Novel lithium-metal batteries will drive the switch to electric cars
A new type of battery could finally make electric cars as convenient and cheap as gas ones.

Why it matters:The performance limitations of batteries have held back the switch to cleaner electric cars and all but ruled out electric planes.
Key players:• QuantumScape
• Samsung Advanced Institute of Technology
• Solid Power
• 24M
Availability:2025
For all the hype and hope around electric vehicles, they still make up only about 2% of new car sales in the US and just a little more globally.
For many buyers, they’re simply too expensive, their range is too limited, and charging them isn’t nearly as quick and convenient as refueling at the pump.
All these limitations have to do with the lithium-ion batteries that power the vehicles. They’re costly, heavy, and quick to run out of juice. To make matters worse, the batteries rely on liquid electrolytes that can burst into flames during collisions.
Making electric cars more competitive with gas-powered ones will require a breakthrough battery that remedies those shortcomings. That, at least, is the argument of Jagdeep Singh, chief executive of QuantumScape, a Silicon Valley startup that claims to have developed just such a technology.
The company asserts it did so by solving a chemistry puzzle that has stumped researchers for nearly half a century: how to use lithium, the lightest metal on the periodic table, to boost the amount of energy that can be packed into a battery without posing a routine risk of fire or otherwise sacrificing performance. The company says it achieved this, in large part, by developing a solid version of the flammable liquid electrolyte.
VW was impressed enough to invest hundreds of millions of dollars in QuantumScape. The German auto giant also agreed to set up a joint venture with the company to mass-produce the batteries and says they’ll be in its electric cars and trucks on the road by 2025.
Faster charging and longer range
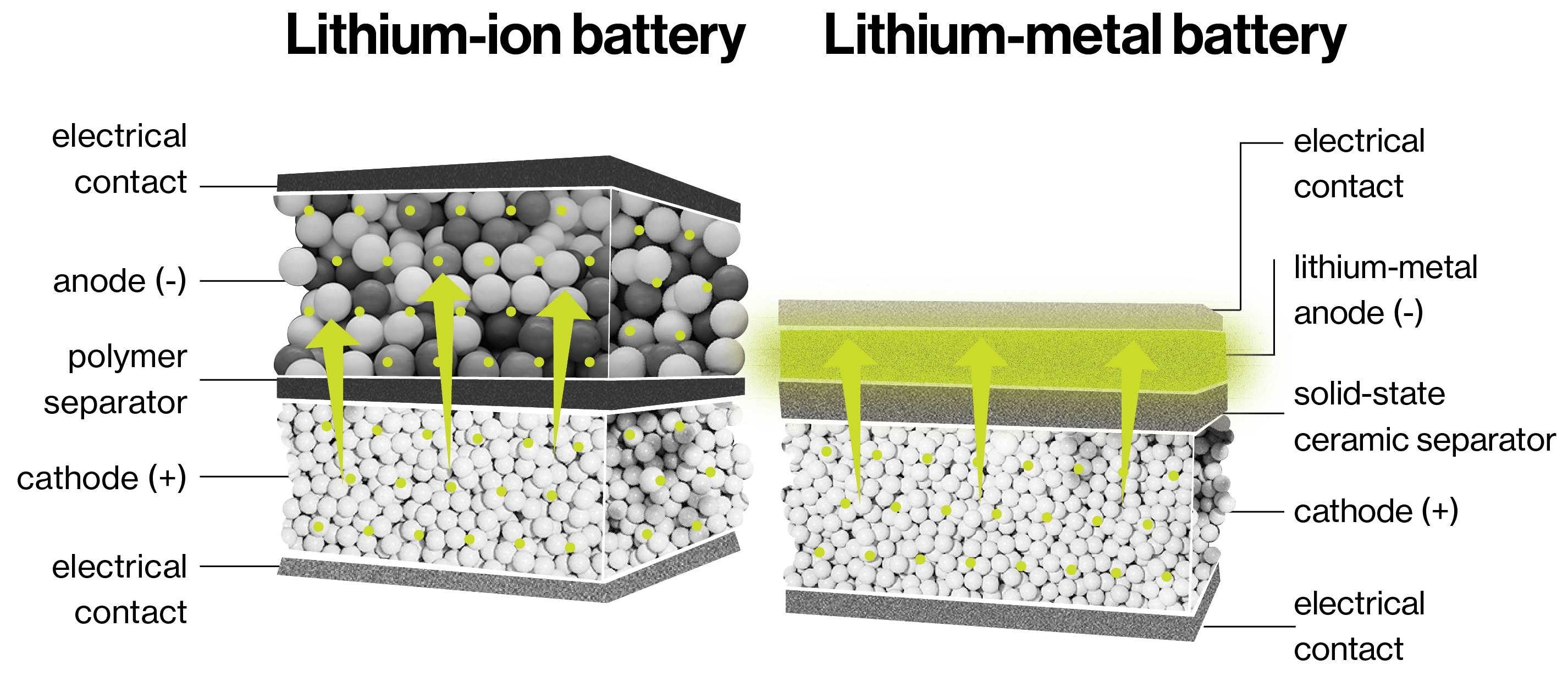
In a conventional lithium-ion battery, one of the two electrodes, the anode, is made mostly from graphite. This is a form of carbon that can easily take up and release the charged lithium ions that shuttle back and forth between the anode and cathode through the electrolyte. That stream of charged particles produces an electric current, which flows out of the battery to power whatever needs powering. But the graphite is merely a host for the lithium ions, which nestle in between sheets of carbon like packages on shelves. It’s dead weight that doesn’t store energy or produce a current itself.
In a lithium-metal battery, the anode itself is made from lithium. This means that nearly every atom in the battery’s anode can also be put to work creating current. Theoretically, a lithium-metal anode-based battery could store 50% more energy than one of the same weight and volume that relied on graphite.
However, because lithium metal is so reactive, being in constant contact with a liquid electrolyte can trigger reactions that degrade the battery or cause it to combust, says Venkat Viswanathan, an associate professor at Carnegie Mellon who works on lithium-metal batteries and is a consultant for QuantumScape. Another issue is that as the lithium ions flow back and forth, needle-like structures known as dendrites can form in the batteries and short-circuit the cell or cause it to catch fire.

QuantumScape, which went public in November after operating in stealth mode for a decade, is still holding back some of the critical details on how its solid-electrolyte battery overcomes these problems. But it appears to perform remarkably well.
In an online presentation in December, the startup displayed a series of charts showing that a single-layer lab version of the battery can be charged to more than 80% of its capacity in 15 minutes, lasts for hundreds of thousands of miles, and works fine at freezing temperatures. The company expects the batteries to be able to boost electric vehicles’ range by more than 80%: a car that can go 250 miles on a single charge today could drive 450 miles instead.
“QuantumScape has set me back on my heels,” says Nancy Dudney, a battery researcher at Oak Ridge National Laboratory, who has done pioneering work on solid-state electrolytes. “At first view, it looks really good,” she says, though she adds, “We’ve been here before with other battery advances.”
Indeed, the battery field is littered with examples of startups that promised breakthrough technologies but ultimately failed. And the challenges ahead of QuantumScape are daunting, particularly when it comes to converting its prototype cells into commercial products that can be manufactured cheaply.
If the company succeeds, it could transform the EV marketplace. Cutting costs, boosting range, and making charging nearly as convenient as filling up at a gas station could broaden demand beyond people who can afford to shell out thousands of dollars for charging ports at home, and ease the anxieties of those who fear being stranded on longer trips.
The added energy density and faster charging could also make it more practical to electrify other forms of transportation, including long-haul trucking and even short-distance flights. (As a bonus, it would also deliver phones and laptops that could last a couple of days on one charge.)
Birth of a battery
The story of lithium-metal batteries began in the early 1970s and is tightly intertwined with the development of the lithium-ion ones we depend on today.
The oil crises of the era, coupled with what would turn out to be very early peak-petroleum fears, suddenly reignited an interest in electric vehicles for the first time since the infancy of the auto industry. By 1972, American Motors, Chrysler, Ford, GM, Toyota, VW, and others were all working on electric cars, as the science writer Seth Fletcher describes in the book Bottled Lighting. Meanwhile, large industrial labs, including those at GE, Dow Chemical, and Exxon, were searching for better battery chemistries.
Batteries back then, which were mostly lead-acid, couldn’t deliver anywhere near the distances or speeds of gas engines. In 1969, General Motors’ experimental 512 electric car boasted a top speed of about 30 miles an hour, with a range of 47 miles.
In 1972, Exxon’s research division hired a young chemist named Stan Whittingham on the strength of his postdoctoral work at Stanford. Specifically, he was developing crystalline materials that allowed ions to easily flow in and out. At Exxon, Whittingham and his colleagues began experimenting with a promising porous material for a cathode: titanium disulfide. They paired it with an anode made from metallic lithium, a highly reactive material that readily releases its electrons. It worked surprisingly well.
The team applied for a patent in 1973, published a landmark paper in Science in 1976, and showed off a larger version of the cells at an auto show in 1977.
By the early 1980s, the oil crisis had passed. Exxon’s new management decided to shed any business line without the potential to become a $100 million annual market. The company dropped its electric-vehicle and battery efforts. “They said, ‘These are too small for us to be involved in,’” says Whittingham.
Lithium-ion takes over
Lithium-metal batteries were far superior to lead-acid batteries, but they also had inherent drawbacks the Exxon team had never resolved, including their habit of sparking fires in the lab.
Others who attempted to commercialize lithium-metal batteries ran into similar problems. In the 1980s, Moli Energy of British Columbia developed a 2.2-volt lithium-metal battery for laptops and cell phones. But in 1989, a Japanese cell phone caught fire, burning its owner. After an investigation pinned the blame on the battery, thousands of cell phones were recalled and the company went into receivership, according to Electric Autonomy Canada.
Meanwhile, others were building on Whittingham’s work. John Goodenough, now a professor at the University of Texas at Austin, used cobalt oxide rather than titanium disulfide to develop a cathode that could store more energy. Akira Yoshino, a professor at Meijo University, swapped the pure lithium anode for coke (another form of carbon), which could still store a lot of lithium ions but reduced the fire dangers. Finally, researchers at Sony assembled the pieces to develop the first commercial lithium-ion batteries in 1992. Whittingham, Goodenough, and Yoshino shared the Nobel Prize in chemistry in 2019 for their roles in the breakthrough.
The runaway success of lithium-ion batteries, which now power our laptops, phones, and electric vehicles, quashed efforts to commercialize lithium-metal technology for years to come. But some never lost sight of lithium-metal’s potential to be a more efficient form of energy storage. And replacing the standard liquid electrolytes, which are effectively combustible solvents, with solid materials seemed a particularly promising avenue of exploration.
Around 2000, a team at Oak Ridge National Laboratory demonstrated thin-film batteries—the kind deployed in small electronics like smart cards and pacemakers—that used solid-state lithium-metal technology. The production process and size and shape of thin-film batteries mostly limit their use beyond anything larger than a watch, says Paul Albertus, a battery expert at the University of Maryland. But the work provided a crucial proof of concept for a working lithium-metal battery.
Road kill
Various startups had begun pursuing the technology again by the late 2000s. But it has proved to be a treacherous road.
Some have already closed down. Seeo, formed in 2007, was bought by the German company Bosch, which later disbanded its battery research efforts. France-based Bolloré was the first to put solid-state lithium-metal batteries into vehicles on the road, launching its Bluecar car-sharing programs in 2011. But its polymer-based electrolytes only work at higher temperatures, limiting their use in consumer vehicles.

A handful of other companies, however, have made more recent advances. Most notably, two days after QuantumScape’s presentation last December, Solid Power, a Colorado startup founded in 2012, announced that it is already producing pilot-scale batches of 22-layer lithium-metal cells that would surpass the range of today’s electric-vehicle batteries.
And in January, the Department of Energy’s ARPA-E division announced it would invest $9 million into an effort by battery company 24M and Carnegie Mellon’s Viswanathan to develop lithium-metal batteries designed for electric planes, where the energy stored and power delivered per kilogram are crucial.
Starting up QuantumScape
The trick for any company developing lithium-metal batteries has been to pinpoint electrolyte materials that prevent fires and dendrites while still allowing ions to easily pass through, and without otherwise degrading the performance of the battery. And that’s precisely what QuantumScape claims it has done.
The origins of the company date back to 2009. As Singh was preparing to step down as CEO of Infinera, a networking company he cofounded, he began talking with Stanford postdoctoral fellow Tim Holme and his advisor, Friedrich Prinz, about forming a company based on their research on novel battery materials.
The trio cofounded QuantumScape the following year, aiming to develop energy-dense batteries with high power output. They first tried to do so by creating an entirely new type of battery, known as an all-electron battery, but found it would be harder than it initially seemed.
By then, the company had raised tens of millions of dollars from venture capital firms like Kleiner Perkins and Khosla Ventures. That left QuantumScape with enough money to quietly shift direction, pursuing the dream of lithium-metal technology.
The company spent the next five years looking for just the right material to develop a solid-state electrolyte, Singh says. It then spent another five working out the right composition and manufacturing process to prevent defects and dendrites. All the company will say about its electrolyte is that it’s a ceramic.
Are we there yet?
All of QuantumScape’s published tests so far were performed on single-layer cells. (After this piece went to press, the company announced they've produced and conducted tests on 4-layer cells that achieved similar results.) To work in cars, the company will need to produce batteries packed with several dozen layers, effectively moving from a single playing card to a deck. And it will still have to find a way to manufacture these cells cheaply enough to compete with lithium-ion, a battery technology that’s dominated for decades.
It’s a daunting engineering task. “They’re partway there—after 10 years and $300 million and 150 people working on this, they have this little playing card now,” says Albertus, from the University of Maryland. “That’s a long way away still from delivering batteries on the thousands-of-metric-tons scale—and it’s a really hard challenge.” Several battery researchers told me they seriously doubt that QuantumScape can scale up and complete full safety tests in time to put batteries in cars on the road just four years from now.
Given the company’s results and the encouraging announcements from other startups, most people in the battery world do think it’s looking more likely that the problems that have held up lithium-metal for decades can be solved—which is why it’s on MIT Technology Review’s list of breakthrough technologies this year. But it’s also clear that for all the progress that’s been made since Whittingham’s time at Exxon, years of work still lie ahead.
Update: This story was updated to correct an error concerning how much additional energy a lithium-metal battery can theoretically store.
Deep Dive
Climate change and energy
The problem with plug-in hybrids? Their drivers.
Plug-in hybrids are often sold as a transition to EVs, but new data from Europe shows we’re still underestimating the emissions they produce.
Harvard has halted its long-planned atmospheric geoengineering experiment
The decision follows years of controversy and the departure of one of the program’s key researchers.
Why hydrogen is losing the race to power cleaner cars
Batteries are dominating zero-emissions vehicles, and the fuel has better uses elsewhere.
Decarbonizing production of energy is a quick win
Clean technologies, including carbon management platforms, enable the global energy industry to play a crucial role in the transition to net zero.
Stay connected
Get the latest updates from
MIT Technology Review
Discover special offers, top stories, upcoming events, and more.