Human brain cells transplanted into baby rats’ brains grow and form connections
When lab-grown clumps of human neurons are transplanted into newborn rats, they grow with the animals. The research raises some tricky ethical questions.
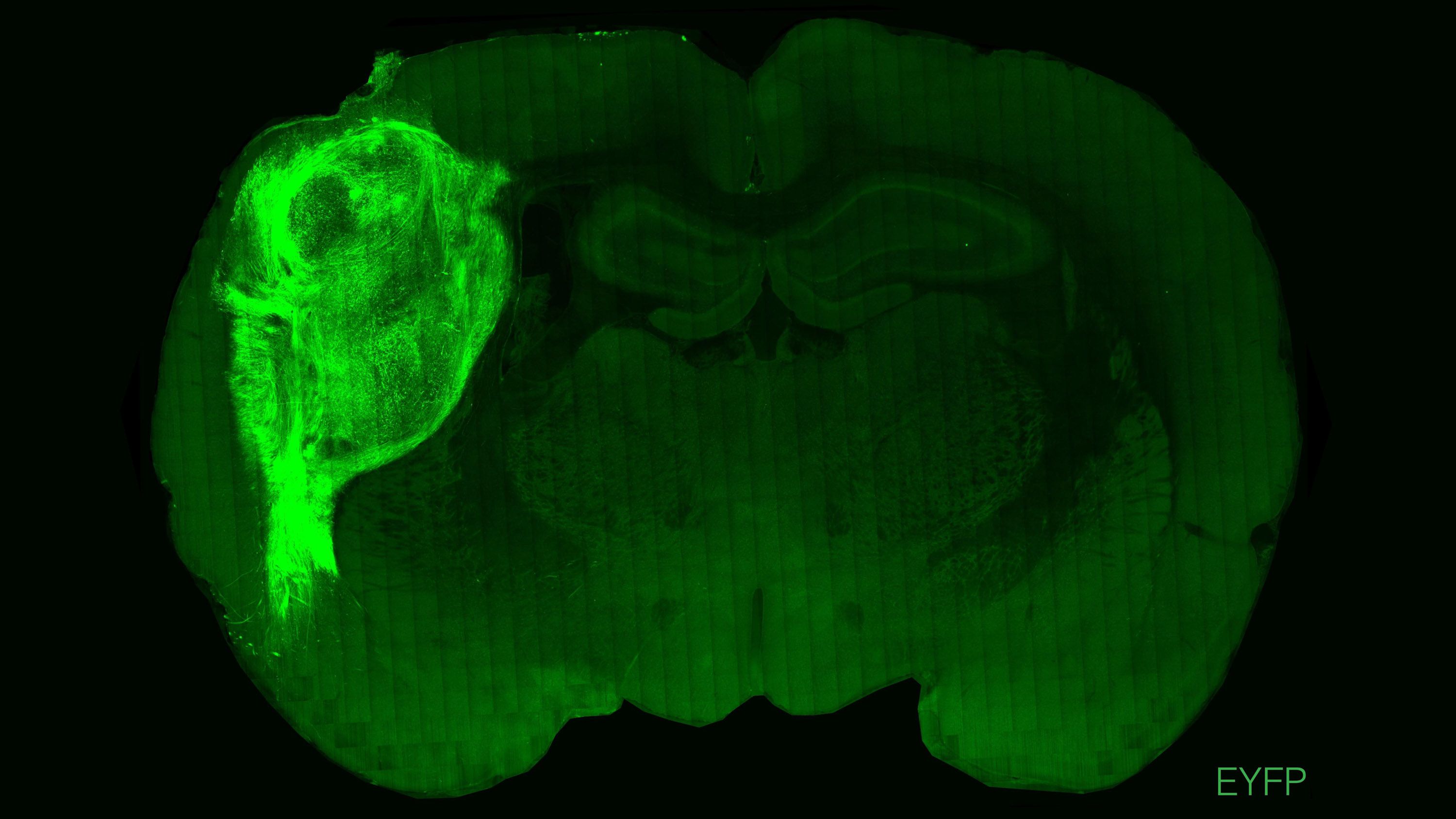
Human neurons transplanted into a rat’s brain continue to grow, forming connections with the animals’ own brain cells and helping guide their behavior, new research has shown.
In a study published in the journal Nature today, lab-grown clumps of human brain cells were transplanted into the brains of newborn rats. They grew and integrated with the rodents’ own neural circuits, eventually making up around one-sixth of their brains. These animals could be used to learn more about human neuropsychiatric disorders, say the researchers behind the work.
“It’s an important step forward in progress into [understanding and treating] brain diseases,” says Julian Savulescu, a bioethicist at the National University of Singapore, who was not involved in the study. But the development also raises ethical questions, he says, particularly surrounding what it means to “humanize” animals.
Sergiu Pașca at the University of Stanford has been working for more than a decade with neural organoids—small clumps of neurons, grown in a dish, that resemble specific brain regions. These organoids are often created from human skin cells, which are first made into stem cells. The stem cells can then be encouraged to form neurons in the lab, under the right conditions. The resulting organoids can be used to study how brain cells fire and communicate—and how they malfunction in some disorders.
But there’s only so much a clump of cells in the lab can tell you. When it comes down to it, these cells don’t really replicate what is happening in our brains—which is why Pașca and many others in the field avoid the commonly used term “mini-brains”. The organoid cells can’t form the same complex connections. They don’t fire in the same way, either. And they aren’t as big as the cells in our brains. “Even when we kept human neurons for hundreds of days … we noticed that human neurons don’t grow to the size to which a human neuron in a human brain would grow,” says Pașca.
It is also impossible to tell how changes to neurons in the lab might lead to symptoms of a neuropsychiatric disorder. If cells in a dish show a change in their shape, the way they fire, or the proteins they make, what does that mean for a person’s memory or behavior, for example?
To get around these issues, Pașca and his colleagues transplanted organoids into the brains of living rats—specifically, newborn rats. The brains of very young animals undergo extensive growth and rewiring as they develop. Neurons transplanted at such an early stage should have the best chance of being integrated with the rats’ own brain circuits, Pașca reasoned.
Building brain organoids
The team used organoids made from skin cells. These cells were made into stem cells in the lab before being encouraged to form layers of cells that resemble those in the human cortex, the folded outer part of the brain that contains regions responsible for thought, vision, hearing, memory, and sensing the environment, among other things. This process took around two months in the lab.
The resulting three-dimensional organoids were then injected into the brains of days-old rats through an incision in the skull. The organoids were transplanted into the sensory cortex, a region that plays a role in helping animals sense their environment.
Within four months, brain scans showed that the organoids had grown to around nine times their original volume—and made up around a third of one brain hemisphere. The cells appeared to have formed connections with rat brain cells and been incorporated into brain circuits.
"It raises the possibility that you’re creating an enhanced rat that might have cognitive capacities greater than an ordinary rat"
Julian Savlescu, bioethicist at the National University of Singapore
The cells themselves were much closer in size to neurons in human brains—six months after being transplanted, they were around six times bigger than those that had been grown in a dish. “They’re just absolutely immense,” says Pașca. Experiments on the cells suggest they fire in a similar way to those in a human brain. “The cells have just moved to another level of maturation,” says Pașca.
Their integration into the rats’ own brain circuits was thorough enough to let them control the animals’ behavior. In another experiment, the team specifically triggered the human cells to fire using optogenetics—a technique that involves shining light on brain cells that have been gene-edited so that they respond to light. By doing this, the researchers were able to influence how often the rats sought out a reward.
“It’s a very important … very cool study,” says Yun Li, a molecular geneticist at the University of Toronto, who was not involved in the research. “The fact that they succeeded in many of these experiments is quite extraordinary.”
Pașca hopes that studying human brain organoids in rats can help researchers understand human diseases. In another experiment, his team created organoids using cells taken from people with Timothy syndrome, a rare genetic disorder that affects the brain and heart. These neurons looked different from healthy ones, and they appeared to work differently too. Pașca’s team is currently testing potential new treatments for the syndrome in these rats.
“[The rat model] gives a lot of information,” says Jeantine Lunshof, a philosopher and ethicist at the Wyss Institute for Biologically Inspired Engineering at Harvard University. “It increases the usefulness of cerebral organoids for human brain disease.”
100% rat?
But are rats with human brain cells still rats? It depends whom you ask. Pașca, Li, and Lunshof believe they are. The animals showed no signs of enhanced cognition or any human-like behaviors, says Pașca. And the human brain is incredibly complex—far more so than a rat brain, even one with a chunk of human cells in it, says Lunshof.
But Savulescu points out that, at the level of cells at least, the rats are no longer fully rat. “The question is: What would be the criteria for a species change?” says Lunshof. There is some agreement that a change in cognition or behavior would be necessary.
Savulescu doesn’t have ethical concerns about this study, because only a small chunk of cells were implanted, and they were placed in a part of the brain that deals with sensing the environment, rather than awareness or consciousness.
But in the future, scientists should be aware of the potential implications of implanting larger organoids in other regions of the brain. “It raises the possibility that you’re creating an enhanced rat that might have cognitive capacities greater than an ordinary rat,” he says.
Deep Dive
Biotechnology and health
How scientists traced a mysterious covid case back to six toilets
When wastewater surveillance turns into a hunt for a single infected individual, the ethics get tricky.
An AI-driven “factory of drugs” claims to have hit a big milestone
Insilico is part of a wave of companies betting on AI as the "next amazing revolution" in biology
The quest to legitimize longevity medicine
Longevity clinics offer a mix of services that largely cater to the wealthy. Now there’s a push to establish their work as a credible medical field.
There is a new most expensive drug in the world. Price tag: $4.25 million
But will the latest gene therapy suffer the curse of the costliest drug?
Stay connected
Get the latest updates from
MIT Technology Review
Discover special offers, top stories, upcoming events, and more.