To Study the Brain, a Doctor Puts Himself Under the Knife
Phil Kennedy no longer saw any other way to get the data. That was how one day he came to lie blissfully unconscious on an operating table in Belize while a neurosurgeon sawed off the top of his skull.

Last year, Kennedy, a 67-year-old neurologist and inventor, did something unprecedented in the annals of self-experimentation. He paid a surgeon in Central America $25,000 to implant electrodes into his brain in order to establish a connection between his motor cortex and a computer.
Along with a small group of pioneers, Kennedy had in the late 1980s developed “invasive” human brain-computer interfaces—literally wires inside the brain attached to a computer, and he is widely credited as the first to allow a severely paralyzed “locked-in” patient to move a computer cursor using her brain. “The father of cyborgs,” one magazine called him.
Kennedy’s scientific aim has been to build a speech decoder—software that can translate the neuronal signals produced by imagined speech into words coming out of a speech synthesizer. But this work, carried out by his small Georgia company Neural Signals, had stalled, Kennedy says. He could no longer find research subjects, had little funding, and had lost the support of the U.S. Food and Drug Administration.
That is why in June 2014, he found himself sitting in a distant hospital contemplating the image of his own shaved scalp in a mirror. “This whole research effort of 29 years so far was going to die if I didn’t do something,” he says. “I didn’t want it to die on the vine. That is why I took the risk.”
This fall, Kennedy presented studies of his own brain at the Society for Neuroscience in Chicago, where his actions provoked both awe and concern among colleagues. By arranging for surgery on a healthy person—even himself, even in the name of science—he’d likely violated his doctor’s oath. “I’m glad he’s fine now,” says Eddie Chang, a University of California, San Francisco, neurosurgeon whose recent work mapping the areas of the motor cortex that control speech helped guide Kennedy’s calculations. “I hope he gets some precious, precious data.”
FDA trouble
Kennedy, who was born in Ireland, says his self-experiment was driven by frustration and by scientific questions. He was so intrigued by the brain as a young physician that he returned to school to earn a Ph.D. in neuroscience. While running a lab at the Georgia Institute of Technology in the 1980s, he developed and patented an innovative type of electrode consisting of a pair of gold wires encased in a tiny glass cone. Filled with a proprietary blend of growth factors, the electrode induced nearby neurons to grow into the device.

In 1996, after tests in animals, the FDA agreed to allow Kennedy to implant his electrodes into locked-in patients with paralysis so severe they could no longer speak or move. His first volunteer was a special education teacher and mother of two named Marjory, or “MH,” who agreed to undergo the procedure at the very end of her life. Marjory had ALS but demonstrated she could turn a switch on and off just by thinking. But she was so sick that only 76 days later, she died. Next, in 1998, came Johnny Ray, a 53-year-old Vietnam veteran and drywall contractor who awoke from a coma with his mind fully intact but unable to move anything except his eyelids.
Kennedy personally oversaw the implantation of the electrodes in at least five subjects, and his team began showing that if it recorded from just a few neurons, patients could move a cursor on a computer screen and communicate by picking words or letters from a menu.
By 2004, Kennedy had implanted his electrodes in the brain of Erik Ramsey, a volunteer who suffered a catastrophic brain stem stroke in a car accident that left him locked in at the age of 16. Thanks to the data collected from Ramsey, Kennedy and his collaborators continued to publish high-profile papers on the results in journals like PLOS One and Frontiers in Neuroscience as recently as 2009 and 2011. One paper described how software could pick out the sounds Ramsey was imagining and allow him to very roughly pronounce a few simple words. Eventually, Ramsey became too ill to keep participating in the research.
By then, the FDA had also withdrawn permission to use the devices in any more patients. Kennedy says the agency began asking him for more safety data, including on the neurotrophic factors he was using to induce neuronal growth. When Kennedy couldn’t provide the data, the FDA refused to approve any more implants.
Kennedy never fully accepted the FDA decision (he took at least one other patient to Belize for an implant). There were also scientific frustrations working with disabled people. Locked-in people can’t communicate, except at times with grunts or their eyes, something that added a confounding variable to his experiments. When a given neuron fired off, he could never be sure what the patient had been thinking.
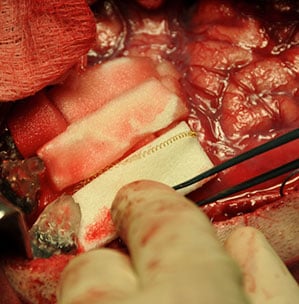
Kennedy became convinced that the way to take his research to the next level was to find a volunteer who could still speak. For almost a year he searched for a volunteer with ALS who still retained some vocal abilities, hoping to take the patient offshore for surgery. “I couldn’t get one. So after much thinking and pondering I decided to do it on myself,” he says. “I tried to talk myself out of it for years.”
The surgery took place in June 2014 at a 13-bed Belize City hospital a thousand miles south of his Georgia-based neurology practice and also far from the reach of the FDA. Prior to boarding his flight, Kennedy did all he could to prepare. At his small company, Neural Signals, he fabricated the electrodes the neurosurgeon would implant into his motor cortex—even chose the spot where he wanted them buried. He put aside enough money to support himself for a few months if the surgery went wrong. He had made sure his living will was in order and that his older son knew where he was.
Walking the walk
Down in Belize, the procedure did not go smoothly, pointing to the dangers of brain-computer interface science to volunteers. There is a small but real chance of death anytime the skull is opened. After waking up from his first surgery, Kennedy says, he could not reply when the surgeons spoke to him. He had lost the ability to speak. The doctors later explained that his blood pressure had spiked during the 12-hour surgery, causing the brain to swell and leading to temporary paralysis. “I wasn’t the least bit scared,” says Kennedy. “I knew what was going on. I invented the surgery.”
The side effects were very serious, but Kennedy says he recovered and returned for a second 10-hour procedure in Belize City several months later so the surgeon could implant electronics that would let him collect signals from his own brain.
Kennedy’s heroics impress some of his former patients. “Talk about walking the walk!” David Jayne, an ALS patient implanted by Kennedy’s team in the early 2000s, said in an e-mail. “I admire the hell out of Phil.”
To some researchers, Kennedy’s decisions could be seen as unwise, even unethical. Yet there are cases where self-experiments have paid off. In 1984, an Australian doctor named Barry Marshall drank a beaker filled with bacteria in order to prove they caused stomach ulcers. He later won the Nobel Prize. “There’s been a long tradition of medical scientists experimenting on themselves, sometimes with good results and sometimes without such good results,” says Jonathan Wolpaw, a brain-computer interface researcher at the Wadsworth Center in New York. “It’s in that tradition. That’s probably all I should say without more information.”
After returning home to Duluth, Georgia, Kennedy began to toil largely alone in his speech lab, recording his neurons as he repeated 29 phonemes (such as e, eh, a, o, u, and consonants like ch and j) out loud, and then silently imagined saying them. He did the same with about 290 short words such as “Dale” and “plum.” There were also phrases to speak: “Hello, world,” “Which private firm,” and “The joy of a jog makes a boy say wow.”
Kennedy says his early findings are “extremely encouraging.” He says he determined that different combinations of the 65 neurons he was recording from consistently fired every time he spoke certain sounds aloud, and also fired when he imagined speaking them—a relationship that is potentially key to developing a thought decoder for speech. At UCSF, Chang says that Kennedy may have learned something new. His own research uses different electrodes placed outside the brain, which he says are able to collect only a “relatively rough” signal (see “A Speech Synthesizer Direct to the Brain”). “I think what he might have access to there is something a lot more detailed,” says Chang.
There was one major disappointment. Kennedy had hoped to live with the implants in his brain for years, collecting data, improving his control, and publishing papers. But the incision in his skull never closed entirely, creating a dangerous situation. After a few weeks of collecting data, last January Kennedy was forced to ask doctors at a local Georgia hospital to remove the implants. The bill came to $94,000. Kennedy submitted the claim to his insurance company (he says it paid $15,000).
Kennedy attributes this setback to his decision to build the electrodes extra large and install them at an unusual angle so they would be easier to work with—a decision he now believes was a mistake. “But I got away with it, so I’m happy,” he says. “I had a few bumps and bruises after the surgery, but I did get four weeks of good data. I will be working on these data for a long time.”
Deep Dive
Biotechnology and health
How scientists traced a mysterious covid case back to six toilets
When wastewater surveillance turns into a hunt for a single infected individual, the ethics get tricky.
An AI-driven “factory of drugs” claims to have hit a big milestone
Insilico is part of a wave of companies betting on AI as the "next amazing revolution" in biology
The quest to legitimize longevity medicine
Longevity clinics offer a mix of services that largely cater to the wealthy. Now there’s a push to establish their work as a credible medical field.
There is a new most expensive drug in the world. Price tag: $4.25 million
But will the latest gene therapy suffer the curse of the costliest drug?
Stay connected
Get the latest updates from
MIT Technology Review
Discover special offers, top stories, upcoming events, and more.