Why We Will Need Genetically Modified Foods

Signs of late blight appear suddenly but predictably in Ireland as soon as the summer weather turns humid, spores of the funguslike plant pathogen wafting across the open green fields and landing on the wet leaves of the potato plants. This year it began to rain in early August. Within several weeks, late blight had attacked a small plot of potatoes in the corner of the neat grid of test plantings at the headquarters of Teagasc, Ireland’s agricultural agency, in Carlow.
It’s now more than a month after the potato plants were first struck and still a few weeks before the crop will be harvested. A large country house, housing the operations of Teagasc, overlooks the field trials, and well-dressed Irish and EU bureaucrats hustle in and out. Much of the sprawling building was constructed in the 1800s, during the worst of the famines that were triggered when blight devastated Ireland’s potato harvest. Such famines are far in the past, but the plant disease remains a costly torment to the country’s farmers, requiring them to douse their crops frequently with fungicides. As part of an EU-wide project called Amiga to study the impact of genetically modified (GM) plants, Teagasc researcher Ewen Mullins is testing potatoes that are engineered to resist blight. (Watch a video of Mullins and GM potatoes in Ireland at the bottom of this page or here.)
It’s breezy, and though the summer is over, it’s still warm and humid. “Perfect weather for blight,” says Mullins. Bending over the conventionally bred plants, he firmly pulls back the wilted stems and leaves to show that the tubers, half-exposed in the ground, are scarred with black blotches. Then he picks at a green leaf from one of the genetically engineered plants, which have been modified with a blight-resistant gene from a wild potato that grows in South America. The defenses of the potato plant have fought off the spores, rendering them harmless. The plant, says Mullins simply, “has performed well.”

It’s the second year of what are scheduled to be three-year field trials. But even if the results from next year are similarly encouraging, Teagasc has no intention of giving farmers access to the plant, which was developed by researchers at Wageningen University in the Netherlands. Such genetically engineered crops remain controversial in Europe, and only two are approved for planting in the EU. Though Mullins and his colleagues are eager to learn how blight affects the GM potatoes and whether the plants will affect soil microbes, distributing the modified plant in Ireland is, at least for now, a nonstarter.
Nevertheless, the fields of Carlow present a tantalizing picture of how genetically modified crops could help protect the world’s food supply. Blight-resistant potatoes would be one of the first major foods genetically engineered to incorporate defenses against plant diseases, which annually destroy some 15 percent of the world’s agricultural harvest. Despite the heavy use of fungicides, late blight and other plant diseases ruin an estimated fifth of the world’s potatoes, a food increasingly grown in China and India. Stem rust, a fungal disease of wheat, has spread through much of Africa and the Arabian Peninsula and is now threatening the vast growing regions of central and south Asia, which produce some 20 percent of the world’s wheat. Bananas, which are a primary source of food in countries such as Uganda, are often destroyed by wilt disease. In all these cases, genetic engineering has the potential to create varieties that are far better able to withstand the onslaught.
GM potatoes could also lead to a new generation of biotech foods sold directly to consumers. Though transgenic corn, soybeans, and cotton—mostly engineered to resist insects and herbicides—have been widely planted since the late 1990s in the United States and in a smattering of other large agricultural countries, including Brazil and Canada, the corn and soybean crops go mainly into animal feed, biofuels, and cooking oils. No genetically modified varieties of rice, wheat, or potatoes are widely grown, because opposition to such foods has discouraged investment in developing them and because seed companies haven’t found ways to make the kind of money on those crops that they do from genetically modified corn and soybeans.
Drought, damaging storms, and very hot days are already taking a toll on crop yields.
With the global population expected to reach more than nine billion by 2050, however, the world might soon be hungry for such varieties. Although agricultural productivity has improved dramatically over the past 50 years, economists fear that these improvements have begun to wane at a time when food demand, driven by the larger number of people and the growing appetites of wealthier populations, is expected to rise between 70 and 100 percent by midcentury. In particular, the rapid increases in rice and wheat yields that helped feed the world for decades are showing signs of slowing down, and production of cereals will need to more than double by 2050 to keep up. If the trend continues, production might be insufficient to meet demand unless we start using significantly more land, fertilizer, and water.
Climate change is likely to make the problem far worse, bringing higher temperatures and, in many regions, wetter conditions that spread infestations of disease and insects into new areas. Drought, damaging storms, and very hot days are already taking a toll on crop yields, and the frequency of these events is expected to increase sharply as the climate warms. For farmers, the effects of climate change can be simply put: the weather has become far more unpredictable, and extreme weather has become far more common.
The central highlands of Mexico, for example, experienced their driest and wettest years on record back to back in 2011 and 2012, says Matthew Reynolds, a wheat physiologist at the International Maize and Wheat Improvement Center in El Batán. Such variation is “worrisome and very bad for agriculture,” he says. “It’s extremely challenging to breed for it. If you have a relatively stable climate, you can breed crops with genetic characteristics that follow a certain profile of temperatures and rainfall. As soon as you get into a state of flux, it’s much more difficult to know what traits to target.”
One advantage of using genetic engineering to help crops adapt to these sudden changes is that new varieties can be created quickly. Creating a potato variety through conventional breeding, for example, takes at least 15 years; producing a genetically modified one takes less than six months. Genetic modification also allows plant breeders to make more precise changes and draw from a far greater variety of genes, gleaned from the plants’ wild relatives or from different types of organisms. Plant scientists are careful to note that no magical gene can be inserted into a crop to make it drought tolerant or to increase its yield—even resistance to a disease typically requires multiple genetic changes. But many of them say genetic engineering is a versatile and essential technique.
“It’s an overwhelmingly logical thing to do,” says Jonathan Jones, a scientist at the Sainsbury Laboratory in the U.K. and one of the world’s leading experts on plant diseases. The upcoming pressures on agricultural production, he says, “[are] real and will affect millions of people in poor countries.” He adds that it would be “perverse to spurn using genetic modification as a tool.”
It’s a view that is widely shared by those responsible for developing tomorrow’s crop varieties. At the current level of agricultural production, there’s enough food to feed the world, says Eduardo Blumwald, a plant scientist at the University of California, Davis. But “when the population reaches nine billion?” he says. “No way, José.”
Failed promises
The promise that genetically modified crops could help feed the world is at least as old as the commercialization of the first transgenic seeds in the mid-1990s. The corporations that helped turn genetically engineered crops into a multibillion-dollar business, including the large chemical companies Monsanto, Bayer, and DuPont, promoted the technology as part of a life science revolution that would greatly increase food production. So far it’s turned out, for a number of reasons, to have been a somewhat empty promise.
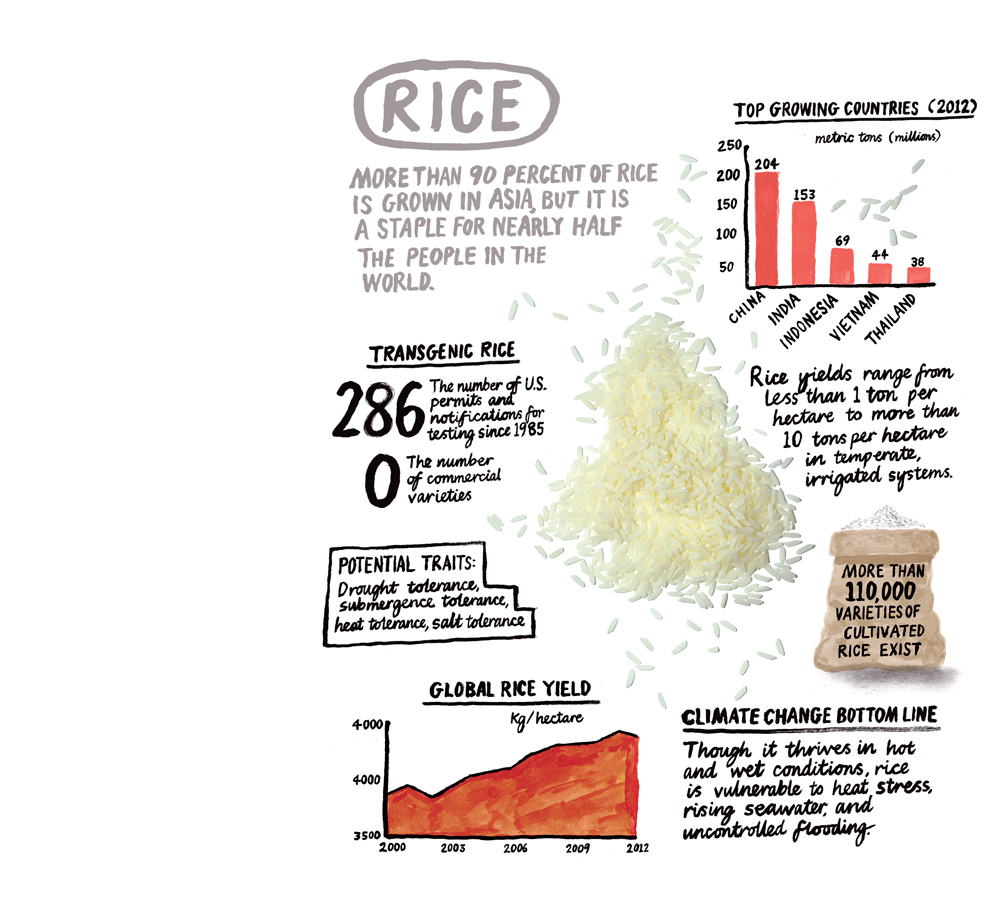
To be sure, bioengineered crops are a huge commercial success in some countries. The idea is simple but compelling: by inserting a foreign gene derived from, say, bacteria into corn, you can give the plant a trait it wouldn’t otherwise possess. Surveys estimate that more than 170 million hectares of such transgenic crops are grown worldwide. In the United States, the majority of corn, soybeans, and cotton planted have been engineered with a gene from the soil bacterium Bacillus thuringensis—Bt—to ward off insects or with another bacterial gene to withstand herbicides. Worldwide, 81 percent of the soybeans and 35 percent of the corn grown are biotech varieties. In India, Bt cotton was approved more than a decade ago and now represents 96 percent of the cotton grown in the country.
Yet it’s not clear whether that boom in transgenic crops has led to increased food production or lower prices for consumers. Take corn, for example. In the United States, 76 percent of the crop is genetically modified to resist insects, and 85 percent can tolerate being sprayed with a weed killer. Such corn has, arguably, been a boon to farmers, reducing pesticide use and boosting yields. But little of U.S. corn production is used directly for human food; about 4 percent goes into high–fructose corn syrup and 1.8 percent to cereal and other foods. Genetically modified corn and soybeans are so profitable that U.S. farmers have begun substituting them for wheat: around 56 million acres of wheat were planted in 2012, down from 62 million in 2000. As supply fell, the price of a bushel of wheat rose to nearly $8 in 2012, from $2.50 in 2000.
So far, the short list of transgenic crops used directly for food includes virus-resistant papaya grown in Hawaii, Bt sweet corn recently commercialized in the United States by Monsanto, and a few varieties of squash that resist plant viruses. That list could be about to grow, however. The Indonesian agricultural agency expects to approve a blight-resistant potato soon, and J. R. Simplot, an agricultural supplier based in Boise, Idaho, is hoping to commercialize its own version by 2017. Monsanto, which abandoned an attempt to develop GM wheat in 2004, bought a wheat-seed company in 2009 and is now trying again. And Cornell researchers are working with collaborators in India, Bangladesh, and the Philippines, countries where eggplant is a staple, to make an insect-resistant form of the vegetable available to farmers.
Only a handful of large companies can afford the risk and expense of commercializing GMOs.
These bioengineered versions of some of the world’s most important food crops could help fulfill initial hopes for genetically modified organisms, or GMOs. But they will also almost certainly heat up the debate over the technology. Opponents worry that inserting foreign genes into crops could make food dangerous or allergenic, though more than 15 years of experience with transgenic crops have revealed no health dangers, and neither have a series of scientific studies. More credibly, detractors suggest that the technology is a ploy by giant corporations, particularly Monsanto, to peddle more herbicides, dominate the agricultural supply chain, and leave farmers dependent on high-priced transgenic seeds. The most persuasive criticism, however, may simply be that existing transgenic crops have done little to guarantee the future of the world’s food supply in the face of climate change and a growing population.
The first generation of insect-resistant and herbicide-tolerant crops offer few new traits, such as drought tolerance and disease resistance, that could help the plants adapt to changes in weather and disease patterns, acknowledges Margaret Smith, a professor of plant breeding and genetics at Cornell University. Nonetheless, she says there is no valid reason to dismiss the technology as plant scientists race to increase crop productivity. Scientists are “facing a daunting breeding challenge,” Smith says. “We will need a second generation of transgenic crops. It would be a mistake to rule out this tool because the first products didn’t address the big issues.”
Developing crops that are better able to withstand climate change won’t be easy. It will require plant scientists to engineer complex traits involving multiple genes. Durable disease resistance typically requires a series of genetic changes and detailed knowledge of how pathogens attack the plant. Traits such as tolerance to drought and heat are even harder, since they can require basic changes to the plant’s physiology.
Is genetic engineering up to the task? No one knows. But recent genomic breakthroughs are encouraging. Scientists have sequenced the genomes of crops such as rice, potatoes, bananas, and wheat. At the same time, advances in molecular biology mean that genes can be deleted, modified, and inserted with far greater precision. In particular, new genome engineering tools known as Talens and Crispr allow geneticists to “edit” plant DNA, changing chromosomes exactly where they want.
Exact Edits
The workshop adjacent to the rows of greenhouses at the edge of Cornell’s campus in Ithaca, New York, smells musty and damp from the crates of potatoes. It is less than a mile from the university’s molecular biology labs, but what you see are wooden conveyer belts, wire screens, and water hoses. Walter De Jong is sorting and sizing harvested potatoes as part of a multiyear effort to come up with yet a better variety for the region’s growers. Boxes are filled with potatoes—some small and round, others large and misshapen. Asked what traits are important to consumers, he smiles slyly and says, “Looks, looks, looks.”
The question of how he feels about efforts to develop transgenic potatoes is not as easily answered. It’s not that De Jong is opposed to genetic engineering. As a potato breeder, he’s well versed in conventional methods of introducing new traits, but he also has a PhD in plant pathology and has done considerable research in molecular biology; he knows the opportunities that advanced genetics opens up. In the northeastern United States, a variety of potato is optimized for about a 500-mile radius, taking into account the length of the growing season and the type of weather in the area. Climate change means these growing zones are shifting, making crop breeding like solving a puzzle in which the pieces are moving around. The speed offered by genetic modification would help. But, De Jong says dismissively, “I don’t expect to use [transgenic] technology. I can’t afford it.”

“It’s a curious situation,” he says. Scientists at public and academic research institutions have done much of the work to identify genes and understand how they can affect traits in plants. But the lengthy testing and regulatory processes for genetically modified crops, and the danger that consumers will reject them, mean that only “a handful of large companies” can afford the expense and risk of developing them, he says.
But De Jong suddenly becomes animated when he’s asked about the newest genome engineering tools. “This is what I have been waiting my whole career for,” he says, throwing his hands up. “As long as I have been a potato scientist, I’ve wanted two things: a sequenced potato genome and the ability to modify the genome at will.” Across campus, De Jong also runs a molecular biology lab, where he has identified the DNA sequence responsible for red pigment in potato tubers. Soon, it could be possible to precisely alter that sequence in a potato cell that can then be grown into a plant: “If I had a white potato I wanted to turn red, I could just edit one or two nucleotides and get the color I want.” Plant breeding “is not the art of shuffling genes around,” De Jong explains. “Basically, all potatoes have the same genes; what they have is different versions of the genes—alleles. And alleles differ from one another in a few nucleotides. If I can edit the few nucleotides, why breed for [a trait]? It’s been the holy grail in plant genetics for a long time.”
One problem with conventional genetic engineering techniques is that they add genes unpredictably. The desired gene is inserted into the targeted cell in a petri dish using either a plant bacterium or a “gene gun” that physically shoots a tiny particle covered with the DNA. Once the molecules are in the cell, the new gene is inserted into the chromosome randomly. (The transformed cell is grown in a tissue culture to become a plantlet and eventually a plant.) It’s impossible to control just where the gene gets added; sometimes it ends up in a spot where it can be expressed effectively, and sometimes it doesn’t. What if you could precisely target spots on the plant’s chromosome and add new genes exactly where you want them, “knock out” existing ones, or modify genes by switching a few specific nucleotides? The new tools allow scientists to do just that.
Talens, one of the most promising of these genome engineering tools, was inspired by a mechanism used by a bacterium that infects plants. Plant pathologists identified the proteins that enable the bacterium to pinpoint the target plant DNA and found ways to engineer these proteins to recognize any desired sequence; then they fused these proteins with nucleases that cut DNA, creating a precise “editing” tool. A plant bacterium or gene gun is used to get the tool into the plant cell; once inside, the proteins zero in on a specific DNA sequence. The proteins deliver the nucleases to an exact spot on the chromosome, where they cleave the plant’s DNA. Repair of the broken chromosome allows new genes to be inserted or other types of modifications to be made. Crispr, an even newer version of the technology, uses RNA to zero in on the targeted genes. With both Talens and Crispr, molecular biologists can modify even a few nucleotides or insert and delete a gene exactly where they want on the chromosome, making the change far more predictable and effective.
One implication of the new tools is that plants can be genetically modified without the addition of foreign genes. Though it’s too early to tell whether that will change the public debate over GMOs, regulatory agencies—at least in the United States—indicate that crops modified without foreign genes won’t have to be scrutinized as thoroughly as transgenic crops. That could greatly reduce the time and expense it takes to commercialize new varieties of genetically engineered foods. And it’s possible that critics of biotechnology could draw a similar distinction, tolerating genetically modified crops so long as they are not transgenic.
Dan Voytas, director of the genome engineering center at the University of Minnesota and one of Talens’s inventors, says one of his main motivations is the need to feed another two billion people by the middle of the century. In one of his most ambitious efforts, centered at the International Rice Research Institute in Los Baños, the Philippines, he is collaborating with a worldwide network of researchers to rewrite the physiology of rice. Rice and wheat, like other grains, have what botanists call C3 photosynthesis, rather than the more complex C4 version that corn and sugarcane have. The C4 version of photosynthesis uses water and carbon dioxide far more efficiently. If the project is successful, both rice and wheat yields could be increased in regions that are becoming hotter and drier as a result of climate change.
Rewriting the core workings of a plant is not a trivial task. But Voytas says Talens could be a valuable tool—both to identify the genetic pathways that might be tweaked and to make the many necessary genetic changes.
The pressure to help feed the growing population at a time when climate change is making more land marginal for agriculture is “the burden that plant biologists bear,” Voytas says. But he’s optimistic. Over much of the last 50 years, he points out, crop productivity has made repeated gains, attributable first to the use of hybrid seeds, then to the new plant varieties introduced during the so-called Green Revolution, and “even to the first GM plants.” The introduction of the new genome engineering tools, he says, “will be another inflection point.”
If he’s right, it might be just in time.
Heat Wave
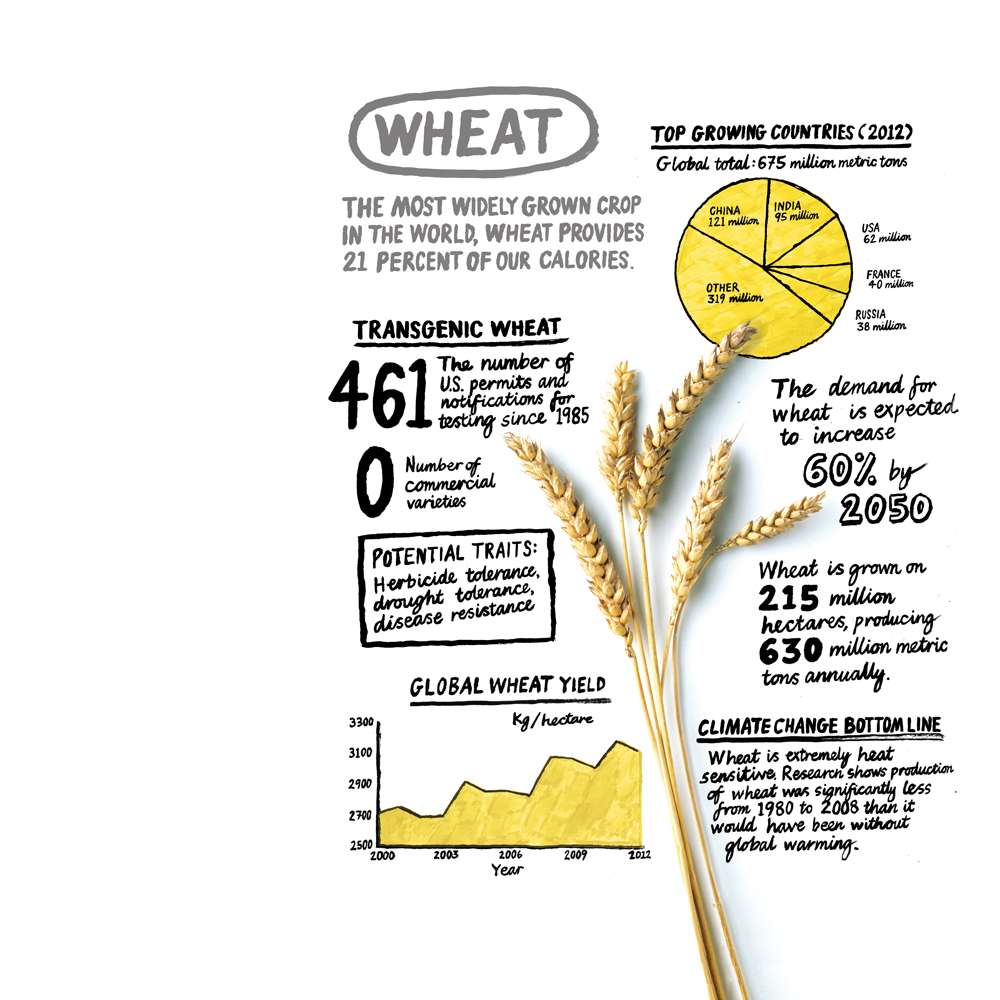
For agronomists, plant breeders, and farmers, it’s all about yield—the amount a crop produces in a hectare. The remarkable increases in crop yields beginning in the middle of the 20th century are the main reason that we have enough food to go from feeding three billion people in 1960 to feeding seven billion in 2011 with only a slight increase in the amount of land under cultivation. Perhaps most famously, the Green Revolution spearheaded by the Iowa-born plant pathologist and geneticist Norman Borlaug substantially increased yields of wheat, corn, and rice in many parts of the world. It did so, in part, by introducing more productive crop varieties, starting in Mexico and then in Pakistan, India, and other countries. But for at least the past decade, increases in the yields of wheat and rice seem to have slowed. Yields of wheat, for example, are growing at roughly 1 percent annually; they need to increase nearly 2 percent annually to keep up with food demand over the long term. Agricultural experts warn that yields will have to improve for other crops as well if we are to feed a rapidly growing population—and yet rising temperatures and other effects of global climate change will make this tougher to achieve.
David Lobell, a professor of environmental earth system science at Stanford University, has a calm demeanor that belies his bleak message about how global warming is already affecting crops. The effects of climate change on agriculture have been widely debated, but recently Lobell and his collaborators have clarified the projections by combing through historical records of weather and agricultural production. They found that from 1980 to 2008, climate change depressed yields of wheat and corn; yields still rose during that time, but overall production was 2 to 3 percent less than it would have been if not for global warming. This has held true across most of the regions where corn and wheat are grown.
The finding is startling because it suggests that global warming has already had a significant impact on food production and will make an even bigger difference as climate change intensifies. “Anything that causes yield [growth] to flatten out is a concern,” says Lobell. And while overall yields of wheat and corn are still increasing, he says, “climate change becomes a concern long before you have negative yield trends.”
Even more disturbing, Lobell and his collaborator Wolfram Schlenker, an economist at Columbia University, have found evidence that in the case of several important crops, the negative effect of global warming is more strongly tied to the number of extremely hot days than to the rise in average temperatures over a season. If that’s true, earlier research might have severely underestimated the impact of climate change by looking only at average temperatures.
Schlenker’s calculations show steady increases in corn and soybean yields as the temperature rises from 10 °C into the 20s—but at around 29 °C for corn and 30 °C for soybeans, the crops are “hit hard” and yields drop dramatically. In subsequent work, Lobell showed that hot days were doing far more damage to wheat in northern India than previously thought.
Agricultural yields will have to improve if we are to feed a rapidly growing population.
A surprising and troubling detail of the research, says Schlenker, is that crops and farmers don’t seem to have adapted to the increased frequency of hot days. “What surprised me most and should inform us going forward,” he says, “is that there has been tremendous progress in agricultural breeding—average yields have gone up more than threefold since the 1950s—but if you look at sensitivity to extreme heat, it seems to be just as bad as it was in the 1950s. We need to have crops that are better at dealing with hot climates.” During the heat wave that hit much of the United States in 2012, he says, yields of corn were down 20 percent, and “2012 is not that unusual a year compared to what the climate models predict will be a new normal pretty soon.”
It’s possible that plants are simply hardwired to shut down at temperatures above 30 °C. Indeed, Schlenker says he’s not convinced that crops can be engineered to adapt to the increased frequency of hot days, though he hopes he’s wrong. Likewise, Lobell wants his work to better define which aspects of climate change are damaging crops and thus help target the needed genetic changes. But, like Schlenker, he is unsure whether genetics can provide much of an answer.
In California’s Central Valley, one of the world’s most productive agricultural areas, UC Davis’s Blumwald acknowledges that scientists have “never bred for stresses” like drought and heat. But he aims to change that. Inserting a combination of genes for tolerance to heat, drought, and high soil salinity into rice and other plants, Blumwald is creating crops that have at least some advantages during extreme weather conditions, particularly during key times in their growth cycle.
The challenge is to avoid reducing yields under good growing conditions. So Blumwald has identified a protein that activates the inserted genes only under adverse conditions. “There’s no cure for drought. If there’s no water, the plant dies. I’m not a magician,” he says. “We just want to delay the stress response as long as possible in order to maintain yields until the water comes.”
Daily Bread
A field just north of London on the grounds of Rothamsted Research, which bills itself as the world’s longest-running agricultural research station (founded in 1843), is one of the focal points of Europe’s continuing battle over genetically modified foods. The controversy here is over an 80-by-80-meter field of wheat, some of it genetically modified to produce a hormone that repels aphids, a common insect pest. In 2012, a protester climbed a low fence and scattered conventional wheat seeds among the GM plants in an attempt to sabotage the trial. The scientists at Rothamsted had the seeds vacuumed up, hired several extra security guards, and built a second fence, this one three meters high and topped with a curved overhang to keep it from being scaled. Later, a few hundred protesters marched arm in arm to the edge of the fenced-in field before they were stopped by the police.
The fuss at Rothamsted is just one hint that the next great GMO controversy could involve transgenic wheat. After all, wheat is the world’s most widely planted crop, accounting for 21 percent of the calories consumed globally. Meddling with a grain that makes the daily bread for countless millions around the world would be particularly offensive to many opponents of genetically modified foods. What’s more, wheat is a commodity grain sold in world markets, so approval of GM wheat in a leading exporting country would likely have repercussions for food markets everywhere.
Wheat is also emblematic of the struggles facing agriculture as it attempts to keep up with a growing population and a changing climate. Not only have the gains in yield begun to slow, but wheat is particularly sensitive to rising temperatures and is grown in many regions, such as Australia, that are prone to severe droughts. What’s more, wheat is vulnerable to one of the world’s most dreaded plant diseases: stem rust, which is threatening the fertile swath of Pakistan and northern India known as the Indo-Gangetic Plain. Conventional breeding techniques have made remarkable progress against these problems, producing varieties that are increasingly drought tolerant and disease resistant. But biotechnology offers advantages that shouldn’t be ignored.
“Climate change doesn’t change [the challenge for plant breeders], but it makes it much more urgent,” says Walter Falcon, deputy director of the Center on Food Security and the Environment at Stanford. Falcon was one of the foot soldiers of the Green Revolution, working in the wheat-growing regions of Pakistan and in Mexico’s Yaqui Valley. But he says the remarkable increases in productivity achieved between 1970 and 1995 have largely “played out,” and he worries about whether the technology–intensive farming in those regions can be sustained. He says the Yaqui Valley remains highly productive—recent yields of seven tons of wheat per hectare “blow your mind”—but the heavy use of fertilizers and water is “pushing the limits” of current practices. Likewise, Falcon says he is worried about how climate change will affect agriculture in the Indo-Gangetic Plain, the home of nearly a billion people.
Asked whether transgenic technology will solve any of these problems, he answers, “I’m not holding my breath,” citing both scientific reasons and opposition to GM crops. But he does expect advances in genetic technologies over the next decade to create wheat varieties that are better equipped to withstand pests, higher temperatures, and drought.
It is quite possible that the first and most dramatic of the advances will come in adapting crops to the shifting patterns of disease. And as Teagasc’s Ewen Mullins puts it, “if you want to study plant diseases, you come to Ireland.”
A hundred kilometers from the idyllic fields in Carlow, Fiona Doohan, a plant pathologist at University College Dublin, is developing wheat varieties that stand up to local diseases and trying to understand how plant pathogens might evolve with climate change. At the school’s agricultural experiment station, she uses growing chambers in which the concentration of carbon dioxide can be adjusted to mimic the higher levels expected in 2050. The experiments have yielded a nasty surprise. When wheat and the pathogens that commonly afflict it are put in the chamber with the increased levels of carbon dioxide, the plant remains resistant to the fungus. But when both are separately grown through several generations under 2050 conditions and then placed together, Doohan says, the plants “crash.” This suggests, ominously, that plant pathogens might be far better and faster than wheat at adapting to increased carbon dioxide.
Next to the building is an apple orchard with representatives of trees grown all over Ireland, including heirloom varieties that have been planted for centuries. Doohan looks at them fondly as she walks past, the ground covered by fallen apples. At the far end of the orchard is a row of greenhouses, including a small one in which genetically modified plants are tested. Inside is a particularly promising transgenic wheat that is proving resistant to the types of scab disease common in Ireland. The new gene is also increasing the plant’s grain production, says Doohan, who created the variety with her colleagues. She’s clearly delighted by the results. But, she quickly adds, there are no plans to test the GM wheat out in the field in Ireland, or anywhere else in Europe. At least for now, the promising variety of wheat is doomed to stay in the greenhouse.
Deep Dive
Biotechnology and health
How scientists traced a mysterious covid case back to six toilets
When wastewater surveillance turns into a hunt for a single infected individual, the ethics get tricky.
An AI-driven “factory of drugs” claims to have hit a big milestone
Insilico is part of a wave of companies betting on AI as the "next amazing revolution" in biology
The quest to legitimize longevity medicine
Longevity clinics offer a mix of services that largely cater to the wealthy. Now there’s a push to establish their work as a credible medical field.
There is a new most expensive drug in the world. Price tag: $4.25 million
But will the latest gene therapy suffer the curse of the costliest drug?
Stay connected
Get the latest updates from
MIT Technology Review
Discover special offers, top stories, upcoming events, and more.