We’re starting to understand the mysterious surge of hepatitis in children
Lockdowns, genes, and viruses that hide in our cells all appear to have played a role.
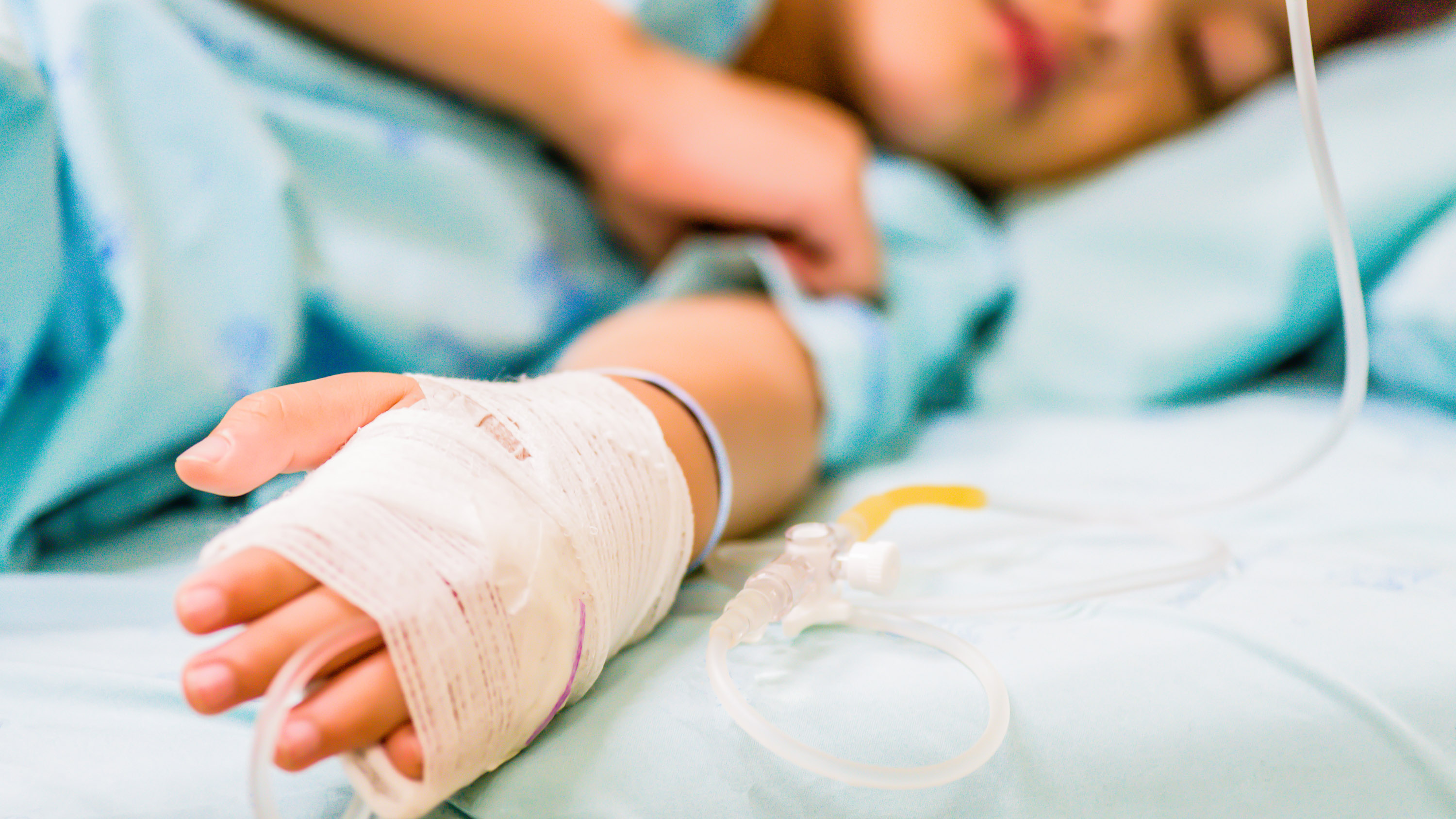
Hundreds of young children around the world have developed severe cases of hepatitis with no obvious cause, leaving doctors baffled. But two new studies reveal the potential culprits: a combination of genetic factors, lockdowns, and at least two viruses working together.
Doctors first noticed a strange cluster of hepatitis cases in young children in Scotland in April this year. By April 8, 60 cases were being investigated across Scotland, England, and Wales, mostly in children aged two to five. As of July 8, the World Health Organization reported 1,010 probable cases of the liver condition in 35 countries—although the true number is likely to be higher, says Antonia Ho, an infectious diseases consultant at the MRC–University of Glasgow Centre for Virus Research, who has been investigating the outbreak. A third of the cases reported so far have been in the US.
These cases are severe—around 5% of the children infected worldwide have needed liver transplants, and 22 have died. And the cause of the outbreak has been something of a mystery. These children don’t have the viruses that usually cause the disease.
Early on, the most obvious suspects were SARS-CoV-2, the virus behind covid-19, and adenovirus, a common virus that often causes cold- and flu-like symptoms. Adenoviruses appeared to surge as lockdowns ended and people began to mingle more, following a period of unusually low transmission.
In an attempt to find out more, Ho, along with Emma Thomson, a professor in infectious diseases at the MRC–University of Glasgow Centre for Virus Research, and their colleagues, have been carefully studying some of the affected children. In a recent study that's yet to be peer reviewed, the team assessed nine children in Scotland with the mystery hepatitis, and compared them with 58 children who did not have the condition.
The team studied blood, liver, and fecal samples taken from the children, as well as throat and nose swabs. While they were unable to find the viruses that usually cause hepatitis, they did find adenovirus in samples from six of the nine children.
The team also found another virus called adeno-associated virus, or AAV-2. This virus was found in samples from all nine children who had the unexplained hepatitis—but was not found in any of the children who did not.
This virus is known to infect most people by the time they are 10, and most people start developing antibodies for it around the age of three. But it has never before been directly linked to human disease, says Thomson.
The virus is unusual in that it relies on other viruses to be able to replicate and make copies of itself. “In this case we think the helper virus is the adenovirus,” Thomson told journalists at a virtual press briefing today. It’s possible that the adenovirus infection followed an AAV2 infection, or that both viruses hit at the same time, she added. “We can’t tell you at the moment which of these viruses is causing the condition,” Thomson said.
But the viruses aren’t the end of the story. In genetic tests, the team noticed that the children with unexplained hepatitis were much more likely to have a gene called DRB1*0401—89% of the affected children had this gene, which is generally found in 16% of the Scottish population. The gene is known to affect the way the immune system works. Essentially, the proteins it codes for help immune cells decide what to destroy.
In another study published today, also awaiting peer review, researcher Sofia Morfopoulou at the UCL Great Ormond Street Institute of Child Health in London and her colleagues performed genetic analysis on samples from 28 children, five of whom had liver transplants.
“Our results absolutely match those of the Scottish group,” coauthor Judy Breuer, also at the UCL GOSH ICH, told journalists at the same briefing. Across both studies, AAV2 was found in 96% of children with unexplained hepatitis but in only 4% of those who didn’t have the condition, Breuer said.
Curiously, while the team found viral genes and evidence that the virus was replicating, they could not find particles of the virus itself. This could mean that the virus is having an indirect effect on the children, and strengthens the case for the role of the children’s own immune system in making them unwell.
“We think that AAV2 and, or together with, human adenovirus may trigger unexplained pediatric hepatitis in children who are susceptible because of [the immune gene],” Breuer said.
The team also looked for SARS-CoV-2. But none of the children had active covid-19 infections. Some had antibodies to the virus, suggesting they had covid-19 in the past—but only at the same rate as the rest of the Scottish population. This, alongside the fact that the hepatitis outbreak occurred two years after the pandemic began, probably rules out covid-19 as a cause, the researchers say.
But the covid-19 pandemic might have indirectly played a part in the outbreak of unexplained hepatitis. Measures to reduce social contact, such as lockdowns, helped reduce the spread of covid—but they also had an impact on the spread of other infectious diseases. Cases of, and deaths from, influenza dropped precipitously in the US and elsewhere, for example.
The sudden surge in adenovirus cases could be the result of children finally returning to school and mingling with each other as they did before. “There’s inevitably a kind of equilibrium that needs to be reestablished,” said Ho. “Lots of seasonal viruses are circulating outside of what we’d normally expect them to.”
Thomson stresses that the puzzle is not yet entirely solved. The two studies were small, and neither has yet been published in a scientific journal. But it’s the clearest picture we’ve had so far. Thankfully, cases of the mysterious condition appear to be declining, although the WHO warns that the organization doesn’t have complete and up-to-date information from all countries.
The researchers think it’s likely that similar cases of hepatitis have been “trickling along” for years, but that they’ve only become obvious now thanks to the outbreak. “Unless you see a cluster [of cases], you don’t investigate in quite the same way,” said Breuer.
Deep Dive
Biotechnology and health
How scientists traced a mysterious covid case back to six toilets
When wastewater surveillance turns into a hunt for a single infected individual, the ethics get tricky.
An AI-driven “factory of drugs” claims to have hit a big milestone
Insilico is part of a wave of companies betting on AI as the "next amazing revolution" in biology
The quest to legitimize longevity medicine
Longevity clinics offer a mix of services that largely cater to the wealthy. Now there’s a push to establish their work as a credible medical field.
There is a new most expensive drug in the world. Price tag: $4.25 million
But will the latest gene therapy suffer the curse of the costliest drug?
Stay connected
Get the latest updates from
MIT Technology Review
Discover special offers, top stories, upcoming events, and more.