A cheap steroid might be the first covid-19 drug to save lives
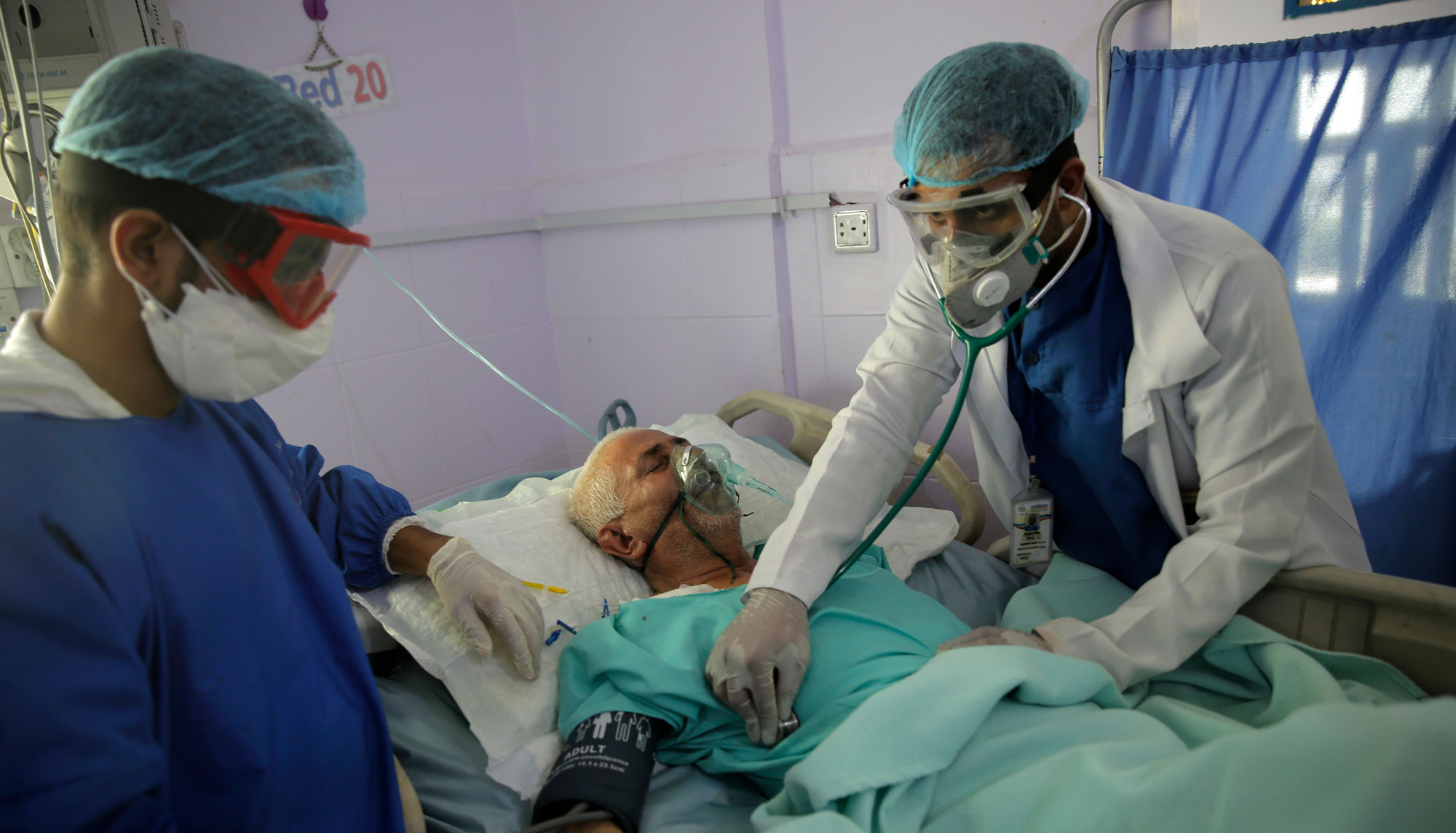
The news: A cheap steroid called dexamethasone significantly cuts the chances that seriously ill covid-19 patients will die, according to the world’s biggest trial of existing treatments. Specifically, it reduced the risk of death for patients on ventilators by a third. For those receiving oxygen, it cut the risk of death by a fifth. The drug is one of six preexisting treatments currently being trialed in coronavirus patients as part of a study led by the University of Oxford.
The details: Researchers enrolled 6,412 patients in the study. They selected 2,104 to receive 6 milligrams of dexamethasone every day for 10 days, as either an injection or a pill, while the remaining 4,321 patients received the usual care alone. While the drug significantly cut the number of deaths among patients on ventilators and those receiving oxygen, there was no benefit to patients who didn’t need respiratory support, who make up the vast majority of people with covid-19. In particular, dexamethasone seems to stop some of the damage that can occur when patients’ immune systems go into overdrive to try to fight off covid-19, a phenomenon known as a cytokine storm.
Good news: The researchers have only released a statement thus far. The paper hasn’t been published yet, and further studies would help confirm the validity of the findings. That said, this discovery could save countless lives. And even better, dexamethasone is readily available and very inexpensive—about $6 per dose. It was first approved as a treatment for inflammation in 1961 and is regularly prescribed to asthmatics.
Other options: The other drug that has shown some promising results is the antiviral remdesivir, which has shown to reduce hospital stays, but has not yet been shown conclusively to reduce the death rate for patients. Remdesivir is also made by pharmaceutical company Gilead and will certainly be sold for many times more than dexamethasone when it comes to market.
The significance: This study finding underlines the value of testing old treatments for covid-19, as opposed to relying on new discoveries alone. “The survival benefit is clear and large in those patients who are sick enough to require oxygen treatment, so dexamethasone should now become standard of care in these patients,” said Peter Horby, professor of emerging infectious diseases at the University of Oxford and one of the chief investigators for the trial.
“This is a major breakthrough: dexamethasone is the first and only drug that has made a significant difference to patient mortality for covid-19,” said Nick Cammack, a researcher at Wellcome, a pharmaceutical research nonprofit, in a statement. However, he cautioned that “to end this pandemic, we still need better diagnostics to detect, medicines to treat, and vaccines to prevent covid-19.”
Deep Dive
Biotechnology and health
How scientists traced a mysterious covid case back to six toilets
When wastewater surveillance turns into a hunt for a single infected individual, the ethics get tricky.
An AI-driven “factory of drugs” claims to have hit a big milestone
Insilico is part of a wave of companies betting on AI as the "next amazing revolution" in biology
The quest to legitimize longevity medicine
Longevity clinics offer a mix of services that largely cater to the wealthy. Now there’s a push to establish their work as a credible medical field.
There is a new most expensive drug in the world. Price tag: $4.25 million
But will the latest gene therapy suffer the curse of the costliest drug?
Stay connected
Get the latest updates from
MIT Technology Review
Discover special offers, top stories, upcoming events, and more.