Tracing a Memory
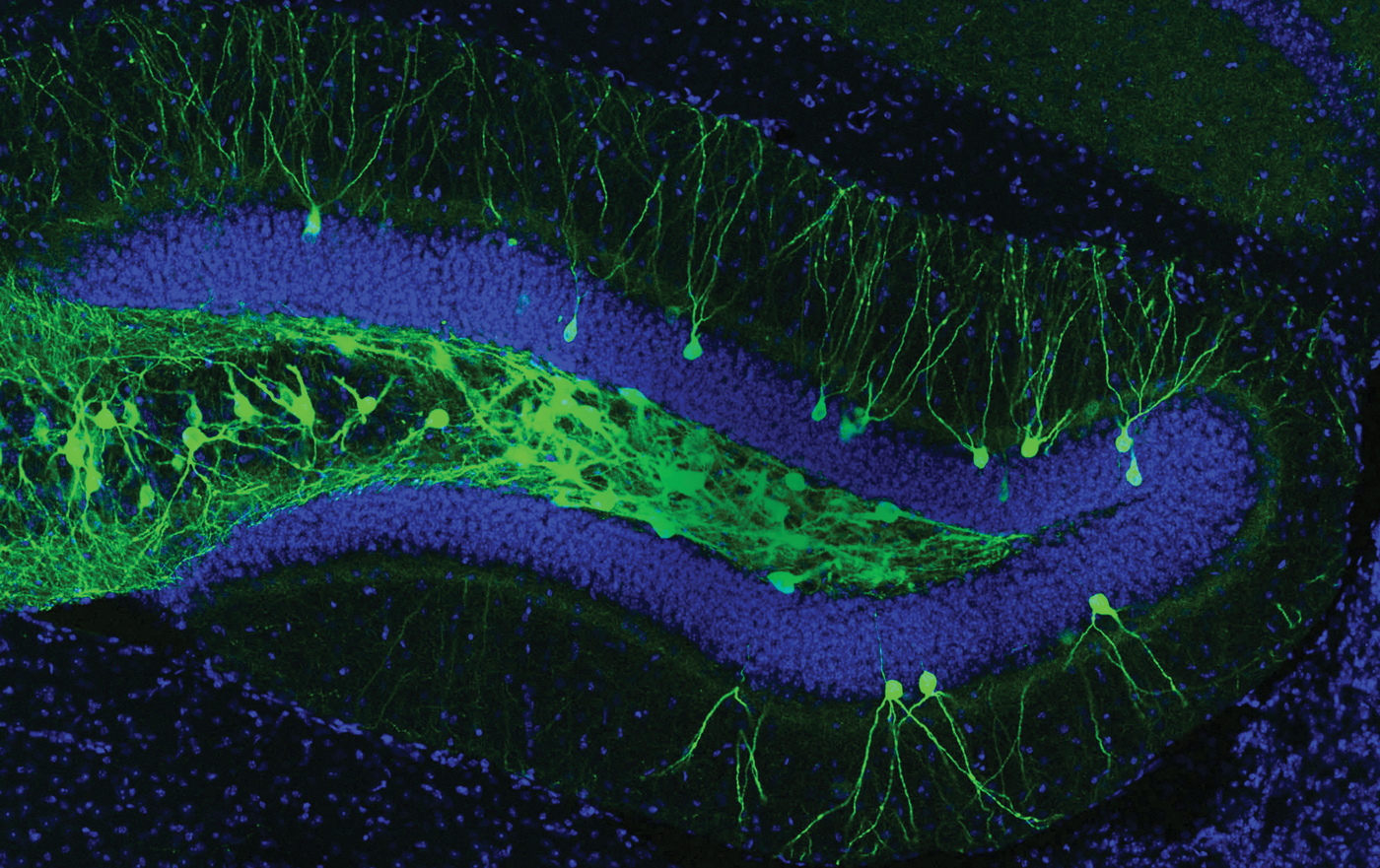
Susumu Tonegawa can make mice remember things that never happened. In a remarkable series of studies that his lab has published in the past four years, his team has devised ways to track and manipulate the animals’ memories—implanting false memories, changing bad memories to good ones, and even restoring memories in mice with amnesia. These studies have advanced the field of memory research and affirmed the 77-year-old Tonegawa as one of its leaders.
But as he recounts this work in his office at MIT’s Picower Institute for Learning and Memory, Tonegawa seems most animated when he’s talking about questions he hasn’t answered yet—such as those about the long-term fate of memories. His research has largely focused on the hippocampus, a region of the brain critical for remembering experiences. Scientists have known for decades that the hippocampus is only a temporary repository, he explains. A memory that we keep for years is held in the neocortex.
I ask him how it’s possible for a memory to move from one place in the brain to another.
How is it possible that an experience like a movie you watched last night is embedded in some fashion in your brain?
“That’s what we are studying now,” he says, his face lighting up. His lab is tracking the interaction between the two brain areas to figure out how memories end up in the neocortex. If the researchers can identify exactly which cells hold a long-term memory, they can begin to study and manipulate these memories as they’ve done with short-term ones. “But this is at the cutting edge of our research,” he says with a laugh, as if he’s giving something away. “This is the next step. We are doing many things, but this is one of the things I’m very excited about.”
Scientists who know Tonegawa say that he is always thinking about the next step. “One of the things that characterize Susumu as a scientist is a relentless focus on what he perceives to be the most important problem in his field,” says David Anderson, a neuroscientist at Caltech. “He is a total scientist. He wants to know what the answer to the question is; he cares about getting the right answer.”
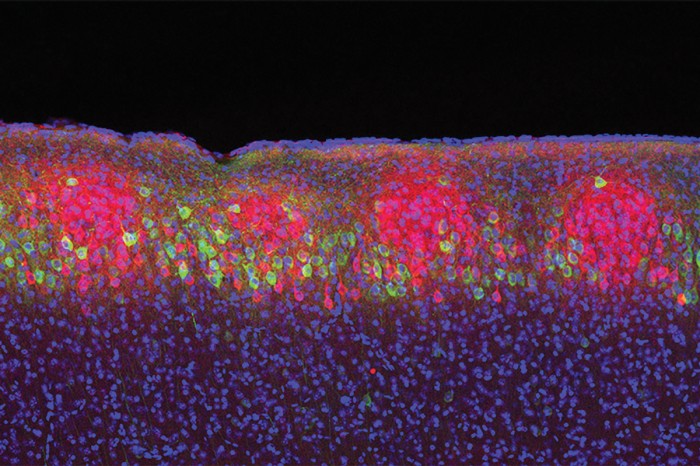
He isn’t big on social events, and he isn’t known for small talk (unless it’s about the Red Sox). Instead, he’s driven by curiosity. Specifically, about questions like this one: How is it possible that an experience like a movie you watched last night is embedded in some fashion in your brain?
“Memory is something that you cannot pin down,” Tonegawa says. “Conceptually, there’s a jump from a physical entity to a nonphysical phenomenon—information. The question is, how do you store the information? There has to be some kind of physical basis for this storage.”
Shocking memories
Brain science is really a second career for Tonegawa, who first gained prominence for work that helped explain how the immune system works. Born in Nagoya, Japan, in 1939, he studied chemistry before becoming fascinated with molecular biology in college. He got his PhD at the University of California, San Diego, and worked at the Salk Institute and then the Basel Institute for Immunology in Switzerland. There he addressed the problem of how the immune system can generate diverse antibodies to defend against pathogens. His experiments, which showed that the DNA used to make antibodies is shuffled around to yield many new combinations during an individual’s lifetime, earned him the Nobel Prize in Physiology or Medicine in 1987.
Tonegawa was recruited to MIT in 1981 to work in its Center for Cancer Research. His research involved genetically engineering mice to study the roles of genes in the immune system, and it was that technology that first led him to neuroscience. In the mid-1980s, he allowed a postdoc, Alcino Silva (now a director of the Integrative Center for Learning and Memory at UCLA), to explore an avenue of research that he was already thinking about: using genetic engineering to study the brain. In 1992, they reported that deleting a particular gene impaired mice’s ability to learn spatial information. It was an important finding: a single gene could affect a complex cognitive process.
Gradually, such work became the sole focus of Tonegawa’s lab. “I felt that the brain is a real subject for the future,” he says. Neuroscience was a burgeoning field, and genetically engineered mice opened up a new way to investigate it.

Tonegawa decided to zero in on learning and memory. He saw that sorting out how the brain acquires information and stores memories is one of the most critical questions in neuroscience. Although the brain is composed of cells like any other organ, it’s different in one important respect. “The brain has acquired the ability to take some information that comes from outside and store it and use it,” he says. “It’s a really fundamental feature of brain function.”
Tonegawa marshaled considerable resources toward addressing this question, founding the Center for Learning and Memory (later named the Picower Institute) at MIT in 1994. He would serve as its director until 2006. By 1996, his lab had helped develop a technique for switching off a gene of interest in specific brain regions and cell types, making it possible to investigate in unprecedented detail how the proteins produced by those genes affect animals’ cognitive processes. More recently, Tonegawa’s lab, like many others in neuroscience, has begun using new technologies such as optogenetics, which make it possible to study interconnected networks of cells in the living animal brain. This is an important advance, he says, because a single memory is formed from a pattern of connections between a unique subset of cells.
These technologies are allowing Tonegawa to probe the particulars of memory formation. Previous research enabled scientists to identify regions of the brain involved generally in memory formation and storage. But identifying a single, unique memory is much more challenging. A German zoologist, Richard Semon, coined the term “engram” in the early 20th century to describe the physical signature of a single memory. When you experience or learn new information, he speculated, some brain cells change, and when you encounter a similar stimulus later, those same cells are reactivated, causing you to recall the memory.
The theory was ignored for decades, but in recent years, scientists have made progress in hunting down the physical signature of specific memory engrams in the hippocampus. They showed, for instance, that deleting the brain cells active during the formation of a memory led animals to forget that information. Yet like all tactics that rely on brain damage, this approach is limited, says Sheena Josselyn, a senior scientist at the Hospital for Sick Children in Toronto. “You can do many things to make a mouse not remember something or not be able to perform a task,” she says. “There’s no specificity there.”
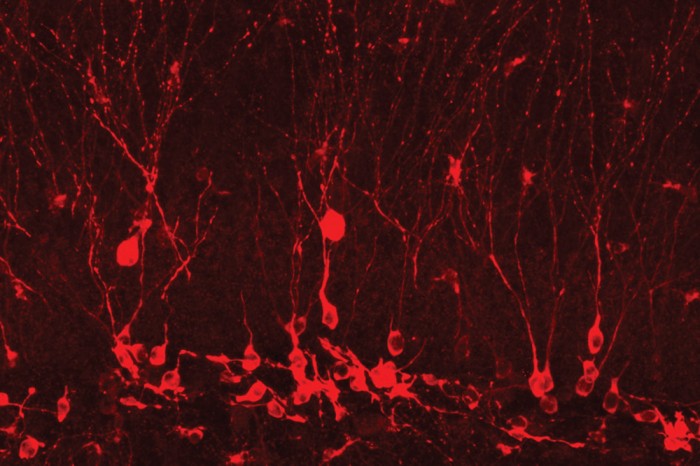
To better understand the process, Tonegawa says, they had to be able to reactivate a memory, not just destroy it. That breakthrough came in 2012, when lab members Xu Liu and Steve Ramirez, PhD ’15, published a paper in Nature demonstrating that they could elicit a specific memory in a mouse at will.
To do it, Liu and Ramirez (a 2013 TR35 winner) brought together and extended two technologies developed in other labs. One is optogenetics, which introduces a light-sensitive protein into cells so they can be activated with light. The other is a technique for tagging brain cells involved in memory formation. Liu and Ramirez kept the mice on the drug doxycycline, which suppresses the tagging; then they took the mice off the drug for a short window of time so they could target memory cells active during a specific event. The result: cells that they identified as forming a specific memory—which the researchers call the engram cells—can later be activated with light.
Liu and Ramirez used the technique to tag cells in the hippocampus of a mouse while it experienced a mild foot shock in a cage. Normally a mouse returned to the same cage would freeze, immobilized by the memory of its bad experience, but it would behave as usual when placed in a cage it didn’t associate with the shock. In this case, however, the researchers placed the mouse in a safe cage and delivered light pulses through tiny fibers to activate the engram cells in the hippocampus. The animal froze just as if it “remembered” being shocked in that environment.
Ramirez, who is now a principal investigator at Harvard’s Center for Brain Science, says that he and Liu were inspired by the movie Inception and “really fell in love with this idea of creating a new or false memory.” (Liu died in 2015, shortly after becoming an assistant professor at Northwestern University.) In a follow-up paper, they tagged memory-forming cells when mice were exploring a normal cage. The next day, they put the mice in a different cage and delivered a foot shock while activating the tagged cells. When the mice were placed again in the safe cage, they froze in fear, suggesting that they had been given the false memory of being shocked in that cage.
Pushing hard
Tonegawa gives the 40 members of his group extraordinary freedom, autonomy, and time to get the right answer or design the perfect experiment. Ramirez calls the lab, which sprawls across two floors of the Picower Institute, “a playground for science,” adding that Tonegawa is not wedded to a particular dogma or outcome. “He encourages people to follow the data,” he says.
Tonegawa is also known for pushing his lab members. Graduate student Dheeraj Roy recalls his excitement when he met with Tonegawa in September 2014 to show the latest data he’d collected. Roy was studying whether he could restore lost memories in mice with a condition similar to early Alzheimer’s disease. He had painstakingly modified the technique that Liu and Ramirez had developed to work in these mice, and his data looked great: by activating engram cells with light, he could make the Alzheimer’s mice remember receiving a foot shock in a cage several days before, something they normally would have forgotten.
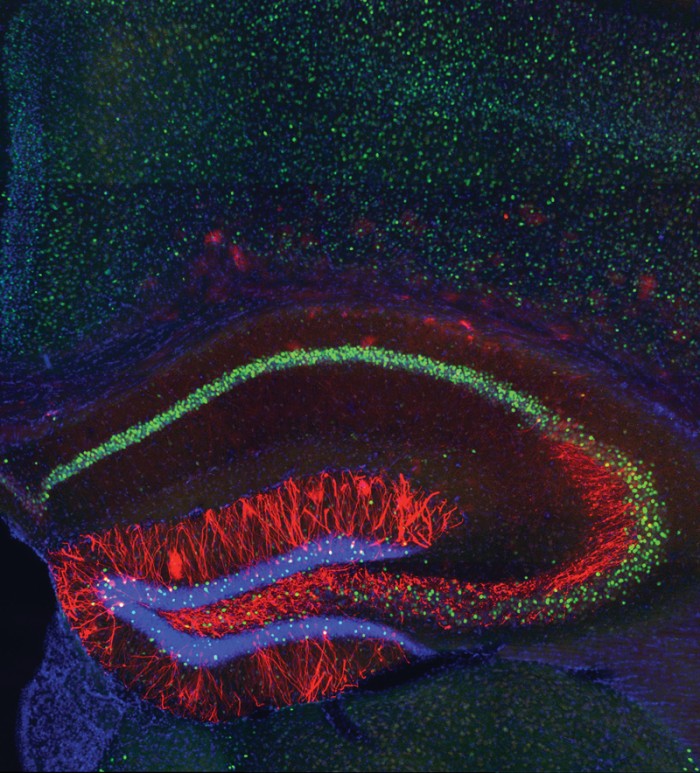
But Tonegawa’s reaction wasn’t exactly what Roy had hoped for. “After the first two minutes of our meeting, he had already moved on,” Roy recalls. What if there was a way to restore the memory permanently, he wondered, not just when the light was on? It was an idea they had never talked about before, but Roy suspects it may have been in Tonegawa’s mind all along.
It was what Roy calls “the famous ‘but.’”
“He says, ‘If only we could do one more experiment.’ We’ve all heard this four hundred times,” Roy says. “He won’t tell you the path, but he’ll show you the finish line.”
It took another 12 months of experiments after that meeting, but Roy went on to discover that connections between engram cells were weaker in memory-deficient mice, and he developed a protocol for pulsing light repeatedly on the cells that caused the connections to strengthen and memories to persist even when the light was off. The research, published earlier this year, not only demonstrated that memories persist in the diseased mice; it offered a new concept for restoring memory in neurodegenerative disease.
Other research in Tonegawa’s lab has explored the role of engram cells in normal behavior as well as disease. Ramirez and Liu led a study that found they could eliminate depression-like behavior in mice exposed to chronic stress by reactivating engram cells associated with an earlier positive experience. Research led by Roger Redondo, then a postdoc, and graduate student Joshua Kim explored whether the perception of a memory as negative or positive could be reversed. And a recent study in Science identified an area of the hippocampus involved in remembering social interactions. When Teruhiro Okuyama worked with mice to activate engram cells linked to the memory of meeting another mouse, the animal reacted as if a novel mouse were familiar.
These studies have fascinated the public with the suggestion that our memories are malleable, but Tonegawa points out that translating rodent experiments into human results is still far off. “We hope that what we do with animal studies is useful for future therapy,” he says. But safely stimulating human brain cells and circuits with such precision is still not possible.
Instead, he’s focused on several questions that have emerged from this research. He is exploring the relationship between the hippocampus and other brain areas, continuing to unravel the role of emotion in memory, and investigating how we remember sequences of past events.
Tonegawa, who directs the RIKEN-MIT Center for Neural Circuit Genetics and the RIKEN Brain Science Institute in Japan, recently reduced the size of his lab and says he may conduct fewer of the exploratory projects that have often led his research in new directions. His interests have narrowed, he says, in the wake of a personal tragedy: the suicide of his son Satto, an MIT undergraduate, in 2011. But his enduring focus on his scientific work has helped him cope with his grief.
“As long as I’m alive and have energy, I’d like to continue,” he says, but he adds with a chuckle: “I think I will stay with memory.”
Note: This story was modified on November 16, 2016.
Keep Reading
Most Popular
Large language models can do jaw-dropping things. But nobody knows exactly why.
And that's a problem. Figuring it out is one of the biggest scientific puzzles of our time and a crucial step towards controlling more powerful future models.
How scientists traced a mysterious covid case back to six toilets
When wastewater surveillance turns into a hunt for a single infected individual, the ethics get tricky.
The problem with plug-in hybrids? Their drivers.
Plug-in hybrids are often sold as a transition to EVs, but new data from Europe shows we’re still underestimating the emissions they produce.
Stay connected
Get the latest updates from
MIT Technology Review
Discover special offers, top stories, upcoming events, and more.