Manufacturing Dopamine in the Brain with Gene Therapy
Parkinson’s patients who take the drug levodopa, or L-Dopa, are inevitably disappointed. At first, during a “honeymoon” period, their symptoms (which include tremors and balance problems) are brought under control. But over time the drug becomes less effective. They may also need ultrahigh doses, and some start spending hours a day in a state of near-frozen paralysis.
A biotech company called Voyager Therapeutics now thinks it can extend the effects of L-Dopa by using a surprising approach: gene therapy. The company, based in Cambridge, Massachusetts, is testing the idea in Parkinson’s patients who’ve agreed to undergo brain surgery and an injection of new DNA.

Parkinson’s occurs when dopamine-making neurons in the brain start dying, causing movement symptoms that afflicted boxing champ Muhammad Ali and actor Michael J. Fox, whose charitable foundation has helped pay for the development of Voyager’s experimental treatment.
The cause of Parkinson’s isn’t well understood, but the reason the drug wears off is. It’s because the brain also starts losing an enzyme known as aromatic L-amino acid decarboxylase, or AADC, that is needed to convert L-Dopa into dopamine.
Voyager’s strategy, which it has begun trying on patients in a small study, is to inject viruses carrying the gene for AADC into the brain, an approach it thinks can “turn back the clock” so that L-Dopa starts working again in advanced Parkinson’s patients as it did in their honeymoon periods.
Videos of patients before and after taking L-Dopa make it obvious why they’d want the drug to work at a lower dose. In the ‘off’ state, people move in slow motion. Touching one’s nose takes an effort. In an ‘on’ state, when the drug is working, they’re shaky, but not nearly so severely disabled.
“They do well at first but then respond very erratically to L-Dopa,” says Krystof Bankiewicz, the University of California scientist who came up with the gene-therapy plan and is a cofounder of Voyager. “This trial is to restore the enzyme and allow them to be awakened, or ‘on,’ for a longer period of time.”

Voyager was formed in 2013 and later went public, raising about $86 million. The company is part of a wave of biotechs that have been able to raise money for gene therapy, a technology that is starting to pay off: after three decades of research, a few products are reaching the market.
Unlike conventional drug studies, those involving gene therapy often come with very high expectations that the treatment will work. That’s because it corrects DNA errors for which the exact biological consequences are known. Genzyme, a unit of the European drug manufacturer Sanofi, paid Voyager $65 million and promised hundreds of millions more in order to sell any treatments it develops in Europe and Asia.
“We’re working with 60 years of dopamine pharmacology,” says Steven Paul, Voyager’s CEO, and formerly an executive at the drug giant Eli Lilly. “If we can get the gene to the right tissue at the right time, it would be surprising if it didn’t work.”
But those are big ifs. In fact, the concept for the Parkinson’s gene therapy dates to 1986, when Bankiewicz first determined that too little AADC was the reason L-Dopa stops working. He thought gene therapy might be a way to fix that, but it wasn’t until 20 years later that he was able to test the idea in 10 patients, in a study run by UCSF.
In that trial, Bankiewicz says, the gene delivery wasn’t as successful as anticipated. Not enough brain cells were updated with the new genetic information, which is shuttled into them by viruses injected into the brain. Patients seemed to improve, but not by much.
Even though the treatment didn’t work as planned, that early study highlighted one edge Voyager’s approach has over others. It is possible to tag AADC with a marker chemical, so doctors can actually see it working inside patients’ brains. In fact, ongoing production of the dopamine-making enzyme is still visible in the brains of the UCSF patients several years later.
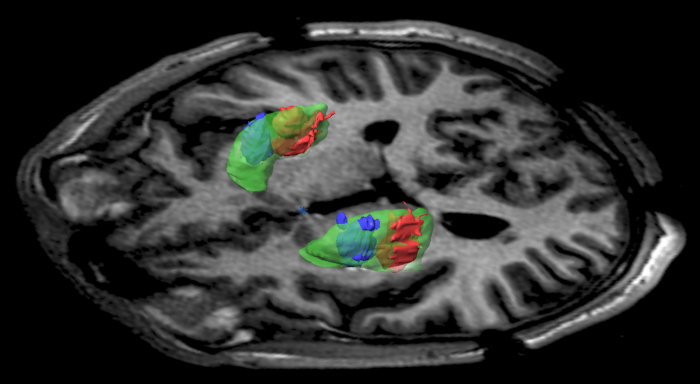
In some past studies of gene therapy, by contrast, doctors had to wait until patients died to find out whether the treatment had been delivered correctly. “This is a one-and-done treatment,” says Paul. “And anatomically, it tells us if we got it in the right place.”
A new trial under way, this one being carried out by Voyager, is designed to get much higher levels of DNA into patients’ brains in hopes of achieving better results. To do that, Bankiewicz developed a system to inject the gene-laden viral particles through pressurized tubes while a patient lies inside an MRI scanner. That way, the surgeon can see the putamen, the brain region where the DNA is meant to end up, and make sure it’s covered by the treatment.
There are other gene therapies for Parkinson’s disease planned or in testing. A trial developed at the National Institutes of Health seeks to add a growth factor and regenerate cells. A European company, Oxford BioMedica, is trying to replace dopamine.
Altogether, as of this year, there were 48 clinical trials under way of gene or cell replacement in the brain and nervous system, according to the Alliance for Regenerative Medicine, a trade group. The nervous system is the fourth most common target for this style of experimental treatment, after cancer, heart disease, and infections.
Voyager’s staff is enthusiastic about a study participant they call “patient number 6,” whom they’ve been tracking for several months—ever since he got the treatment. Before the gene therapy, he was on a high dose of L-Dopa but still spent six hours a day in an “off” state. Now he’s off only two hours a day and takes less of the drug.
That patient got the highest dose of DNA yet, covering the largest brain area. That is part of what makes Voyager think higher doses should prove effective. “I believe that previous failure of gene-therapy trials in Parkinson’s was due to suboptimal delivery,” says Bankiewicz.
Keep Reading
Most Popular
Large language models can do jaw-dropping things. But nobody knows exactly why.
And that's a problem. Figuring it out is one of the biggest scientific puzzles of our time and a crucial step towards controlling more powerful future models.
How scientists traced a mysterious covid case back to six toilets
When wastewater surveillance turns into a hunt for a single infected individual, the ethics get tricky.
The problem with plug-in hybrids? Their drivers.
Plug-in hybrids are often sold as a transition to EVs, but new data from Europe shows we’re still underestimating the emissions they produce.
Google DeepMind’s new generative model makes Super Mario–like games from scratch
Genie learns how to control games by watching hours and hours of video. It could help train next-gen robots too.
Stay connected
Get the latest updates from
MIT Technology Review
Discover special offers, top stories, upcoming events, and more.