Abraham Stroock
Microfluidic biomaterials.
When Abraham Stroock looks at a tree, he sees a complex feat of engineering. Inside the trunk, the branches, and the leaves, an intricate network of capillaries draws water dozens of meters into the air, with nary a pump in sight. This incredible system inspires Stroock’s approach to microfluidics.
Microfluidics involves moving tiny volumes of liquid through channels that are usually etched into a rigid material such as glass or silicon. Stroock, however, works with hydrogels, soft polymers that absorb water. Recently, he molded a capillary system that mimics a tree’s into a slab of hydrogel. This “synthetic tree” uses evaporation to pull water through its capillaries. The force it achieves is equivalent to that required to move liquid up a vertical column 85 meters high–the height of a redwood.
Liquid diffuses out of the hydrogel capillaries and into the surrounding material, just as it would in living tissue. Hydrogels are also biologically compatible, so such systems could serve as wound dressings that remove fluid and deliver drugs to promote healing. They could also act as three-dimensional scaffolds for engineered tissues; oxygen and nutrients could travel to cells inside the scaffolds. To mimic real tissues, the materials will ultimately have to provide conduits for proteins and cells–another step in Stroock’s plan to “give the material life.”
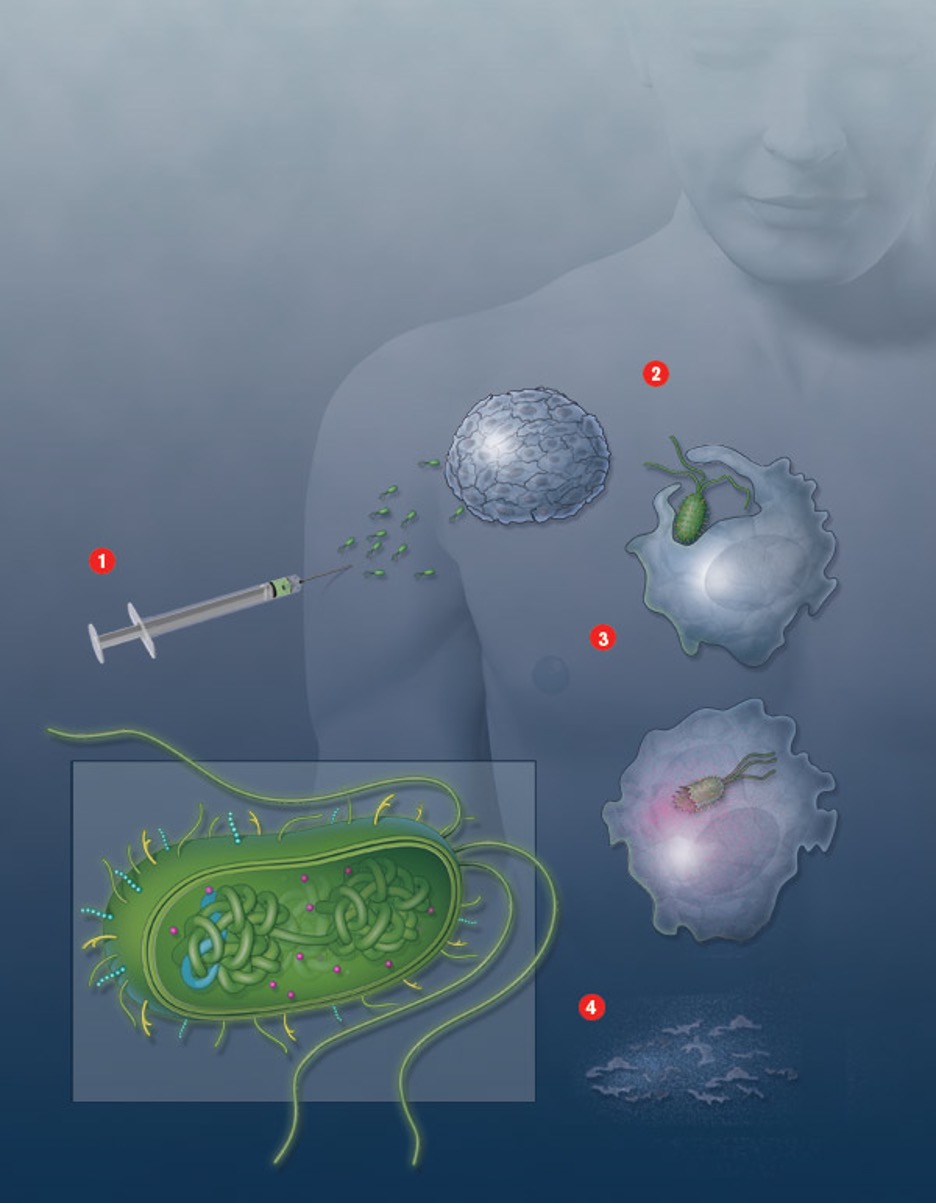
J. Christopher Anderson
Creating tumor-killing bacteria.
Using the engineering approach of synthetic biology, Chris Anderson has set out to program bacteria to selectively kill cancer cells. He is combining DNA sequences from different types of bacteria and inserting them into the bacterium E. coli to create an organism that can evade the immune system, home in on tumors, and trick cancer cells into letting it inside, where it releases a toxin.
Anderson has built and tested all the biological parts for the cancer-killing bug and is now working on putting them together. “All of these things exist as little genetic programs,” he says. He also expects to be able to engineer bacteria for other medical purposes, because “everything is designed in a modular way, so the parts can be used for a totally different application that shares some of the same problems.” For example, the genetic parts he has developed could be used to deliver medicine to an HIV-infected immune cell.
Kristala Jones Prather
Reverse-engineering biology.
Scientists are increasingly looking for ways to make compounds using biological processes rather than chemical reactions. Such techniques could provide environmentally cleaner ways to manufacture everything from biofuels to drugs, avoiding the harsh solvents and toxic by-products associated with more conventional synthesis. Kristala Jones Prather, an assistant professor of chemical engineering, is developing a promising strategy for synthesizing commercial molecules biologically, from start to finish.
Organic chemists often begin with the structure of a molecule they want to make, then look for the simplest pathway of precursors to produce it. The strategy is called retrosynthesis. Prather believes that biologists can use similar reverse-engineering principles–she calls it “retrobiosynthesis”–to build compounds, stringing natural and engineered enzymes together in novel combinations inside microbial hosts such as E. coli.
“What I’m interested in is designing organisms to be chemical factories,” says Prather, who spent four years in the Bioprocess Research and Development department at Merck. “We used biological systems to do one reaction, and we passed that back to a group of chemists who would do the rest of the fun stuff, and I started thinking, ‘Why can’t biology do more?’”
In 2004, Prather left industry and joined academia so she could help biology do more. Enzymes catalyze a wide range of chemical reactions. Prather is developing a database of these reactions; it includes algorithms that will identify the enzymes most useful for constructing novel metabolic pathways–in many cases leading to chemicals that are not produced through any natural biosynthetic pathway. When multiple enzymes might fit the bill, her software will help select the best one; when no appropriate enzyme is known, the program will help determine which existing enzyme should be modified in order to fill the hole. Prather’s software should be a boon to other synthetic biologists who now construct metabolic pathways by painstakingly combing the literature for possible enzymes, says Jay Keasling, a leading synthetic biologist at the University of California, Berkeley, who supervised her PhD thesis.
Adds John Woodley, professor of chemical engineering at the Technical University of Denmark, “It’s a very clever idea.”
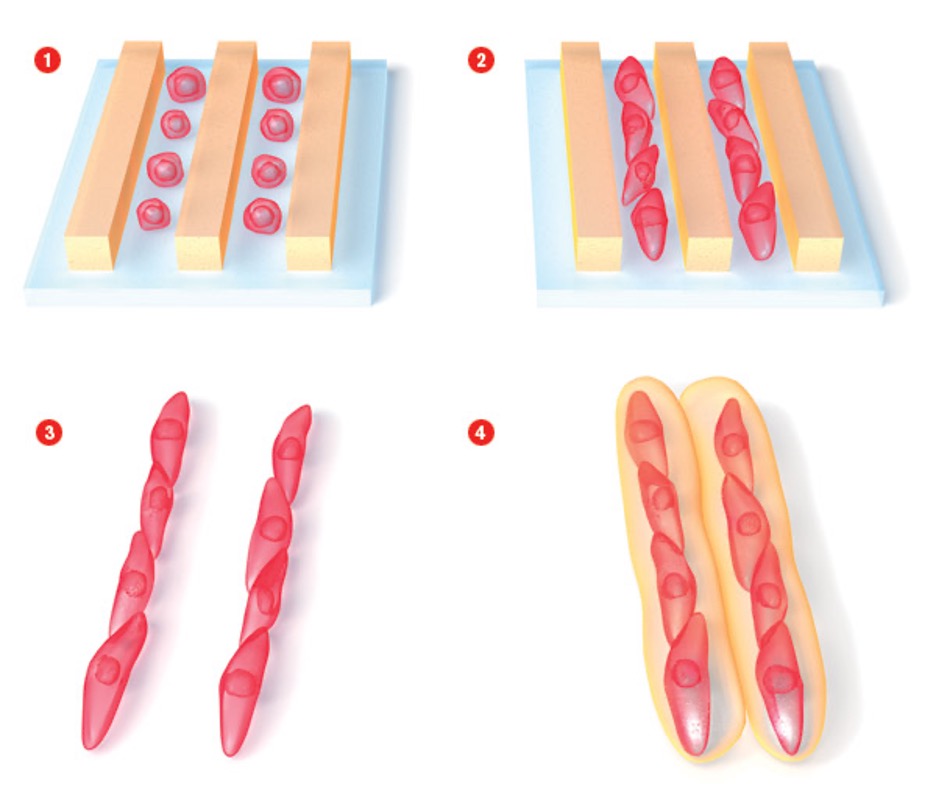
Ali Khademhosseini
Living Legos.
The ability to create living tissues and organs in the lab holds great promise for transplant medicine. But the traditional approach to tissue engineering–seeding the outside of a biodegradable scaffold with cells, without regard to their organization–hasn’t gotten cells to behave the way they would in the body.
Ali Khademhosseini, assistant professor in medicine and health sciences and technology at Harvard Medical School, hopes to improve engineered tissues with an approach he likens to building with living Legos. As a first step toward creating a heart, he aligns cardiac muscle cells to form small, beating strings. He then embeds these strings in a supportive, gelatinous polymer to make building blocks that can be assembled into bundles resembling the sheets of muscle that make up the heart. He can also add other types of cells to the building blocks to provide support for the muscle. This aspect of the system is crucial, since natural tissues comprise cells of multiple types in structurally complex arrangements. By giving cells the same interconnections they have in the body, Khademhosseini hopes to create tissues that can be used to test new drugs and, eventually, to rebuild organs.
Christopher Loose
Beating up bacteria.
Problem: Each year, a million Americans suffer infections related to medical devices such as the intravenous lines that deliver chemotherapy and liquid nutrition. Adding slow-release antibiotic coatings to the devices helps prevent such infections, but the coatings become inactive when all the drugs have been released, and bacteria can become resistant to them.
Solution: As a graduate student at MIT, Christopher Loose created a design tool to optimize formulations of naturally occurring antibiotics called antimicrobial peptides (AMPs), and developed a way to use them in medical devices. Found in bacteria, human sweat, and plants, these short proteins puncture bacteria like balloons. The mechanism is nonspecific, so microbes have trouble developing resistance to the peptides.
But AMPs are too expensive for routine oral or intravenous use. So Loose incorporated optimized peptides into coatings for medical devices, which are effective with a small amount of peptide. When bacteria approach a hip implant or catheter coated with the peptides, they “see a bed of nails,” says Loose. The coating doesn’t release the drugs the way typical antibacterial coatings do, so its activity is potentially permanent. Loose founded SteriCoat to commercialize the technology and is currently its chief technology officer; the company is testing coated intravenous lines in animals and hopes to bring them to market in 2011.
Neil Renninger
Hacking microbes for energy.
As a former member of the infamous MIT blackjack team, Neil Renninger knows what it means to make big, calculated risks and see them pay off. Three years ago, he took just such a risk, cofounding synthetic-biology startup Amyris while a postdoc at the University of California, Berkeley. The company’s new approach to biofuels is now generating serious buzz among investors and interest from corporations such as Virgin, which recently opened a fuel division.
Amyris started by commercializing a microbial approach to producing a precursor of artemisinin, a potent malaria drug (see “10 Emerging Technologies: Bacterial Factories,” May 2005). Artemisinin is currently derived from sweet wormwood, but Renninger outlined a way that it could be made more cheaply in bacteria–helping land a share of a $42 million grant from the Gates Foundation. He is also playing a key role in Amyris’s biofuels venture. He began by identifying molecules that would work well as fuels and were compatible with existing engines and delivery infrastructures; then he found a way to combine biological and chemical processes to manufacture them. So far, Amyris has created microbes that can produce candidate replacements for biodiesel, jet fuel, and gasoline. “Now we need to tinker with the bug to squeeze out the last bit of metabolic flux that turns something from interesting to cheap enough to burn,” he says.
Shetal Shah
Cushioning preemies.
As a fellow in neonatology, Shetal Shah spent hundreds of hours jouncing around in ambulances, transporting dangerously ill premature babies to New York University Medical Center’s specialized neonatal unit. “You have a lot of time to think when you’re sitting there,” he says. “I noticed how disruptive these vibrations were to me, and I started thinking, Well, what does it mean for the infant?” Shah, now an assistant professor of pediatrics at SUNY Stony Brook, knew that preemies who have to be transferred between hospitals tend to have more problems than those who don’t–problems that include bleeding in the brain and chronic lung disease. So he set about finding out what role those jolts might play. He adapted an accelerometer, attached it to the head of a neonatal mannequin, and drove around the city in a borrowed ambulance. This gave him approximate measurements of the forces a transported baby experiences every minute. To damp those forces, Shah initially used a free sample of memory foam from a mattress store but eventually developed a patent-pending transport system. Some companies have expressed interest, and the military is studying its potential to help protect soldiers with head trauma.
Doris Tsao
Shedding light on how our brains recognize faces.
Glance at a newsmagazine and you probably recognize the face on the cover right away–Al Gore looking serious in profile, or perhaps a smirking Dick Cheney. But in that instant, your brain performs a lot of complex computations: identifying the object as a face (regardless of size or viewing angle), interpreting its expression, and accessing memory to determine whether it’s familiar.
Little is known about how the brain does all that, but Doris Tsao aims to unravel the process by combining two of the most important tools in neuroscience: brain imaging and electrical recordings from single neurons.
Last year, Tsao used functional magnetic resonance imaging, a technology that indirectly measures brain activity, to identify areas of the monkey brain that are active only when the animals look at faces. She then used a detailed MRI picture to guide an electrode to several of those spots. Using the electrode to record activity from individual neurons, she found that different cells respond to different facial characteristics–say, the shape of the face or the size of the eyes. This exquisite level of detail would have been impossible with imaging alone.
Tsao’s work yields important clues to how neural activity leads to conscious visual perception, says Christof Koch, a neuroscientist at Caltech. “It’s a step toward answering the age-old question ‘What is the relation between the mind and the brain?’”
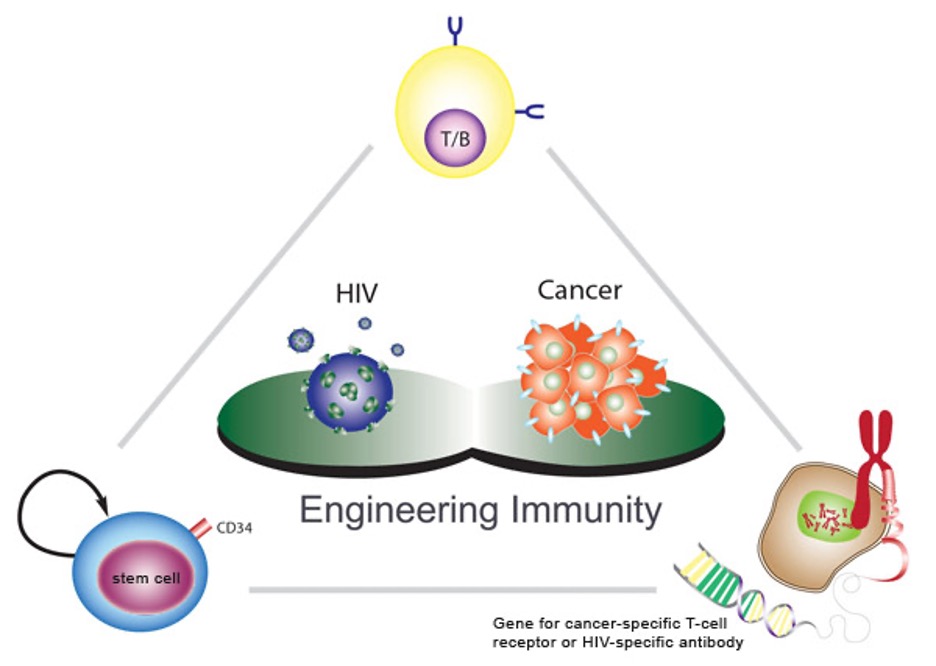
Lili Yang
Engineering immunity.
The immune system is a sophisticated machine, designed to fend off a constant barrage of disease-causing microbes. Unfortunately, it’s not as good at fighting cancer, which disguises itself as normal tissue. Using gene therapy, Lili Yang is reprogramming the immune system to recognize and kill cancer cells.
Stimulating the immune system to fight cancer is one of today’s hottest research areas. Some scientists hope to genetically modify patients’ white blood cells to do the job, but Yang is altering the body’s blood-forming stem cells, a technique that could prove much more powerful. Because stem cells are self-renewing, they could generate a lifelong supply of immune cells programmed to combat, or even prevent, the disease.
A project manager and lead scientist in Caltech’s Engineering Immunity Program, Yang created a viral “vector” that simultaneously delivers two genes to the stem cells: the genes encode the T-cell receptor protein, which enables white blood cells called T cells to recognize and kill cancer cells. The modified stem cells then give rise to T cells that bear the receptor. The technique has so successfully suppressed tumors in mice that Yang plans to begin trials this spring in melanoma patients.
In order to treat patients, Yang will have to isolate and modify their blood-forming stem cells in the lab and then reinject them–a laborious, costly process. So she is collaborating with her husband, Pin Wang of the University of Southern California, to design viral vectors that can deliver therapeutic genes to only one cell type. They have succeeded in mice, a significant advance in gene therapy.
In the future, says Yang, treating cancer could be as simple as injecting patients with such targeted vectors. Gene therapy has yet to live up to its huge potential, but “Lili has the ability to make it a part of modern medicine,” says Caltech biologist and Nobel Prize winner David Baltimore, Yang’s supervisor. Indeed, Yang is also creating vectors to stimulate specific immune cells to make antibodies against HIV. If successful, that project could at last lead to an AIDS vaccine.
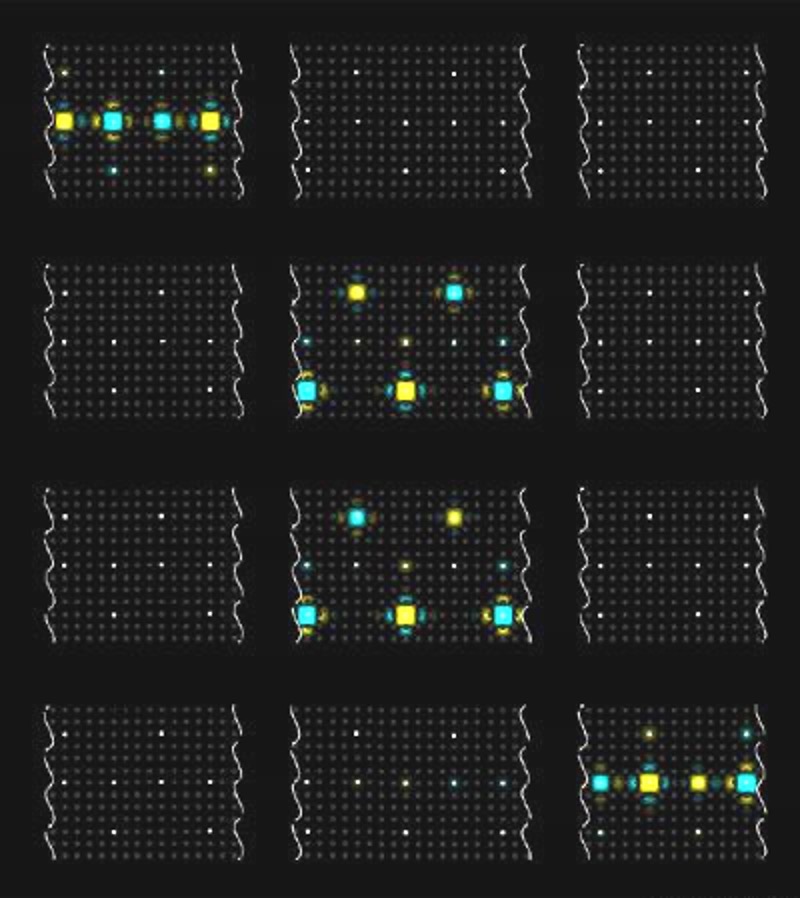
Mehmet Yanik
Stopping light on microchips.
Mehmet Yanik, an assistant professor of electrical engineering and computer science, has invented a way to stop light pulses on a chip and release them at will. The technology could allow engineers to route and store optical data in telecommunications networks and on microchips without having to convert it to electricity.
Yanik’s system traps a pulse of light in an arrangement of microscopic cavities. A fundamental challenge in designing the system was that the faster light can enter the cavities, the faster it escapes, severely limiting how long it can be stored. To overcome this problem, Yanik developed a way to adjust the cavities’ refractive index, a property related to the way a material bends or reflects light. At first, light easily slips into a cavity. Once the light is inside, a small change to the index traps it. Changing the index yet again releases it. The system works quickly, making it ideal for processing data.