Meet the Test-Tube Turkey That Costs $34,000

This Thanksgiving, Paul Mozdziak will be giving thanks that people are finally paying attention to his big idea.
He wants to grow turkey meat in 5,000-gallon tanks.
Mozdziak is an expert in growing avian muscle cells in a lab flask. That obscure corner of research recently landed the North Carolina State University professor of poultry science at the cutting-edge of “cellular agriculture,” or the idea that animal protein could be manufactured in bioreactors rather than by animals.
The technology, also known as in vitro meat cultivation, may sound strange. But it has been drawing a following of environmentalists, animal-rights activists, and investors who think meat can be made by biotech companies rather than on farms.
“Years from now, when people are [in] the grocery store trying to decide if they want to buy traditional versus cultivated meat, I am 100 percent sure that cultured meat is going to be just as cheap, if not cheaper,” says Mozdziak.
The idea of cultured meat took flight in 2013, when Dutch scientist Mark J. Post went on British TV and cooked and bit into the first lab-grown hamburger. The experiment cost more than $300,000 and was paid for by Google founder Sergey Brin.
Proponents say in vitro meat could reduce reliance on farm animals and save resources. This summer, Mozdziak was a featured panelist at a conference put on by New Harvest, a foundation that promotes “animal products without animals.” The San Francisco event featured innovators presenting their versions of lab-grown beef, gelatin, egg whites, and milk.

Mozdziak’s research begins with a pencil-eraser-size biopsy of a turkey breast. Then the job is to isolate stem cells known as satellite cells, which multiply and fuse to build up existing muscle fibers. By manipulating these prolific cells in a warm broth of glucose and amino acids, Mozdziak essentially tricks them into behaving as if they are still inside a turkey.
In theory, the growth potential is enormous. Assuming unlimited nutrients and room to grow, a single satellite cell can undergo 75 generations of division during three months. That means one cell could turn into enough muscle to manufacture over 20 trillion turkey nuggets. Surveys suggest about half of vegetarians would eat meat if it came from a lab.
“Muscle to me is the most fascinating tissue and cell type that exists,” says Mozdziak, who earned his PhD studying satellite cells. “Let’s just put it this way—I find a lot of beauty in turkeys.”
Lab-grown meat is still far from being economical. In Mozdziak’s lab, his team grows cells as a thin layer inside plastic flasks. If the cells become too thick, nutrients can’t get in. Growing a turkey-size amount of white meat this way would require about 11,340 flasks and about $34,000 worth of growth serum.
Hultz Smith, a scientist charged with studying long-term innovation for Tyson Foods, the Arkansas food processing giant, says his company is keeping an eye on cultured meat, but isn’t yet ready to invest. He thinks scientists are still far away from making a dent in the $675 billion global meat trade.
At the New Harvest conference, Smith asked the audience of cultured meat impresarios how many believed they were ready now to scale up their research into a real business. “Crickets,” says Smith. “Not one hand raised.”
One company, Memphis Meats, has turned cattle stem cells into ground beef, but so far at a cost of $18,000 a pound. Another, Modern Meadow, dropped its highly publicized plans for laboratory beef chips and instead is working on synthesizing leather, a far more valuable commodity.
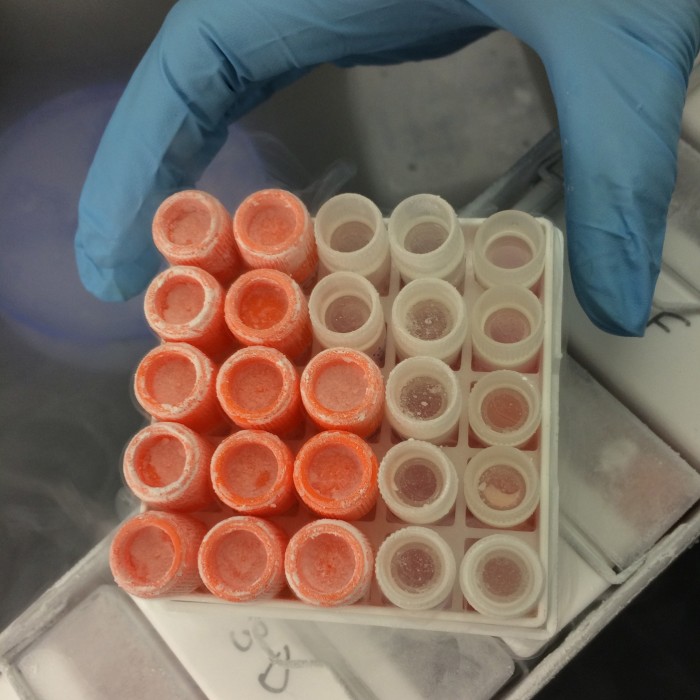

Scientists at North Carolina State University are working on manufacturing meat in the lab. Turkey stem cells are stored in liquid nitrogen and later transferred to plastic flasks, where they are fed with warm nutrients and allowed to multiply.
Mozdziak has been thinking about animal protein since he rated carcasses as a teenage judge in the Future Farmers of America student competition. He went on to study agricultural science at Cornell. “I was chasing the pure science,” he says. “But what really clicked for me was the connection between biology and practical applications. How do we make muscle bigger? How do we treat patients? How do we produce more food?”
Until recently, Mozdziak says, no one wanted to fund his ideas for mass producing turkey meat without turkeys. But earlier this year, New Harvest gave Mozdziak a $118,800 grant, which has allowed him to create supplies of cells that other researchers can use by request. In November, he agreed to send some to biomedical engineer David Kaplan at Tufts University.
Kaplan says he’s searching for muscle cells that can be grown in “3-D,” or in tanks called bioreactors, and not just in thin sheets. That’s a necessary technical leap if in vitro meat is ever to scale up. “Our interest is in new ways to think about generating food-like tissues in a laboratory setting to better understand texture, structure, function, and nutrition,” says Kaplan, who says turkey cells look promising.
Biotech Startups Manufacturing Animal Tissue
Gelcor, San Leandro, California
Biotech production of animal gelatin
Perfect Foods, Berkeley, California
Animal-free dairy products
Clara Foods, San Francisco
Laboratory generated egg whites
Memphis Meats, San Leandro, California
Lab cultured beef and pork
Mosa Meat, Maastricht, Netherlands
Created first in vitro hamburger
SuperMeat, Israel
Developing cultured chicken liver
Mozdziak says breast meat has qualities that make it a good bet for growing at larger scale. The satellite cells can be induced to form either protein or fat cells—important because fat helps impart flavor. (A complaint against Post’s burger, which had no fat, was that it was too dry. A food writer called it “like an animal-protein cake.”)
Yet it will not be easy to compete with factory farms, known as concentrated animal-feeding operations. At peak efficiency, turkeys raised in such facilities are able to turn two pounds of feed into one pound of weight. This explains why a 16-pound frozen Butterball turkey costs just $15.84 at Target, only 99 cents a pound.
Despite this, Mozdziak believes the price of cultured meat will eventually be competitive. One reason is that you’d be creating only the white meat you will eat, not the rest of the bird. “You’re not giving nutrients to bone, you’re not giving nutrients to the alimentary tract, all body systems,” he says.
Inside his lab, Mozdziak’s students begin their work with a small cutting of live turkey breast. After breaking the tissue apart with enzymes, the challenge is to get the satellite cells to multiply, but without forming muscle too soon. His team has kept a single supply of cells growing for more than six months.
Some studies of in vitro meat suggest there could be large environmental benefits. Hanna Tuomisto, an agroecologist at the London School of Hygiene and Tropical Medicine, says producing beef in vitro could reduce greenhouse gas emissions from cattle by over 90 percent, and land use by 99 percent, but only if the vats were fed with pond scum called cyanobacteria.
Carolyn Mattick of Arizona State University is considerably more skeptical. Her calculations show that manufacturing chicken tissue with conventional nutrients, like glucose, would use more energy and release more greenhouse gases than farming chickens does. One reason is because ingredients would have to be heated to body temperature.
During a week in which Americans will consume 46 million turkeys, other researchers say the ethical benefits of in vitro meat must also factor into what technology is developed. Mozdziak’s graduate student Marie Gibbons, who cultivates the cells in his lab, says she quit studying to be a veterinarian after seeing farm animals treated cruelly.
"It would be better if farm animals didn’t have to exist,” she says.
Keep Reading
Most Popular
Large language models can do jaw-dropping things. But nobody knows exactly why.
And that's a problem. Figuring it out is one of the biggest scientific puzzles of our time and a crucial step towards controlling more powerful future models.
The problem with plug-in hybrids? Their drivers.
Plug-in hybrids are often sold as a transition to EVs, but new data from Europe shows we’re still underestimating the emissions they produce.
Google DeepMind’s new generative model makes Super Mario–like games from scratch
Genie learns how to control games by watching hours and hours of video. It could help train next-gen robots too.
How scientists traced a mysterious covid case back to six toilets
When wastewater surveillance turns into a hunt for a single infected individual, the ethics get tricky.
Stay connected
Get the latest updates from
MIT Technology Review
Discover special offers, top stories, upcoming events, and more.