Genome Surgery

Over the last decade, as DNA-sequencing technology has grown ever faster and cheaper, our understanding of the human genome has increased accordingly. Yet scientists have until recently remained largely ham-fisted when they’ve tried to directly modify genes in a living cell. Take sickle-cell anemia, for example. A debilitating and often deadly disease, it is caused by a mutation in just one of a patient’s three billion DNA base pairs. Even though this genetic error is simple and well studied, researchers are helpless to correct it and halt its devastating effects.
Now there is hope in the form of new genome-engineering tools, particularly one called CRISPR. This technology could allow researchers to perform microsurgery on genes, precisely and easily changing a DNA sequence at exact locations on a chromosome. Along with a technique called TALENs, invented several years ago, and a slightly older predecessor based on molecules called zinc finger nucleases, CRISPR could make gene therapies more broadly applicable, providing remedies for simple genetic disorders like sickle-cell anemia and eventually even leading to cures for more complex diseases involving multiple genes. Most conventional gene therapies crudely place new genetic material at a random location in the cell and can only add a gene. In contrast, CRISPR and the other new tools also give scientists a precise way to delete and edit specific bits of DNA—even by changing a single base pair. This means they can rewrite the human genome at will.
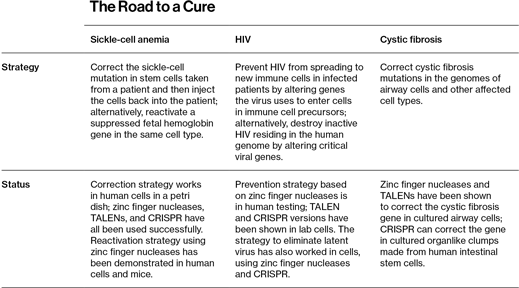
It is likely to be at least several years before such efforts can be developed into human therapeutics, but a growing number of academic researchers have seen some preliminary success with experiments involving sickle-cell anemia, HIV, and cystic fibrosis (see table below). One is Gang Bao, a bioengineering researcher at the Georgia Institute of Technology, who has already used CRISPR to correct the sickle-cell mutation in human cells grown in a dish. Bao and his team started the work in 2008 using zinc finger nucleases. When TALENs came out, his group switched quickly, says Bao, and then it began using CRISPR when that tool became available. While he has ambitions to eventually work on a variety of diseases, Bao says it makes sense to start with sickle-cell anemia. “If we pick a disease to treat using genome editing, we should start with something relatively simple,” he says. “A disease caused by a single mutation, in a single gene, that involves only a single cell type.”
Bao has an idea of how such a treatment would work. Currently, physicians are able to cure a small percentage of sickle-cell patients by finding a human donor whose bone marrow is an immunological match; surgeons can then replace some of the patient’s bone marrow stem cells with donated ones. But such donors must be precisely matched with the patient, and even then, immune rejection—a potentially deadly problem—is a serious risk. Bao’s cure would avoid all this. After harvesting blood cell precursors called hematopoietic stem cells from the bone marrow of a sickle-cell patient, scientists would use CRISPR to correct the defective gene. Then the gene-corrected stem cells would be returned to the patient, producing healthy red blood cells to replace the sickle cells. “Even if we can replace 50 percent, a patient will feel much better,” says Bao. “If we replace 70 percent, the patient will be cured.”
Though genome editing with CRISPR is just a little over a year old, it is already reinventing genetic research. In particular, it gives scientists the ability to quickly and simultaneously make multiple genetic changes to a cell. Many human illnesses, including heart disease, diabetes, and assorted neurological conditions, are affected by numerous variants in both disease genes and normal genes. Teasing out this complexity with animal models has been a slow and tedious process. “For many questions in biology, we want to know how different genes interact, and for this we need to introduce mutations into multiple genes,” says Rudolf Jaenisch, a biologist at the Whitehead Institute in Cambridge Massachusetts. But, says Jaenisch, using conventional tools to create a mouse with a single mutation can take up to a year. If a scientist wants an animal with multiple mutations, the genetic changes must be made sequentially, and the timeline for one experiment can extend into years. In contrast, Jaenisch and his colleagues, including MIT researcher Feng Zhang (a 2013 member of our list of 35 innovators under 35), reported last spring that CRISPR had allowed them to create a strain of mice with multiple mutations in three weeks.

Because a CRISPR system can easily be designed to target any specific gene, the technology is allowing researchers to do experiments that probe a large number of them. In December, teams led by Zhang and MIT researcher Eric Lander created libraries of CRISPRs, each of which targets a different human gene. These vast collections, which account for nearly all the human genes, have been made available to other researchers. The libraries promise to speed genome-wide studies on the genetics of cancer and many other human diseases.
Genome GPS
The biotechnology industry was born in 1973, when Herbert Boyer and Stanley Cohen inserted foreign DNA that they had manipulated in the lab into bacteria. Within a few years, Boyer had cofounded Genentech, and the company had begun using E. coli modified with a human gene to manufacture insulin for diabetics. In 1974, Jaenisch, then at the Salk Institute for Biological Studies in San Diego, created the first transgenic mouse by using viruses to spike the animal’s genome with a bit of DNA from another species. In these and other early examples of genetic engineering, however, researchers were limited to techniques that inserted the foreign DNA into the cell at random. All they could do was hope for the best.
It took more than two decades before molecular biologists became adept at efficiently changing specific genes in animal genomes. Dana Carroll of the University of Utah recognized that zinc finger nucleases, engineered proteins reported by colleagues at Johns Hopkins University in 1996, could be used as a programmable gene-targeting tool. One end of the protein can be designed to recognize a particular DNA sequence; the other end cuts DNA. When a cell then naturally repairs those cuts, it can patch its genome by copying from supplied foreign DNA. While the technology finally enabled scientists to confidently make changes where they want to on a chromosome, it’s difficult to use. Every modification requires the researcher to engineer a new protein tailored to the targeted sequence—a difficult, time-consuming task that, because the proteins are finicky, doesn’t always work.
TALENs, another significant advance in gene editing, came along in 2010. TALENs are also proteins that find and cut a desired DNA sequence—but tailoring them to new gene targets is much easier. While they represented a great improvement over zinc fingers, however, TALENs are large proteins that are cumbersome to work with and deliver into cells.
CRISPR changed everything. It replaces the DNA-targeting proteins with a short bit of RNA that homes in on desired genes. Unlike the complex proteins, RNA—which has nearly the same simple structure as DNA—can be made routinely in the lab; a technician can quickly synthesize the roughly 20-letter-long sequences the method requires. The system makes it easy for medical researchers to modify a genome by replacing, deleting, or adding DNA.
In little more than a year, CRISPR has begun reinventing genetic research.
CRISPR stands for “clustered regularly interspaced short palindromic repeats”—clusters of brief DNA sequences that read similarly forward and backward, which are found in many types of bacteria. Scientists first observed the puzzling DNA segments in the 1980s but didn’t understand for almost two decades that they are part of a bacterial defense system. When a virus attacks, bacteria can incorporate sequences of viral DNA into their own genetic material, sandwiching them between the repetitive segments. The next time the bacteria encounter that virus, they use the DNA in these clusters to make RNAs that recognize the matching viral sequences. A protein attached to one of these RNAs then cuts up the viral DNA.
In 2012, Emmanuelle Charpentier, a medical microbiologist who studies pathogens at the Helmholtz Centre for Infection Research, and Jennifer Doudna, a collaborator at the University of California, Berkeley, showed they could use a single RNA in conjunction with the cutting protein, an enzyme called Cas9, to slice any desired sequence of DNA in test tubes. It was still uncertain whether the method would work in animal cells, but in January 2013 came a dramatic breakthrough. Zhang and George Church, a Harvard Medical School geneticist, separately reported that the CRISPR/Cas9 system could be used for gene editing in the cells of animals, including humans.
Now a researcher who wants to go after a new gene need only synthesize the Cas9 protein and a bit of RNA that matches the sequences of the targeted region. The RNA then guides the enzyme to the DNA the researcher wants to cut. And because the same cutting protein is used regardless of the target, researchers can design experiments in which they change multiple genes in an organism simultaneously using Cas9 and multiple RNA guides. “It offers the potential to do experiments that in the past were very difficult or essentially not possible,” says Doudna.
Complex Mysteries
MIT’s Zhang, who is a member of the Broad Institute and the McGovern Institute for Brain Research, is interested in the genetics behind mental illness. To try to understand these complex conditions, Zhang has helped develop multiple gene- and neuron-modifying tools, including TALENs and optogenetics, a technique that involves controlling neuron activity with laser light. When he first heard about CRISPR, in 2011, he began to engineer it for use in human cells. Now he’s using CRISPR to help reveal the genetic secrets behind such devastating and poorly understood conditions as schizophrenia and autism.
The tool allows Zhang to begin systematically testing some of the DNA variants that have been linked to the illnesses. While much progress has been made over the last decade toward identifying genes that are common in people with these conditions, understanding how these genes relate to the symptoms is a daunting challenge. “What you learn from sequencing is only an observation,” says Zhang: in order to understand whether a suspected gene is actually causing the condition, you have to introduce the specific mutation into healthy cells or organisms and see what goes wrong. If the mutated cell or organism has features that mimic the human disease, that’s evidence implicating the gene.
Zhang can re-create, in both lab mice and cultured human cells, genetic variants found in people with autism and schizophrenia. “You can put a human mutation into the corresponding gene in a lab animal and then see: does that animal become less social or have a learning deficit?” he says. Then, he adds, you can study differences in the behavior and physiology of lab-cultured neurons grown from stem cells that have been modified with the same mutation. “With single-gene mutations, we will start to see aspects of the biological function that are involved in autism,” he says.
Zhang is also using CRISPR to make multiple genetic changes at once. That becomes particularly important with complex diseases like autism and schizophrenia, which for the most part are not caused by the type of single DNA change behind sickle-cell anemia. Different patients are affected by different collections of mutations. Solving a puzzle of such immense complexity will require large, systematic studies on the effects of various genes and the way they interact. CRISPR makes such studies possible, says Zhang, and will be important in finding treatments for a variety of complex diseases. “We will understand more about pathways and disease mechanisms,” he says. “This knowledge will inform all kinds of drug development.”
Designer Babies
Late last year, Doudna, Zhang, Church, and two other pioneers of genome editing founded a startup that will develop novel treatments for human genetic diseases. In November the company, Editas Medicine, announced that it had raised $43 million in venture capital and said it plans to use genome-editing technologies against a broad range of illnesses.
The launch of Editas should benefit from a resurgence of interest in gene therapy thanks to years of technological improvements, including safer mechanisms for delivering treatment. “The landscape has changed for gene therapy,” says Church. (There are still no gene therapies approved in the United States, though a number are in human trials.) But he says the therapies that Editas will develop will be fundamentally different from the older approaches that use a virus to insert a gene into cells.
Perhaps scientists could rewrite normal genes so that humans could better fight infections.
“Making a change or a deletion is out of range for most of those simple viral methods,” Church says. And deleting a bit of DNA, rather than adding a gene, may indeed be the key to treating many illnesses. Take Huntington’s disease. The fatal brain condition arises from a buildup of a toxic protein in neurons. Adding a healthy copy of the gene to the cell would not affect that protein’s poisonous activity: the original dysfunctional version must be rewritten. With the new genome-editing tools, says Church, rewriting the defective DNA may be possible: “You aren’t limited to adding back something that is missing.” And, he adds, “when you start realizing that the most common versions of genes are not necessarily the ideal versions, then you realize this is a much bigger field.” Perhaps scientists could rewrite normal genes so that humans can better fight infectious diseases. They might even be able to shake up the molecular pathways involved in aging.
Church also predicts that if genome editing is used to cure childhood diseases, some scientists will be tempted to use it to engineer embryos during in vitro fertilization. Researchers have already shown that genome editing can rewrite DNA sequences in rat and mouse embryos, and in late January, researchers in China reported that they had created genetically modified monkeys using CRISPR. With such techniques, a person’s genome might be edited before birth—or, if changes were made to the eggs or sperm-producing cells of a prospective parent, even before conception.
These possibilities raise ethical questions. But if researchers prove they can safely correct diseases by editing the genome, it’s inevitable that some parents will also want to alter the genomes of healthy embryos. “If you can prevent mental retardation with gene therapy, presuming that that’s permissible, then there’s a whole spectrum of intellectual challenges that will be discussed,” says Church.
Such discussions are likely to heat up as CRISPR becomes more widely used. For now, though, the technology is still evolving: while researchers like Bao, Church, and Zhang ultimately hope to cure some of our most intractable diseases, much of their time is still spent simply fine-tuning the tool and exploring its possibilities. But even in these early days, CRISPR has already transformed how these researchers think about manipulating the genome. They are ham-fisted no longer.
Deep Dive
Biotechnology and health
How scientists traced a mysterious covid case back to six toilets
When wastewater surveillance turns into a hunt for a single infected individual, the ethics get tricky.
An AI-driven “factory of drugs” claims to have hit a big milestone
Insilico is part of a wave of companies betting on AI as the "next amazing revolution" in biology
The quest to legitimize longevity medicine
Longevity clinics offer a mix of services that largely cater to the wealthy. Now there’s a push to establish their work as a credible medical field.
There is a new most expensive drug in the world. Price tag: $4.25 million
But will the latest gene therapy suffer the curse of the costliest drug?
Stay connected
Get the latest updates from
MIT Technology Review
Discover special offers, top stories, upcoming events, and more.