The race to find covid-19 drug treatments that actually work
We have a bounty of vaccines and more on the way, but drugs that treat the disease are vital too if we want to keep people alive and bring the pandemic under control.
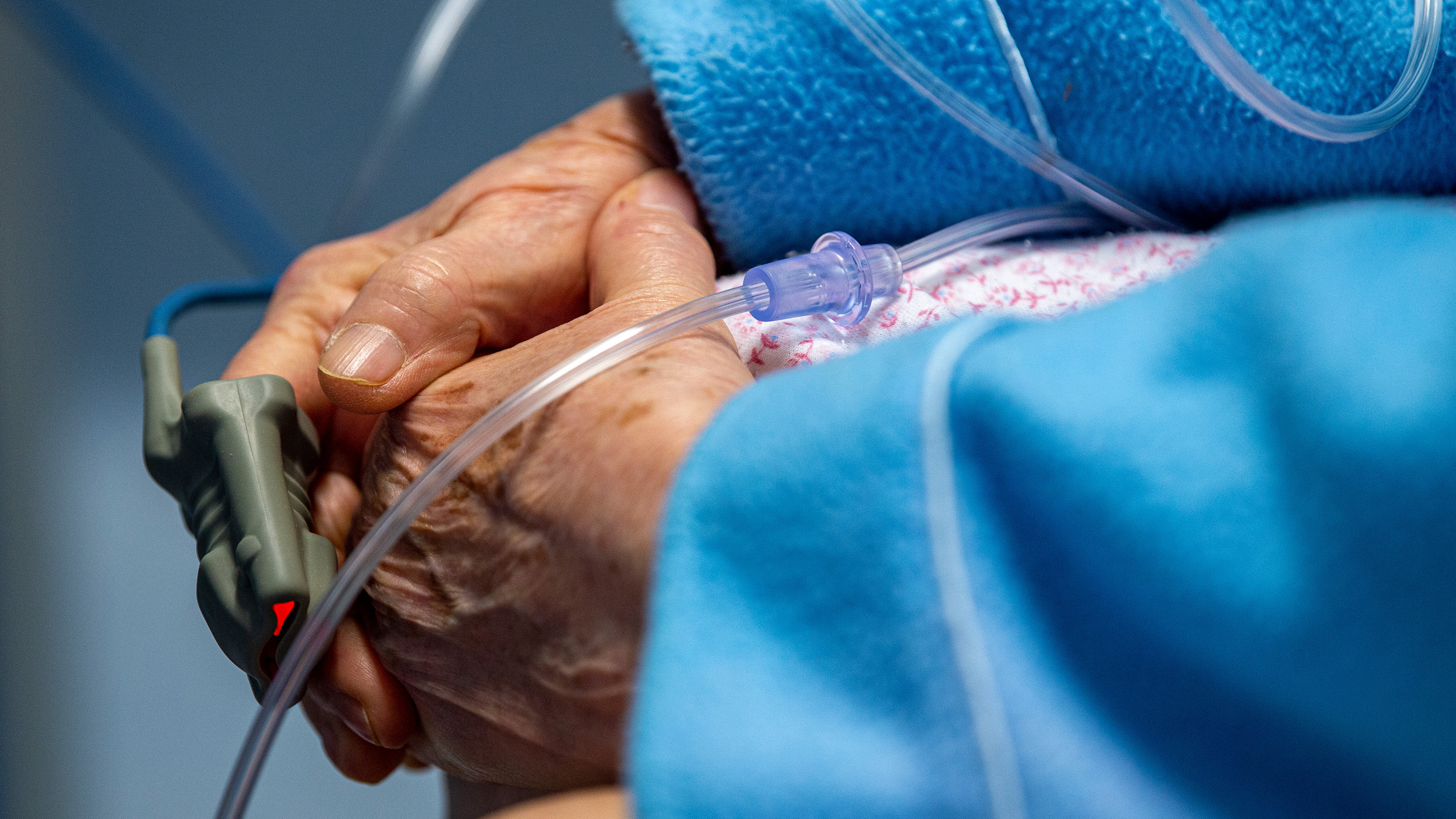
The global effort to develop vaccines against covid-19 has been a scientific triumph. The search for new therapies, however, has had far less success.
More than a year and a half into the pandemic, few treatment options for covid-19 exist, and those that are available seem to have only modest impact on the course of the disease.
Vaccination rates may be high enough in some wealthy countries to put herd immunity within reach, but the need for new therapies is still urgent. The virus is raging in some countries with limited access to vaccines. And even in places where they are widely available, pockets of unvaccinated people remain. “The need for treatments for all stages of the disease is, in fact, more pronounced than ever,” says Rachel Cohen, North American executive director of the Drugs for Neglected Diseases Initiative (DNDi), a program aimed at developing new medicines for diseases that have historically been ignored by pharmaceutical companies. “Never in the history of infectious-disease control have we been satisfied with just one set of tools.”
New drugs might help people with early cases of covid avoid hospitalization, “especially in places where ICU and hospitalization capacity is very limited,” Cohen says. They might also prevent those who are hospitalized from dying.
New therapies could also be a crucial stopgap if SARS-CoV2 mutates enough to evade the immune response in people who are vaccinated. And there’s another reason to keep hunting for new medications. SARS-CoV2 is the third coronavirus to make the jump from animals to humans in the last 20 years. “This may not be the last coronavirus that we see,” says Michael Diamond, a virologist at the Washington University School of Medicine.
The Biden administration hopes to help stock the medicine cabinet. In mid-June, officials announced that the federal government would put $3.2 billion toward antiviral drug discovery and development for treatments targeting covid-19 and future pandemic viruses. That’s just a fraction of the more than $10 billion the Trump administration spent on vaccine development with Operation Warp Speed, but it’s still “massive,” Cohen says. A number of other initiatives to bolster antiviral development are under way as well.
Sparse arsenal
Only a handful of therapies exist to treat covid-19. For the sickest patients, studies have shown that the steroid dexamethasone reduces the risk of death by a third by dampening the runaway inflammatory response.
Other therapies target the virus itself. A few companies have received emergency-use authorization for monoclonal antibodies. These lab-made antibodies work like natural ones: they bind to the virus and block it from infecting cells. When given early, monoclonal antibodies or combinations of these therapies reduced hospitalization or death by 70% to 87% in individuals at high risk of severe symptoms or hospitalization. These medicines work best in patients who aren’t yet seriously ill. However, Regeneron’s antibody combination—the therapy former president Donald Trump received—also seems to help people who are already hospitalized but have not generated their own antibodies. In mid-June, researchers announced that in a 9,000-person trial, it curbed deaths in this group by 20%. That benefit did not extend to people who had natural antibodies.
But antibody therapies have drawbacks. They’re expensive and must be given via infusion or injection. That makes them poor options for many low- and middle-income countries. And they may not perform as well against some circulating variants. In fact, on June 25 the FDA paused distribution of Lilly’s antibody cocktail nationwide because of the increasing prevalence of two variants of concern that don’t seem to respond to the medicines.
When it comes to antiviral drugs, which interrupt the virus’s ability to replicate, even fewer options are available. Remdesivir is the only such medication approved to treat covid-19, in large part because it was one of the few candidates that had been tested for safety in humans when the pandemic hit, so it had a head start. But just how well it works is still an open question. Some studies have found it to shorten the length of the illness, while others suggest it has little impact. The World Health Organization does not recommend its use.
Tricky therapies
Antiviral development has lagged for a variety of reasons. Until covid-19, companies didn’t have much of a financial incentive to produce these drugs. The antivirals that do exist target just 10 viruses, and half of them treat HIV. Chronic infections require lengthier treatments and thus make more money. “If there is not an obvious market for a therapeutic, then generally speaking, they’re not going to invest in those types of therapeutics,” says John Bamforth, interim executive director of READDI, a public-private partnership at the University of North Carolina at Chapel Hill founded to develop novel antivirals.
There are also a number of scientific hurdles. To inhibit replication, a drug has bind to some essential viral protein or enzyme and block its activity without harming the host cell. But unlike bacteria, viruses rely on the machinery inside the cells they inhabit to copy themselves, so they have few proteins of their own. And even when researchers do come across a compound that works, its effectiveness tends to be short-lived because viruses are constantly evolving.
Some researchers, including those at READDI, are working on medicines that target cellular proteins crucial for viral replication. Most antivirals work on only a single virus. The hope is that these compounds would be effective against entire families of them. They may also be less likely to drive resistance.
But novel therapies take more time to develop. That’s why the quickest way to get drugs on the shelves is to repurpose compounds that have already been approved. They have been tested for safety, and there are fewer regulatory hurdles to getting a new use approved for an existing drug. DNDi is testing a variety of existing compounds in a clinical trial called ANTI-COV. The latest study looks at the anti-parasite medicine nitazoxanide combined with an inhaled steroid. “The consensus that is emerging is that you would need a strong antiviral or a combination of antivirals with different mechanisms of action, combined with some kind of anti-inflammatory,” Cohen says.
Biden’s Antiviral Program for Pandemics aims to speed up testing of 19 antivirals that are already in development. It will also provide about a billion dollars to create a drug discovery program to search for compounds that can treat not only SARS-CoV-2 but other viruses as well. The administration also announced that it will purchase up to 1.7 million doses of Merck’s antiviral molnupiravir for $1.2 billion, pending regulatory authorization. Researchers once envisioned this drug as a flu treatment, but when the pandemic hit, they had to pivot. The compound has already made it through phase 2 testing for covid-19.
More than 20 biotech and pharma companies have banded together to form the INTREPID alliance to deliver 25 novel, late-stage drug candidates targeting viral pathogens that pose the greatest pandemic risks, including coronaviruses. And the COVID Moonshot is an international consortium of scientists from academia, biotech, and pharma who work pro bono or at cost to develop drugs to inhibit one particular enzyme in SARS-CoV-2. The project relies on crowdfunding and crowdsourcing. Anyone can submit a drug design and view the ones that have already been submitted. As of June 28, the project had collected 17,976 molecule designs and synthesized and tested nearly 1,500.
The payoff for all these new initiatives might not come for years. But the hope is that when the next pandemic hits, we will be better prepared. “We have to stop this cycle of neglect and then panic,” Cohen says.
Unequal access
Vast inequities have emerged in the rollout of covid vaccines. In the United States and the United Kingdom, more than 45% of the population is vaccinated. In the Democratic Republic of the Congo and Chad, fewer than 0.1% of people have gotten the jab. And where vaccine coverage is low, the virus can surge. “We’re seeing spiraling crises in the Indian subcontinent, in Latin America, and over the last few weeks in Africa,” Cohen says. These are the places in desperate need of new therapies, but she worries that any new treatments developed won’t reach these countries.
It’s something that READDI has been thinking about. “If we were to license a drug, we would probably put within the contract some language around global access,” Bamforth says. “We need to make sure that these drugs are accessible to all the countries in the world, not just in the First World that can afford them”
Deep Dive
Biotechnology and health
How scientists traced a mysterious covid case back to six toilets
When wastewater surveillance turns into a hunt for a single infected individual, the ethics get tricky.
An AI-driven “factory of drugs” claims to have hit a big milestone
Insilico is part of a wave of companies betting on AI as the "next amazing revolution" in biology
The quest to legitimize longevity medicine
Longevity clinics offer a mix of services that largely cater to the wealthy. Now there’s a push to establish their work as a credible medical field.
There is a new most expensive drug in the world. Price tag: $4.25 million
But will the latest gene therapy suffer the curse of the costliest drug?
Stay connected
Get the latest updates from
MIT Technology Review
Discover special offers, top stories, upcoming events, and more.