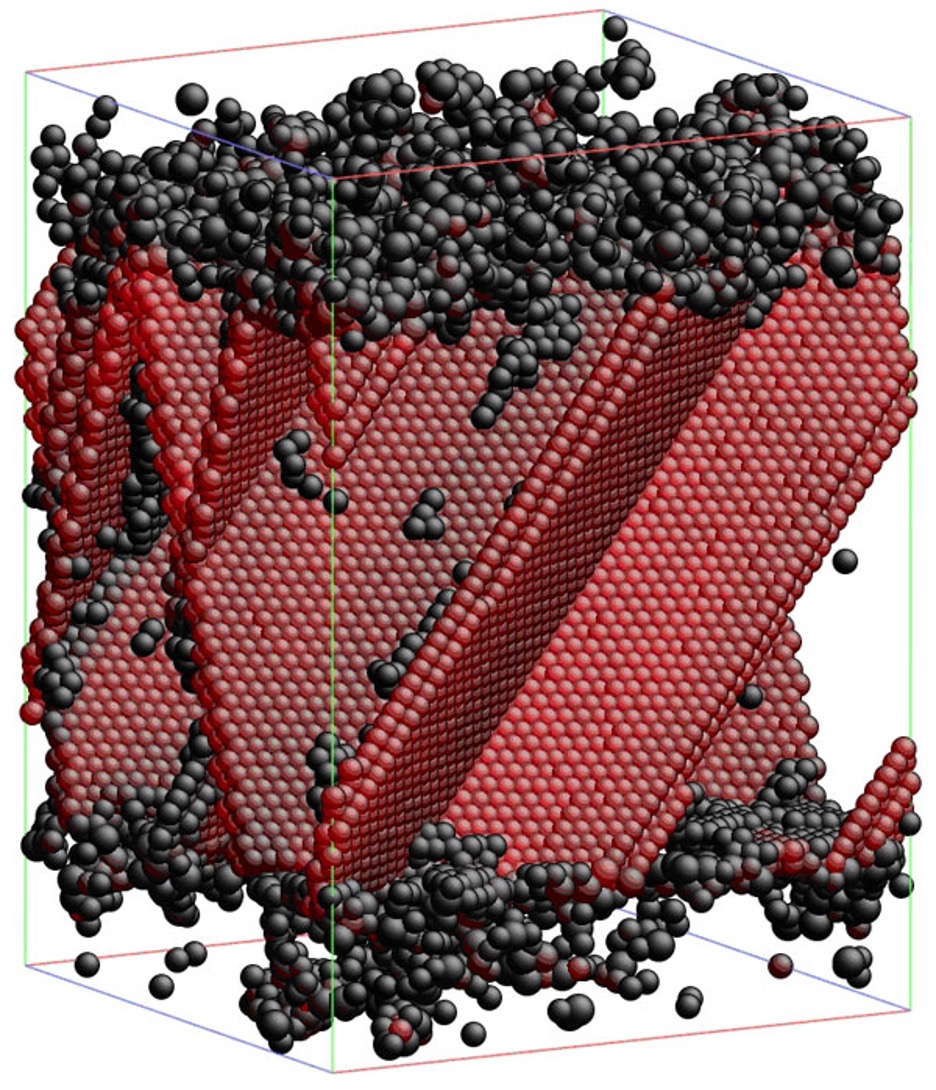
Researchers have long hoped that computer simulations of how atoms interact would allow them to design useful new materials from scratch. But the physics of atomic interactions rapidly becomes so complex that using it to predict the properties and performance of real-world materials has proved extremely difficult. Ju Li, an assistant professor of materials science and engineering, has developed new algorithms to model some of the hardest-to-understand phenomena in his field: the mechanical properties of complex, nanostructured materials. In one model, illustrated above, Li shows that combining nanoscale layers of amorphous copper zirconium and crystalline copper yields a material as much as ten times as strong as copper, without making it too brittle to be useful. In the crystalline region in the middle, atoms in one plane slip by atoms in neighboring planes, allowing the material to easily change shape under stress. The outer amorphous layers don’t change shape that way, so they keep the planes from slipping too far. Li’s collaborators have already shown experimentally that this structure results in a strong but malleable material.